Clear Sky Science · en
Cacna1c deficiency in parvalbumin-expressing neurons promotes anxiety and passive stress-coping behavior
Why tiny brain switches matter for stress and anxiety
Anxiety and how we cope with stress can feel like products of personality or life experience alone. Yet deep inside the brain, specialized cells act as microscopic switches that help decide whether we freeze, give up, or actively fight through a challenge. This study explores one such switch: a calcium channel protein called Cav1.2, produced by the CACNA1C gene, in a particular set of inhibitory brain cells. By turning this protein off only in those cells in mice, the researchers reveal how subtle shifts in brain wiring can tilt behavior toward anxiety and passive coping, offering clues for future, more targeted treatments for psychiatric disorders.
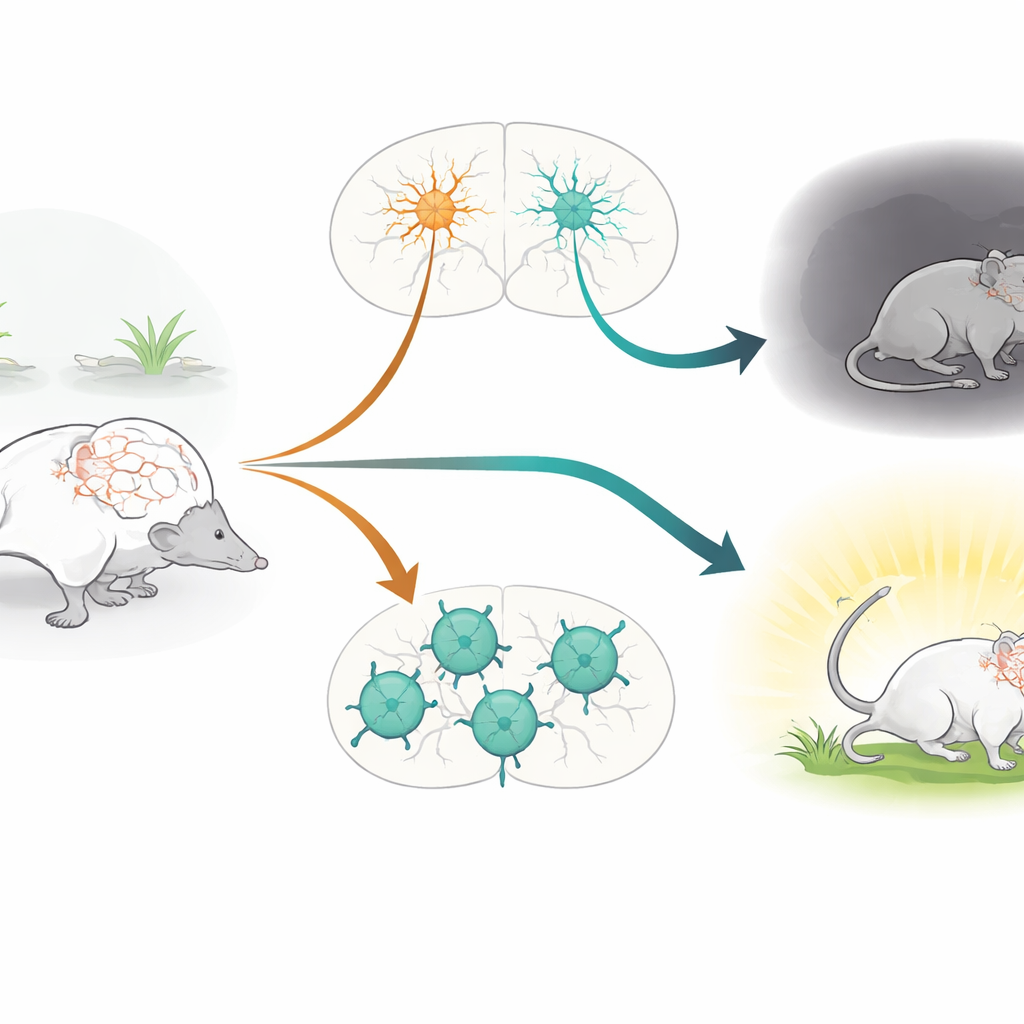
The brain’s rhythm keepers
The work focuses on parvalbumin-positive (PV+) neurons, a class of fast-firing inhibitory cells that act as rhythm keepers in the brain. These cells help maintain a delicate balance between excitation and inhibition, shaping brain rhythms that underlie attention, memory, and emotion. PV+ neurons have long been implicated in conditions such as schizophrenia, depression, autism, and epilepsy. At the same time, the CACNA1C gene, which encodes the Cav1.2 calcium channel, is one of the most consistently identified genetic risk factors across multiple psychiatric disorders. Cav1.2 channels allow calcium ions to flow into cells when they are active, influencing how they develop, connect, and adjust their strength over time. Despite this, the specific role of Cav1.2 within PV+ neurons had not been directly tested in living animals.
Engineering mice with targeted changes
To isolate this question, the researchers engineered mice in which Cav1.2 was selectively removed only from PV+ neurons, leaving other cell types untouched. They used a genetic system that activates a “cutting” enzyme (Cre) specifically in PV+ cells, deleting the Cacna1c gene there. A molecular tag was added to ribosomes to verify where this recombination occurred, confirming that PV-rich regions such as cortex, hippocampus, amygdala, and cerebellum were effectively targeted. The mice then underwent an extensive behavioral test battery that assessed basic movement, anxiety-like behavior, social interaction, learning and memory, and how they responded to an inescapable stressor.
Anxious but mentally intact
Mice lacking Cav1.2 in PV+ neurons showed a clear increase in anxiety-like behavior. In standard tests where animals choose between safe, dark or sheltered areas and more exposed, brightly lit spaces, these modified mice avoided the open or lit zones more strongly than their normal littermates, even though their overall movement levels were unchanged. Strikingly, however, these same mice performed normally on a wide range of tasks probing social preference, recognition of new objects, spatial memory, and learning the location of a hidden platform in a water maze. This suggests that, at least in this model, Cav1.2 in PV+ neurons is particularly important for emotional regulation and stress responses, rather than for social or cognitive abilities.
Opposite stress styles from different cell types
When placed in a forced swim test, which is often used to study stress-coping styles, PV-specific Cav1.2 knockout mice adopted a more passive strategy: they spent less time actively swimming and more time immobile. Earlier work from the same group had shown the opposite effect when Cav1.2 was deleted in excitatory glutamatergic neurons: those mice became more actively engaged in coping with the stressful situation. Direct comparison of the two models confirmed this bidirectional pattern. To connect behavior with brain activity, the team measured cFos, a marker of recently activated neurons, after the swim test. Loss of Cav1.2 in PV+ neurons led to heightened activity in areas such as the nucleus accumbens, lateral habenula, and paraventricular thalamus—regions linked to negative mood and stress processing—while deletion in excitatory neurons instead boosted activity in the lateral septum, a region involved in emotional regulation and resilience.
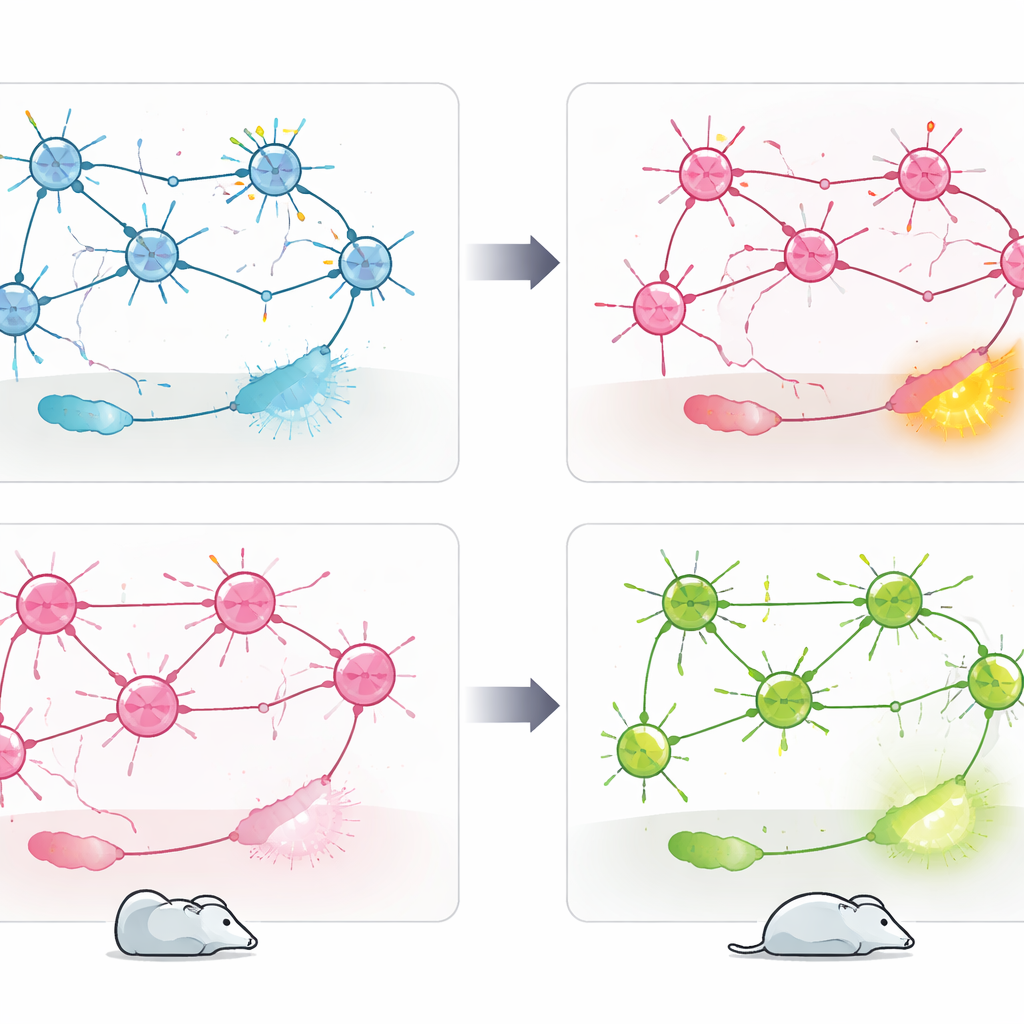
What this means for future treatments
Taken together, the findings show that the same risk gene, CACNA1C, can push stress behavior in opposite directions depending on which cell type is affected. Cav1.2 in PV+ neurons appears to act as a brake on anxiety and passive coping, while its role in excitatory neurons supports a different, more active style of responding to stress. Rather than a single “good” or “bad” gene, CACNA1C emerges as a context-dependent regulator embedded in distinct brain circuits. For people living with psychiatric disorders linked to this gene, these results hint that future therapies might need to target specific cell populations or circuit nodes, instead of broadly blocking or enhancing Cav1.2 throughout the brain.
Citation: Loganathan, S., Zhao, C. & Deussing, J.M. Cacna1c deficiency in parvalbumin-expressing neurons promotes anxiety and passive stress-coping behavior. Sci Rep 16, 12870 (2026). https://doi.org/10.1038/s41598-026-48841-4
Keywords: anxiety, stress coping, parvalbumin neurons, CACNA1C, mouse behavior