Clear Sky Science · en
Synthesis, spectroscopic, characterization, antimicrobial, DNA interaction, DFT and molecular docking studies of a new Cu(II)-Schiff base complex
Why a Vitamin-Inspired Metal Drug Matters
Many of today’s antibiotics and cancer drugs are losing their edge, either because microbes are becoming resistant or side effects limit how much medicine patients can tolerate. This study explores an inventive approach: building a new copper-based compound from folic acid—the familiar vitamin B9—and testing whether this tailored molecule can attack bacteria, latch onto DNA, and slow the growth of breast cancer cells. By combining a nutrient that naturally seeks out rapidly dividing cells with a biologically active metal, the researchers aim to create a smarter, more versatile therapeutic tool.
Turning a Common Vitamin into a Custom Tool
The team began with folic acid, a vitamin that our cells use for DNA building and repair and that cancer cells often hoard. They chemically linked folic acid to salicylaldehyde, a simple aromatic chemical, to form a new "Schiff base" ligand—essentially a custom-designed hook for grabbing a metal ion. When this ligand met copper(II) ions, it formed a binuclear complex: two copper centers bridged by the organic framework. The scientists prepared this copper–ligand system in both ordinary (bulk) and nanometer-sized forms and carefully confirmed its structure using a battery of standard lab techniques, including infrared and visible light spectroscopy, nuclear magnetic resonance, mass spectrometry, magnetic measurements, and thermal analysis. They also used electron microscopy to show that the nano version forms well-defined particles around 15–27 nanometers in size. 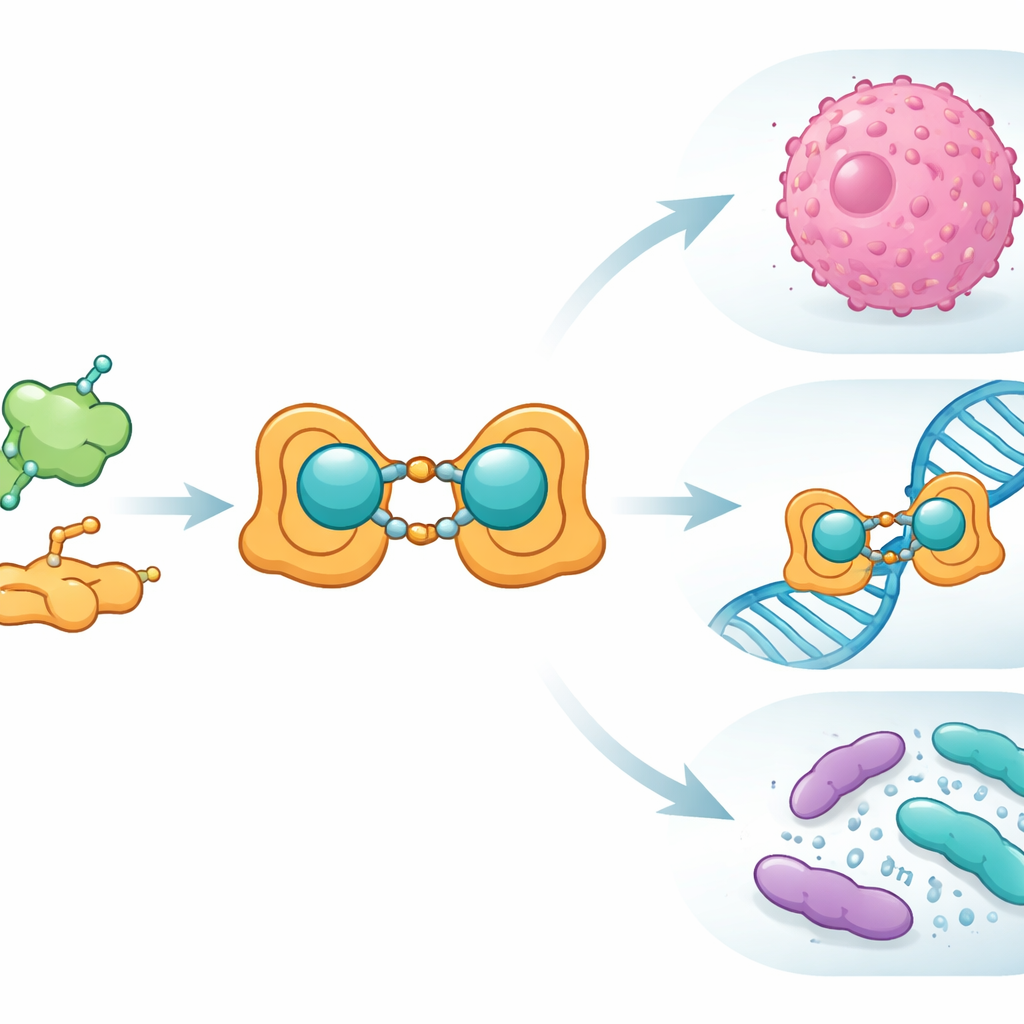
How the New Compound Behaves
Physical measurements painted a consistent picture of the compound’s shape and bonding. Shifts in the infrared signals revealed that both nitrogen and oxygen atoms of the Schiff base ligand bind strongly to copper, while magnetic and electron spin studies indicated a distorted tetrahedral environment around the metal centers and strong interaction between the two copper ions. Mass spectrometry and size-exclusion chromatography suggested that the complex can assemble into higher-mass polymer-like chains, stabilized by interactions between neighboring units. To supplement these experiments, the researchers turned to modern quantum-chemical calculations, which supported the proposed geometry and electronic structure. The calculations also showed that once copper is bound, the energy gap between the compound’s frontier orbitals shrinks markedly, implying a more reactive, charge-transfer-friendly system with enhanced optical and electronic responsiveness.
Putting Bacteria, DNA, and Cancer Cells to the Test
With the structure in hand, the team examined how the new copper complex behaves in biological settings. In antimicrobial tests against common bacterial and fungal strains, the free ligand alone did essentially nothing, but the nano-sized copper complex produced clear zones of inhibited growth for disease-related bacteria such as Escherichia coli and Staphylococcus aureus, and for the yeast Candida albicans. Next, they probed how the compounds interact with DNA using a trio of methods: changes in ultraviolet light absorption, displacement of a fluorescent dye that nestles between DNA base pairs, and measurements of solution viscosity. Across these experiments, the copper complex bound to DNA more strongly than the ligand alone and behaved much like classic agents that slide between base pairs, subtly lengthening and unwinding the helix. Molecular docking simulations, which computationally “fit” the molecules into protein structures from bacteria and fungi, showed favorable binding pockets and negative interaction energies, again with the copper complex outperforming its metal-free counterpart.
Promise Against Breast Cancer Cells
The researchers then challenged human MCF-7 breast cancer cells with the new molecules. Using a standard cell viability test, they found that the free folate-derived ligand was relatively weak, requiring high concentrations to halve cell growth. By contrast, the nanoscale copper complex achieved the same level of growth inhibition at much lower doses, approaching the potency of the benchmark chemotherapy drug cisplatin, though it was still less powerful overall. The folic-acid fragment may help steer the complex toward cancer cells that overexpress folate receptors, while the copper centers can generate reactive species and disturb DNA and key enzymes. Theoretical analyses of charge distribution and electrostatic potential supported this picture by identifying specific nitrogen and oxygen atoms as prime sites for donating electrons to copper and to biological targets. 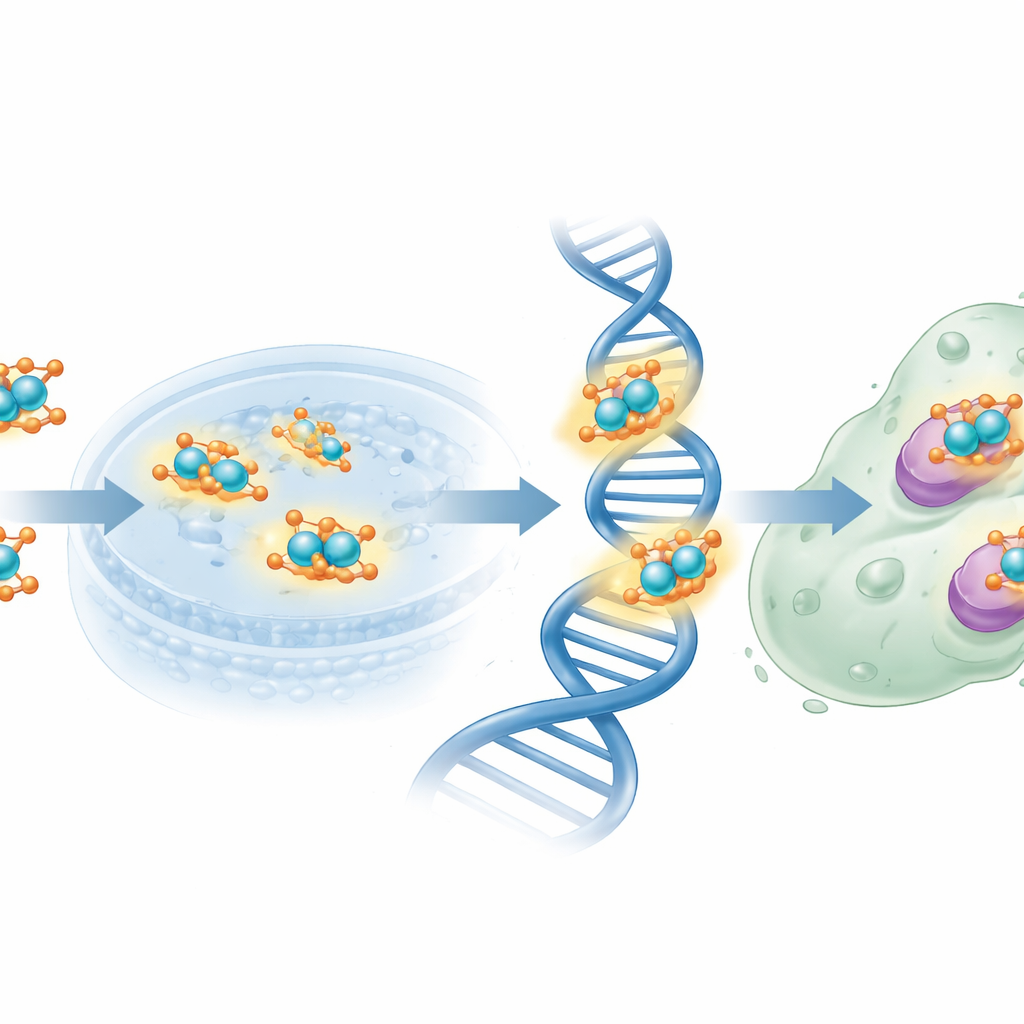
What This Could Mean for Future Medicines
In accessible terms, this study shows that cleverly combining a vitamin “address label” with an active metal center can transform an otherwise mild organic molecule into a multifaceted weapon against microbes and cancer cells. The folic-acid-based Schiff base on its own is modestly active, but when paired with copper and shaped into a binuclear, nanoscale complex, it becomes better at slipping into DNA, disrupting bacterial machinery, and slowing the growth of breast cancer cells. While much work remains before such a compound could reach the clinic—especially detailed safety and selectivity studies—the results highlight a promising design strategy: use nutrients that naturally home in on fast-growing cells as scaffolds for metal complexes that can attack disease at the level of DNA and essential proteins.
Citation: Mahmoud, E.G., Ismail, E.H., Abdel Aziz, A.A. et al. Synthesis, spectroscopic, characterization, antimicrobial, DNA interaction, DFT and molecular docking studies of a new Cu(II)-Schiff base complex. Sci Rep 16, 13636 (2026). https://doi.org/10.1038/s41598-026-44842-5
Keywords: copper complexes, folic acid, Schiff base, DNA binding, anticancer agents