Clear Sky Science · en
ROS-NLRP3 participates in the pyroptosis response of excretory-secretory products from protoscoleces of Echinococcus granulosus in hepatocytes
Why a tiny parasite matters for liver health
Cystic echinococcosis is a disease caused by a small tapeworm that usually lives in animals like sheep and dogs but can accidentally infect people. The parasite forms fluid-filled cysts, most often in the liver, which can quietly grow for years before causing pain, digestive problems, or serious complications. Doctors know that liver cells are damaged during this infection, but it has been unclear exactly how the parasite’s presence translates into cell death. This study looks closely at a specific kind of fiery cell death, driven by molecules called reactive oxygen species and an internal alarm system, to understand how parasite secretions may directly injure liver cells.
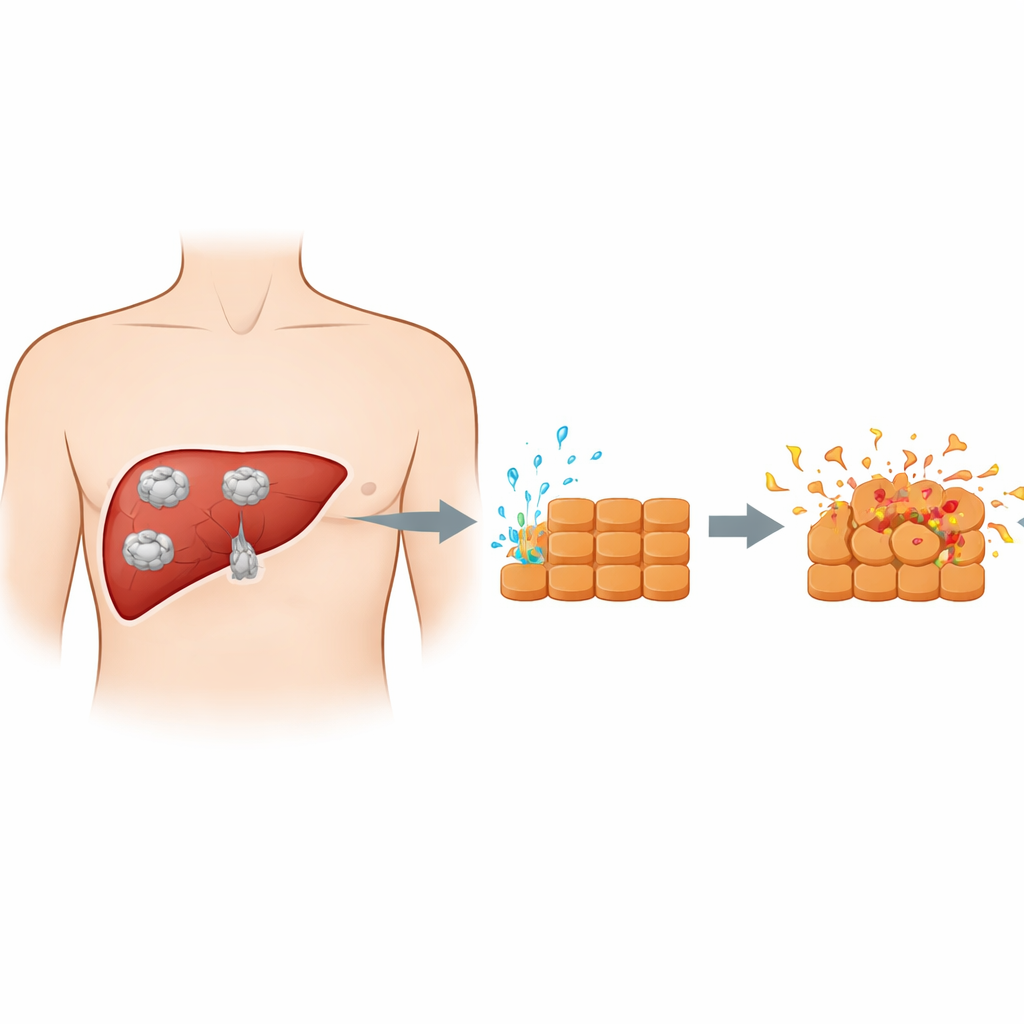
How the parasite and liver cells meet
When people swallow parasite eggs, the young forms travel from the intestine to the liver and develop into cysts. These cysts are not just passive bubbles; the growing parasite constantly releases a mixture of proteins and other substances into the surrounding tissue. Earlier work often used whole cyst fluid, which also contains many host-derived components, making it difficult to see what the parasite itself is doing. In this study, the researchers instead collected purified secretions from an early parasite stage called protoscoleces. They then exposed mouse liver cells grown in the lab to these excretory–secretory products, creating a cleaner model of the direct chemical conversation between parasite and liver cells as a cyst expands.
A fiery form of cell death inside the liver
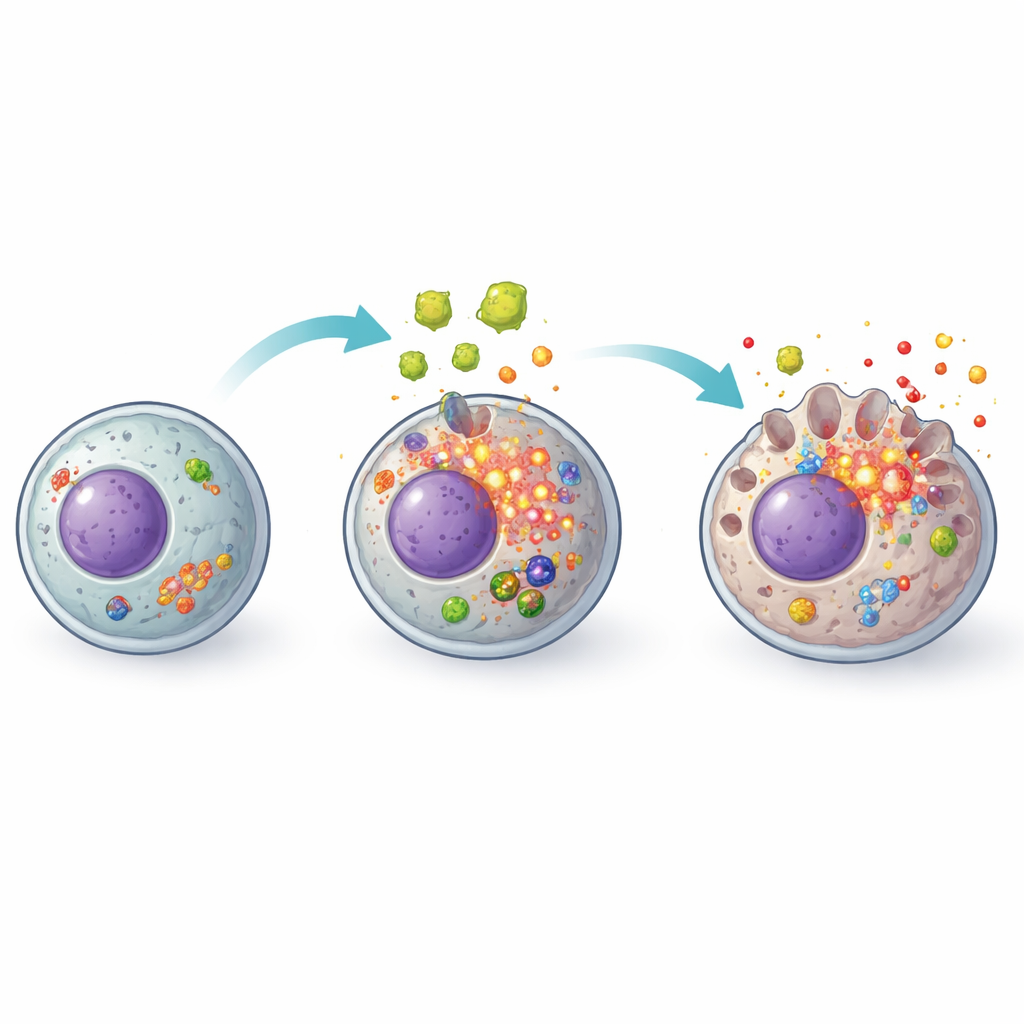
The team focused on pyroptosis, a form of programmed cell death that differs from the quiet, tidy shutdown seen in normal cell turnover. In pyroptosis, cells swell, their membranes become perforated, and they burst open, spilling inflammatory molecules that summon immune cells and intensify tissue damage. This process is controlled by a molecular machine known as the inflammasome, in particular a version built around a sensor protein called NLRP3. When the researchers treated liver cells with parasite secretions, they saw classic warning signs: reduced cell survival, increased leakage of a cell enzyme (LDH) that signals membrane damage, more dead cells under the microscope, and higher levels of key pyroptosis-related proteins that drive membrane pore formation and the release of inflammatory messengers.
Switching off the danger alarm
To test whether this destructive response really depended on the NLRP3 alarm system, the researchers used a small molecule called MCC950 that specifically blocks NLRP3 activity. Liver cells pretreated with this blocker were much more resilient when exposed to parasite secretions. They showed better survival, fewer membrane-damaged cells, lower leakage of LDH, and reduced production of inflammatory proteins associated with pyroptosis. Under fluorescence imaging, the amount of NLRP3 visible inside the cells dropped sharply. Together, these results indicate that the parasite’s secretions do not simply poison liver cells in a nonspecific way; instead, they actively trigger the cells’ own NLRP3-based self-destruct and inflammation machinery.
The spark: oxidative stress inside the cell
The study then asked what lights the fuse on this alarm system. A prime suspect was reactive oxygen species, chemically reactive byproducts of normal metabolism that become harmful when they build up. Parasite secretions caused liver cells to accumulate high levels of these reactive molecules while weakening their antioxidant defenses. When the researchers added N-acetylcysteine, a well-known antioxidant used clinically to protect the liver in other settings, the picture changed. Reactive oxygen levels fell, antioxidant markers recovered, and the activation of the NLRP3 complex and its downstream pyroptosis machinery was strongly dampened. Cell survival improved, and fewer inflammatory molecules were released, suggesting that oxidative stress is a crucial upstream trigger for this chain reaction.
What this means for people at risk
Taken together, the findings reveal a clear storyline: proteins secreted by Echinococcus granulosus around its liver cysts can overload nearby liver cells with reactive oxygen species, which in turn activate the NLRP3 inflammasome and drive an explosive, inflammatory form of cell death. This mechanism helps explain how a seemingly quiet parasitic cyst gradually erodes liver tissue and fuels chronic inflammation. For patients, the work points to new potential treatment angles beyond surgery and standard antiparasitic drugs. Approaches that reduce oxidative stress or specifically block the NLRP3 inflammasome might one day help protect liver cells, slow disease progression, and lower the risk of complications in cystic echinococcosis.
Citation: Cao, J., Chen, J., Li, H. et al. ROS-NLRP3 participates in the pyroptosis response of excretory-secretory products from protoscoleces of Echinococcus granulosus in hepatocytes. Sci Rep 16, 14316 (2026). https://doi.org/10.1038/s41598-026-45127-7
Keywords: cystic echinococcosis, liver inflammation, pyroptosis, reactive oxygen species, NLRP3 inflammasome