Clear Sky Science · en
Lipid nanoparticle delivery of circle RNA vaccine induces potent immune responses
Why a New Kind of Vaccine Matters
Vaccines based on genetic material have transformed how we fight infections like COVID-19, but today’s RNA shots can be fragile and short‑lived. This study explores a next‑generation approach that uses a sturdier form of RNA, packaged in tiny fat bubbles, to spur the immune system against both cancer and infectious diseases. For readers, it offers a glimpse of how future vaccines might become more powerful, longer lasting, and easier to store and use around the world. 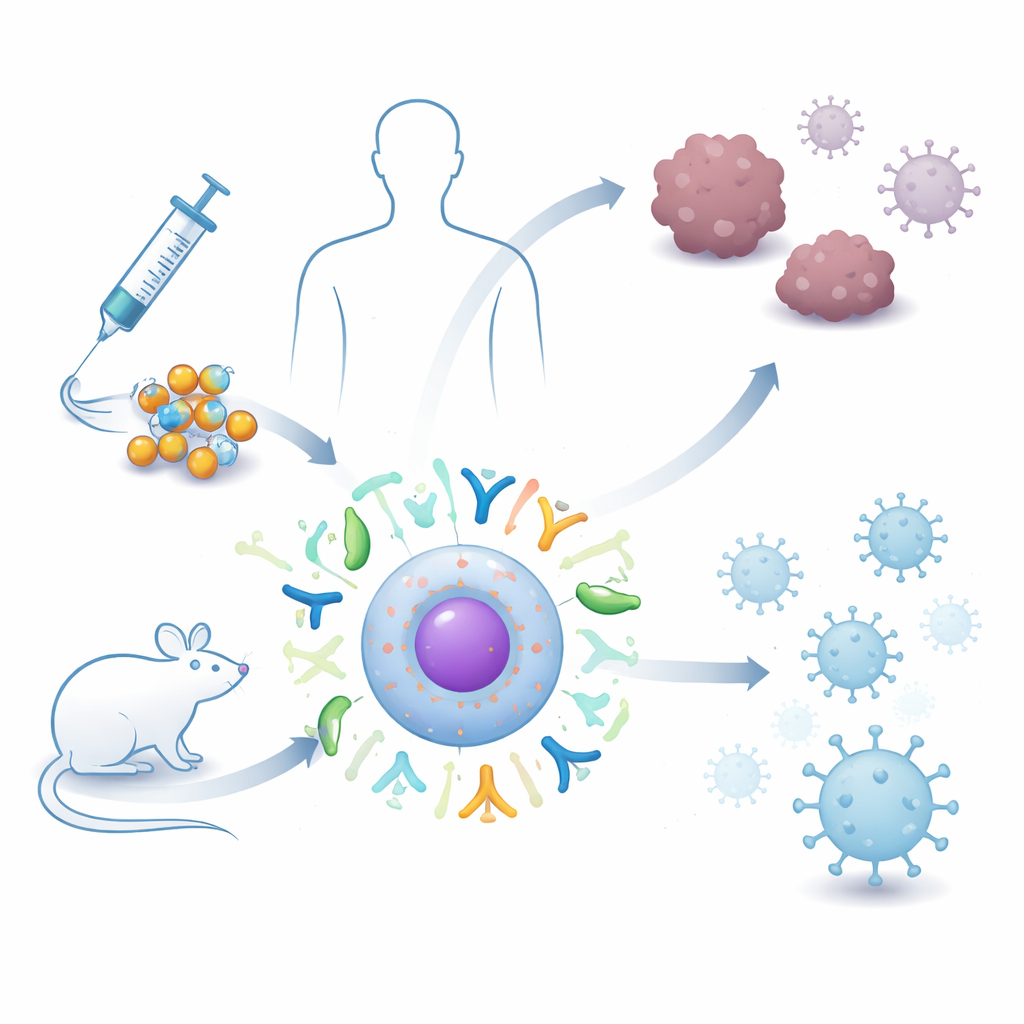
A Loop-Shaped Instruction Manual
Most people have heard of mRNA, the single‑stranded genetic message used in current RNA vaccines. Circle RNA, or circRNA, is different: instead of having two ends, it forms a closed loop. This simple change makes it much harder for the body’s enzymes to chew up, so it survives longer and can direct cells to produce proteins for an extended period. The authors explain that circRNA can be engineered to carry blueprints for tumor markers or viral pieces, and because it never enters the DNA in our chromosomes, it avoids the risk of permanently altering our genes. Combined with the ability to be manufactured in large quantities, these properties make circRNA an appealing candidate for a durable, flexible vaccine platform.
Tiny Fat Bubbles as Delivery Trucks
Getting fragile RNA safely into cells is a major hurdle. This is where lipid nanoparticles—tiny spheres made of fat‑like molecules—come in. They wrap around the circRNA, shielding it as it travels through the body and helping it slip into cells after injection. The researchers focused on studies in which circRNA was delivered to mice using these particles, through routes such as intramuscular, subcutaneous, intravenous, and intranasal injection. By reviewing and combining data from multiple independent animal experiments, they asked how well these circRNA–nanoparticle vaccines shrink tumors, control infections, and avoid side effects like weight loss or serious toxicity.
Stronger Immune Soldiers, Smaller Tumors
The combined animal data paint a striking picture. Across ten tumor studies, mice that received circRNA vaccines had much smaller tumors than control animals, regardless of whether the shot went into muscle, under the skin, or into the bloodstream. At the same time, body weight—a rough marker of overall health—remained stable, suggesting the treatment was not broadly toxic. Detailed immune measurements showed that key white blood cells became more active: T cells that directly kill abnormal cells expanded, macrophages increased, and dendritic cells—the sentinels that show fragments of invaders to the rest of the immune system—were markedly boosted. These changes indicate that the vaccines do not just attack tumors mechanically; they re‑educate the immune system to recognize and destroy cancer cells more efficiently. 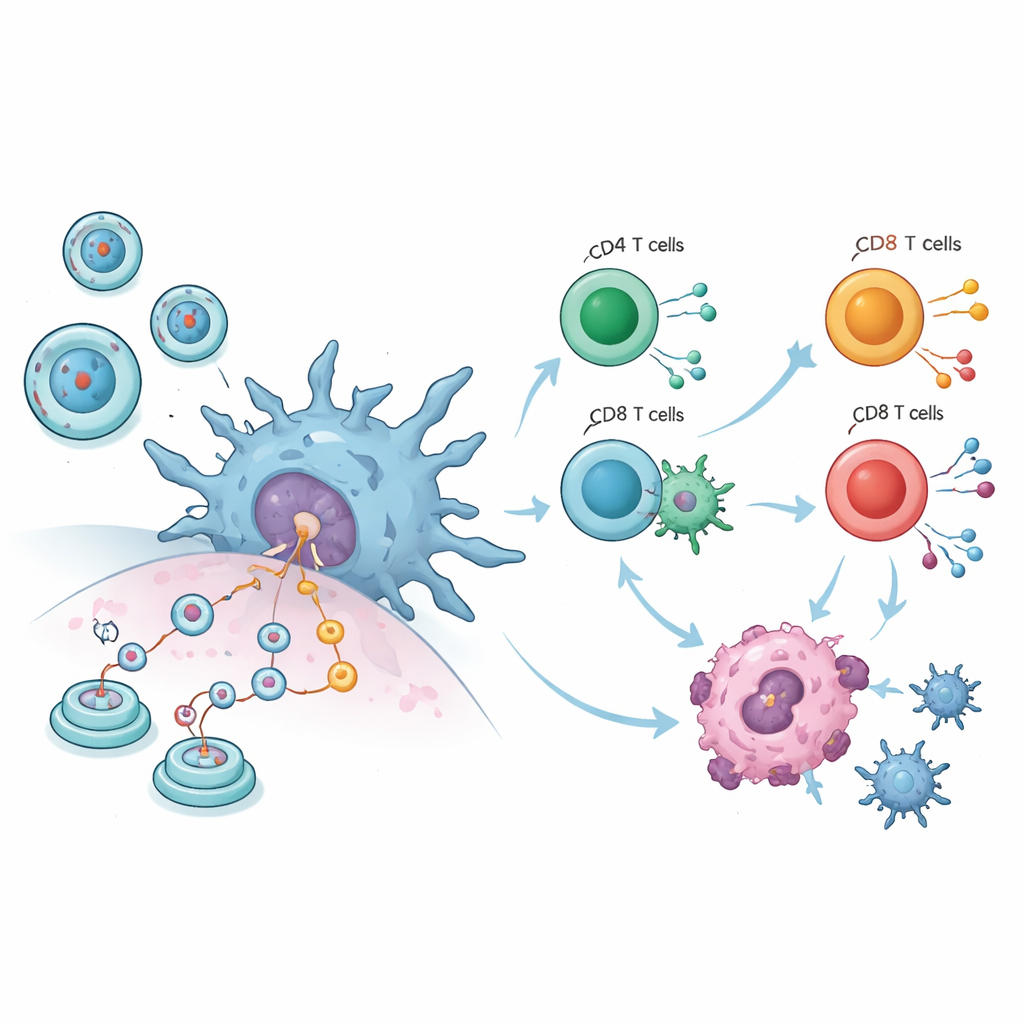
Fighting Viruses with the Same Tool
The same circRNA technology also proved powerful in infectious disease models, including influenza, Zika, SARS‑CoV‑2 and other viruses. In vaccinated mice, antibody levels in the blood rose sharply, lung viral loads dropped, and signaling molecules like interferon‑gamma and interleukin‑4 increased, showing that both rapid and longer‑term arms of the immune system were engaged. Intranasal delivery, which targets the lining of the airways, often produced especially strong local protection, while other routes favored broader responses through the whole body. Overall, the analysis suggests that a single circRNA platform, tuned to different targets, could be adapted to protect against a wide range of pathogens as well as cancers.
Checking the Mechanism in Human Cells
To move beyond animal data, the authors ran laboratory experiments using human blood‑derived immune cells. They produced purified circRNA that encoded a test protein and removed contaminating double‑stranded RNA that might trigger nonspecific alarm signals. When dendritic cells were exposed to lipid‑wrapped circRNA, they began making the new protein, switched on key surface markers of maturity, and released inflammatory messengers such as IL‑6, IL‑1β, and TNF‑α. When these activated dendritic cells were mixed with human T cells, both helper and killer T cells switched on and secreted interferon‑gamma, and more antigen‑specific T cells appeared in an assay that counts individual responding cells. These findings confirm that circRNA can precisely direct human immune cells, not just those of mice.
Promise, Caveats, and What Comes Next
For non‑specialists, the bottom line is that circular RNA vaccines, carried by lipid nanoparticles, look like a sturdier, longer‑acting cousin of current RNA shots. In mice they shrink tumors, curb viral infections, and do so without obvious harm, while in human cells they awaken the very immune players needed for targeted defense. However, all of this evidence is still preclinical, and different animal strains, disease types, and injection routes produced variable results. Large, carefully designed human trials will be required to learn how well this approach works in people and to fine‑tune dosing and delivery. If those hurdles are cleared, circRNA vaccines could become a versatile new tool for treating cancer and preventing future outbreaks of infectious disease.
Citation: Yang, R., Jia, L. & Cui, J. Lipid nanoparticle delivery of circle RNA vaccine induces potent immune responses. Sci Rep 16, 13268 (2026). https://doi.org/10.1038/s41598-026-46871-6
Keywords: circular RNA vaccines, lipid nanoparticles, cancer immunotherapy, infectious disease vaccines, T cell activation