Clear Sky Science · en
Gut microbiota-dependent protection by Lactobacillus reuteri against renal injury in brain-dead rats
Why this research matters
Most kidneys used for transplants come from donors who have suffered brain death, but these organs are often damaged before surgery even begins. This study in rats explores an unexpected ally living in the gut: a probiotic bacterium called Lactobacillus reuteri. The researchers show how this microbe can help shield the kidneys from injury after brain death by stabilizing gut bacteria, tightening the gut barrier, and calming harmful immune reactions that would otherwise weaken transplant-ready organs.
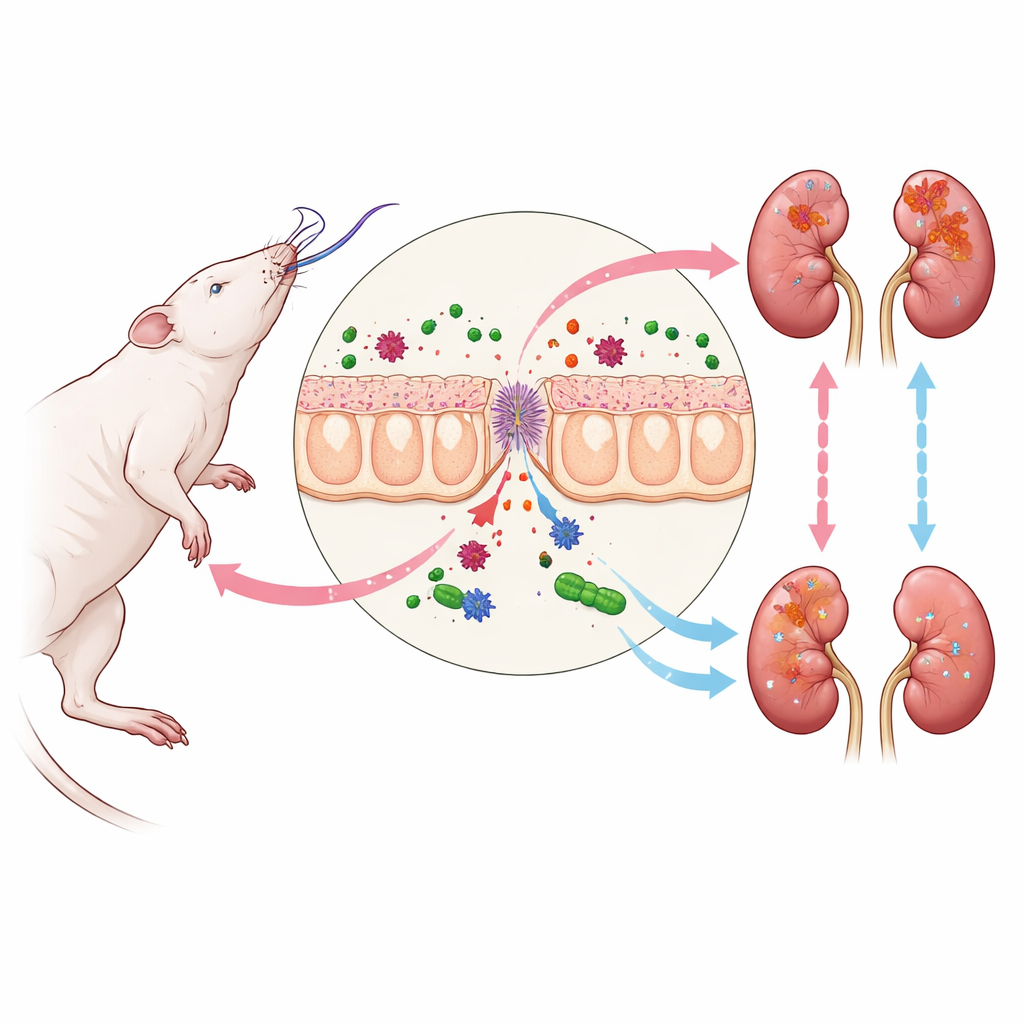
Brain death and the chain reaction to the kidneys
When brain death occurs, the body experiences a storm of hormonal and circulatory changes that ripple through many organs. One of the first weak points is the intestine. Its protective lining becomes leaky, allowing bacterial fragments from gut microbes to slip into the bloodstream. Among these fragments is LPS, a component of certain bacterial cell walls that strongly activates the immune system. As LPS travels to the kidneys, it locks onto a sensor on kidney cells that triggers the TLR4–NF-kappaB pathway, a signaling route well known for driving inflammation. In the rat model used here, this process led to higher blood levels of creatinine and urea, increased expression of the kidney damage marker KIM-1, and subtle structural damage in kidney tubule cells, even when standard microscopic views looked almost normal.
A probiotic rewires the gut environment
The team then asked whether feeding rats Lactobacillus reuteri for a week before brain death could change this story. They found that the probiotic reshaped the gut’s microbial community: harmful, LPS-rich Proteobacteria were reduced, while Bacteroidota and other potentially beneficial groups became more abundant. At the same time, the physical barrier of the intestine improved. Under the microscope and in electron micrographs, treated animals showed more intact cell junctions between intestinal cells and less widening of the spaces where leaks form. Blood levels of iFABP, a marker of gut injury and permeability, dropped back toward normal, indicating a tighter, more selective intestinal wall.
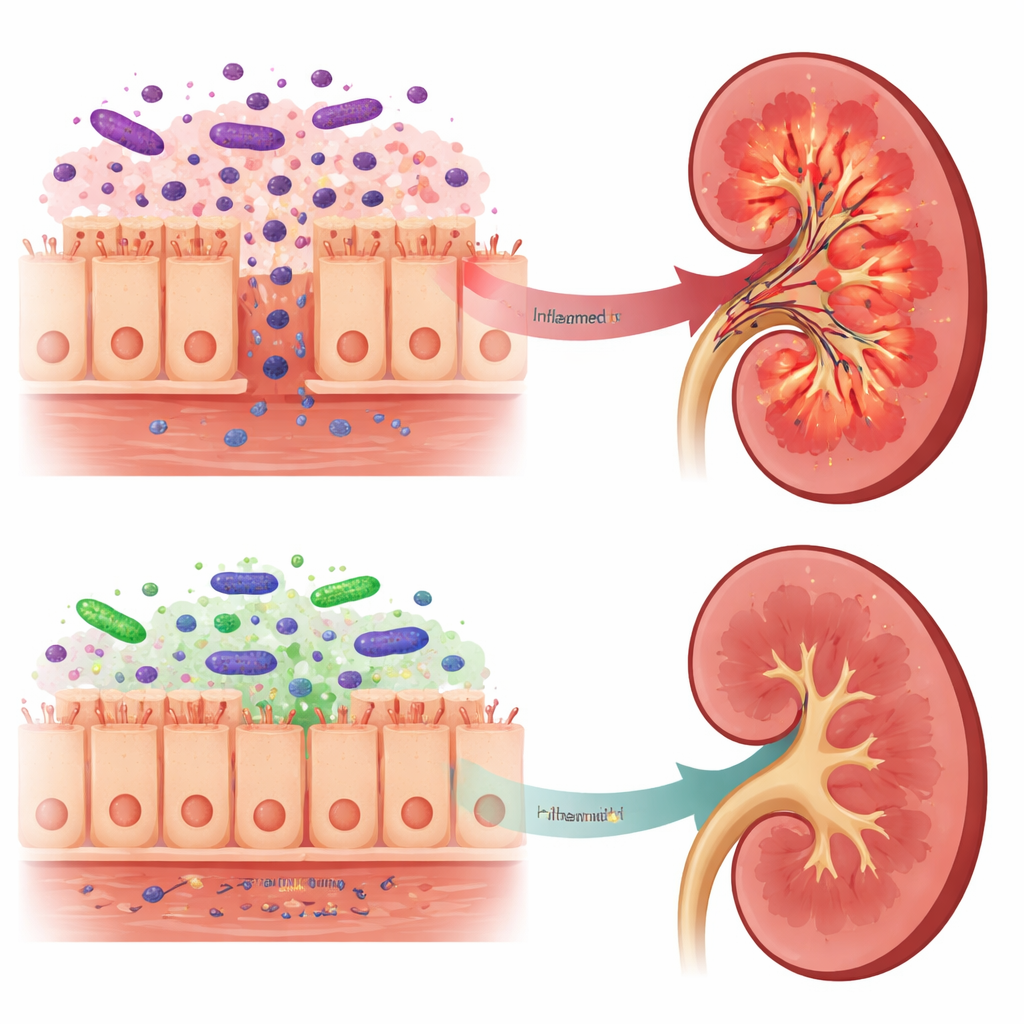
From toxic signals to gentler messages
These shifts in the gut were mirrored by changes in what reached the kidneys. In untreated brain-dead rats, kidney tissue contained more LPS, higher levels of TLR4 and its helper protein LBP, and stronger activation of the NF-kappaB pathway, all signs of a robust inflammatory response. By contrast, rats pretreated with Lactobacillus reuteri had lower kidney LPS, weaker TLR4 signaling, and reduced production of several inflammatory molecules, including IL-6 and IFN-gamma. Interestingly, total LPS in the blood actually rose in the probiotic group, but analysis of the gut microbes suggested that much of this LPS came from Bacteroidota, whose LPS structures are thought to be far less inflammatory and may even act as partial blockers of the TLR4 sensor. The authors propose that this “gentler” LPS competes with the more damaging form, reducing the kidney’s exposure to the most toxic signals.
Balancing the immune response along the gut–kidney route
The probiotic did more than just alter microbes and barriers; it also tuned the immune system on both sides of the gut–kidney connection. In the intestine, markers of activated immune cells such as CD4 and CD25 were elevated after brain death, suggesting an overactive response at the mucosal surface. Lactobacillus reuteri scaled back this activation in the lining itself but increased CD25-positive cells in the underlying glands, hinting at a more controlled and localized defense. In the kidneys, the probiotic reduced signs of neutrophil buildup and oxidative stress, while selectively boosting the chemokine CXCL2, which may help coordinate a more balanced repair response. Correlation analyses linked specific Bacteroidota members with these immune changes, supporting the idea of a gut-driven immune network that helps protect the kidneys.
What this could mean for future transplants
Taken together, the findings point to a dual protective role for Lactobacillus reuteri in brain-dead donor rats: it rebuilds a healthier gut community and barrier while also reshaping immune and inflammatory signals that reach the kidneys. As a result, kidney cells face less exposure to highly inflammatory LPS and show better functional and structural health. Although the work is preclinical and relies on pretreatment before brain death, it highlights the gut and its microbes as promising targets for strategies to improve organ quality before transplantation, potentially giving future recipients a better starting point for long-term kidney function.
Citation: Liu, T., Zhu, G., Li, H. et al. Gut microbiota-dependent protection by Lactobacillus reuteri against renal injury in brain-dead rats. Sci Rep 16, 14709 (2026). https://doi.org/10.1038/s41598-026-47230-1
Keywords: gut microbiota, probiotics, kidney injury, brain death donors, Lactobacillus reuteri