Clear Sky Science · en
Ecoepidemiological determinants of Borrelia infection in sigmodontine rodents from the Delta and Parana Islands ecoregion, Argentina
Why tiny island rodents matter for health
On the river islands near Buenos Aires, wild rodents, ticks, cattle and people share the same changing landscape. Scientists wanted to know whether spiral-shaped bacteria of the Borrelia group, which elsewhere include the agents of Lyme disease, quietly circulate in these wetlands. By following rodents and their ticks over two years, they uncovered a hidden infection cycle that depends more on local animal numbers than on weather, and that may hold clues for managing disease risks at the wildlife–livestock–human interface. 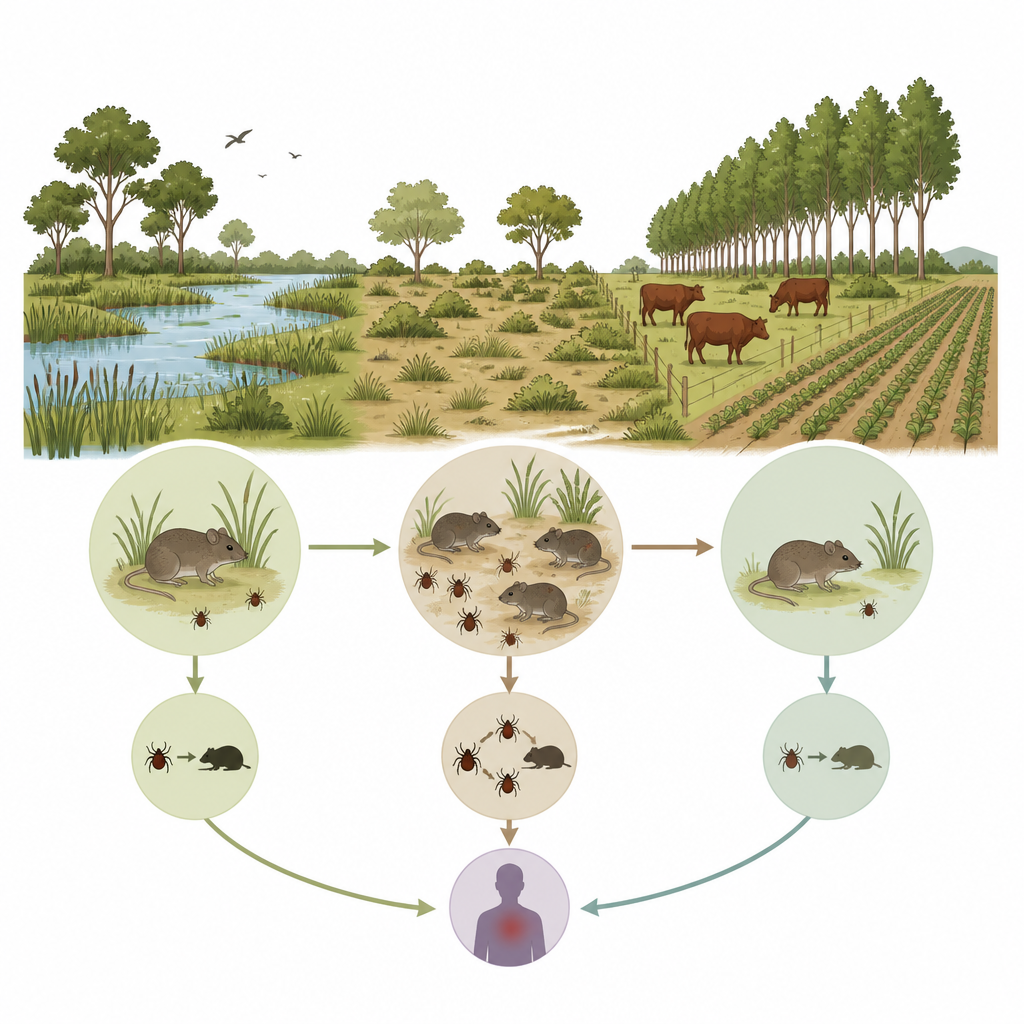
Watching wildlife over time
The team worked in the Delta and Paraná Islands ecoregion, a mosaic of natural grasslands and commercial tree plantations, some grazed by cattle and others left ungrazed. Every five weeks from 2010 to 2012, they trapped small mammals across 16 sites, recorded each animal’s size, sex and species, and collected blood, organs and attached ticks. In total, they sampled 770 rodents from eight species, most of them Azara’s grass mouse and the long-nosed mouse Oxymycterus rufus. This careful, repeated sampling allowed the researchers to link infection patterns to changes in rodent and tick numbers at each place and time.
Finding hidden bacteria
Under the microscope, none of the blood smears showed obvious Borrelia. But when the scientists tested lung tissue using sensitive DNA methods, 17 rodents, or about 2 in every 100, were positive. Most infected animals were Azara’s grass mice, though five other rodent species also carried the bacteria. By comparing gene sequences from these samples with global databases, the team confirmed that all belonged to the Borrelia burgdorferi “sensu lato” group, the same broad family that includes Lyme disease agents in North America and Europe. The sequences formed three main genetic lineages in the study area, and one variant of a key surface protein (ospC) had not been seen before in this region. 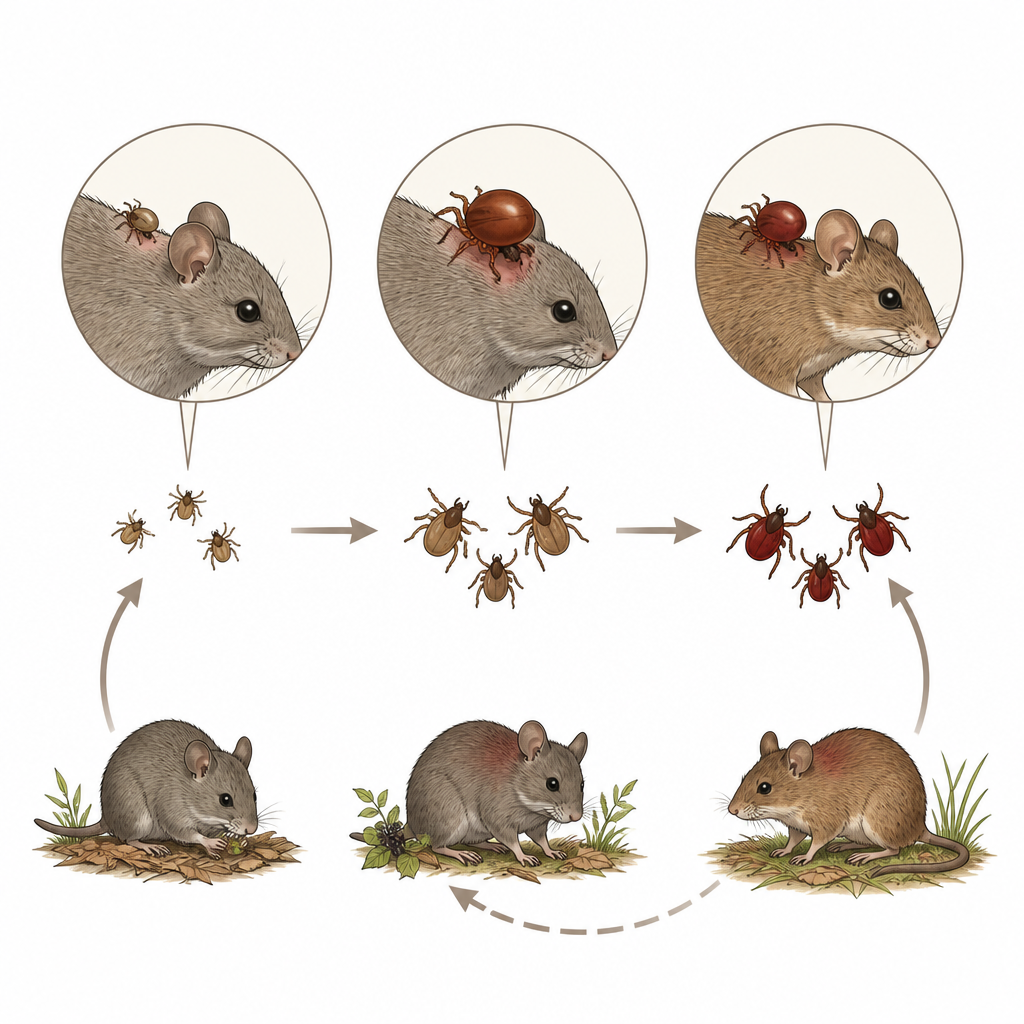
Who gets infected and when
To understand what drives infection, the researchers used statistical models that accounted for site and sampling date. Larger rodents, a proxy for older age, were more likely to be infected, suggesting that animals accumulate risk as they live longer and encounter more ticks. Species also differed: Oxymycterus rufus had a much lower chance of carrying Borrelia than Azara’s grass mouse, even though it often hosted many ticks. The key seemed to be which tick species fed on which rodent. The tick Ixodes loricatus, especially in its nymph stage, was strongly linked to infection in several rodent species, while another common tick, Amblyomma triste, was not. This points to Ixodes loricatus as the main local vector in this wetland system.
Role of cattle, land use and climate
The team also examined broader environmental influences. Periods with more Azara’s grass mice two sampling sessions earlier, and with more Ixodes nymphs one session earlier, were followed by higher infection odds, indicating that recent surges in host and immature tick numbers fuel transmission. In contrast, weather measurements such as temperature, rainfall and humidity added little explanatory power, at least at the coarse scale available. Cattle grazing told a more nuanced story. In simple analyses focused on Azara’s grass mouse, infection was about ten times less frequent in grazed areas than in cattle‑free areas. Earlier work in the same landscape had shown that cattle reduce Ixodes ticks on these mice, while favoring Amblyomma. When the new models included direct measures of tick and rodent abundance, the apparent protective effect of cattle disappeared, suggesting that livestock influence Borrelia mainly by altering the key tick population rather than through a separate pathway.
What this means for animals and people
Overall, the study reveals a stable, low-level Borrelia cycle carried by rodents and Ixodes ticks in a productive South American wetland. The bacterial lineages detected differ from those commonly linked to human Lyme disease in the Northern Hemisphere, and current evidence does not show that they cause typical Lyme in people. Still, their presence at the boundary between wildlife, cattle and nearby cities highlights the importance of combined monitoring of rodents, ticks and domestic animals. For now, the work shows that local tick and host demographics, more than broad climate patterns, shape where and when Borrelia appears, offering a clearer picture of how landscape change might shift future risks.
Citation: Barolin, J., Antoniazzi, L.R., Colombo, V.C. et al. Ecoepidemiological determinants of Borrelia infection in sigmodontine rodents from the Delta and Parana Islands ecoregion, Argentina. Sci Rep 16, 15024 (2026). https://doi.org/10.1038/s41598-026-45332-4
Keywords: Borrelia, rodents, ticks, Argentina, Lyme-like infection