Clear Sky Science · en
Pathogenic mechanism of the PLS1 gene variant in hearing loss and functional validation in a zebrafish model
Why tiny ear structures matter
Hearing depends on rows of microscopic hair like projections deep inside the inner ear. When sound vibrations bend these hairs, they open channels that let charged particles flow into the cells, creating the signals our brains interpret as sound. This study looks at how a single genetic change in a protein that helps shape these hair bundles can disturb that process and lead to inherited hearing loss, using both human cells and zebrafish as models.
A gene behind quieted sound
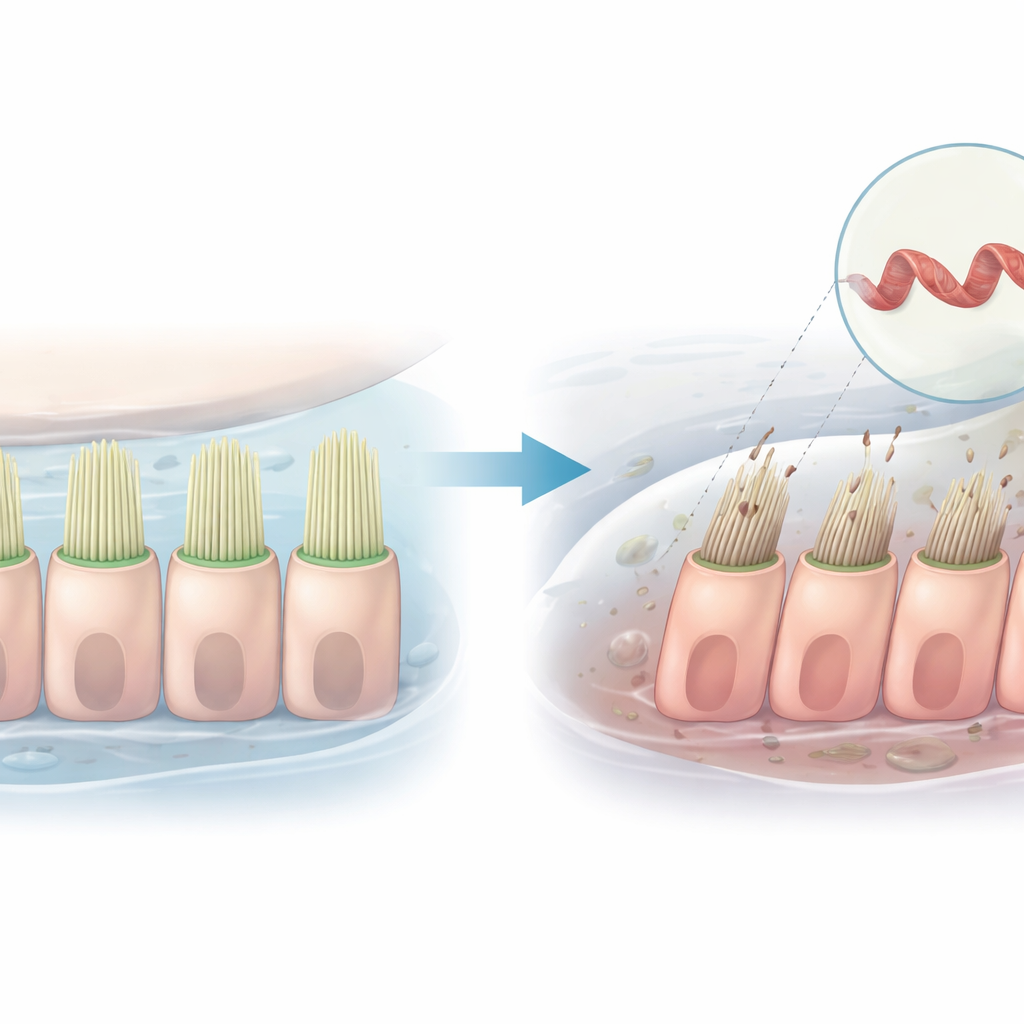
The research focuses on a gene called PLS1, which provides instructions for making plastin 1, a protein that bundles together slender strands inside the hair like projections of inner ear cells. Families with a form of progressive, non syndromic hearing loss have been found to carry specific PLS1 variants. In one Chinese family, a change in the gene causes the loss of a small segment, known as exon 8. Affected family members start to lose high pitched hearing in childhood, and their hearing worsens with age, but the cellular reason for this decline had not been clear.
How a structural change ripples through the cell
Plastin 1 has two main regions that grab onto internal strands, and under normal conditions one region keeps the other in check so that the bundles remain flexible and orderly. The team created versions of plastin 1 with and without exon 8 and expressed them in cultured cells. They found that the exon 8 deletion disrupts the normal self interaction between these two regions. As a result, the mutant protein binds differently to the internal strands and to partner proteins that help control movement and tension. Three dimensional imaging of the internal scaffolding showed that cells with the mutant protein had fewer, shorter, and thinner bundles, and their overall shape became more rounded, pointing to a disturbed internal architecture.
Consequences for stability and partnerships
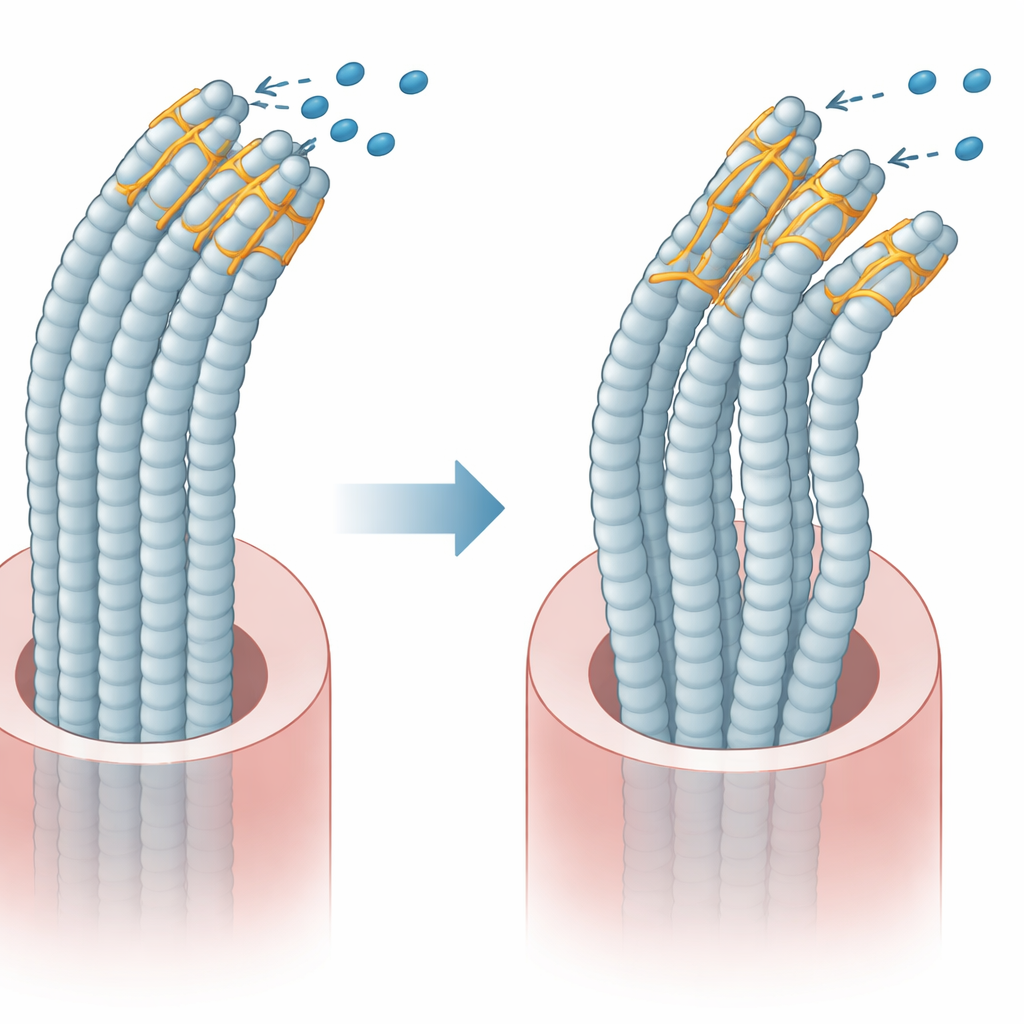
The scientists also learned that the mutant plastin 1 is more heavily tagged for disposal by the cell’s waste system, which means it is broken down more quickly and does not accumulate to normal levels. At the same time, the mutant form clings more tightly to certain key partners, including a common strand forming protein and a motor protein called MYO1C that helps adjust tension at the tips of the hair like projections. In effect, the altered plastin 1 both loses some of its usual function and interferes with the fine tuning of the mechanical system that opens and closes the channels sensing sound.
Lessons from zebrafish ears and behavior
To see how these cellular changes play out in a living ear, the researchers turned to zebrafish, whose inner ear structures and genes are remarkably similar to our own. They used gene editing to create fish that completely lack the fish version of PLS1 and then tried to rescue this loss by adding either normal human PLS1 or the exon 8 deleted version. Fish lacking PLS1 showed mild changes in the size and shape of their inner ear sacs and the tiny stones inside that help sense movement, and these defects were mostly corrected by the normal human gene. However, fish given the mutant human gene continued to show structural abnormalities and did not swim as vigorously in response to sound, suggesting reduced hearing like responses.
Channels that no longer open properly
The team then asked whether the key channels on the hair cells were working correctly. They used a fluorescent dye that can slip into hair cells only through open channels and measured how much dye entered. Fish expressing the mutant gene had noticeably lower dye uptake, meaning these channels were less likely to open. In contrast, fish that simply lacked PLS1, without the mutant form present, actually showed a slight increase in dye entry, as if their channels were more open than usual. This contrast suggests that the mutant protein actively interferes with normal channel behavior, rather than just removing helpful function.
What this means for inherited hearing loss
Taken together, the cell and zebrafish experiments point to a dual impact of the exon 8 PLS1 variant. The mutant plastin 1 is less stable and cannot fully perform its usual role in shaping and supporting the internal framework of hair cells, but it also binds abnormally to key partners and stiffens or misaligns the structures that control channel opening. This combination likely underlies the hearing loss seen in affected families. For future therapies, the work suggests that simply adding more normal PLS1 may not be enough; treatments may also need to silence or bypass the harmful mutant version to restore healthy sound detection.
Citation: Xu, T., Yang, T., Wang, H. et al. Pathogenic mechanism of the PLS1 gene variant in hearing loss and functional validation in a zebrafish model. Sci Rep 16, 14708 (2026). https://doi.org/10.1038/s41598-026-47079-4
Keywords: hereditary hearing loss, PLS1 gene, hair cells, zebrafish model, mechanotransduction