Clear Sky Science · en
GABARAPL1 is important for the activation of HRI during eIF2α phosphorylation-dependent stress response to sodium arsenite
Why this matters for our cells under stress
Every day, our cells are bombarded by stresses from the environment, including toxic metals such as arsenic. To survive, they must quickly slow down normal activities and switch into damage-control mode. This study uncovers an unexpected player, a protein called GABARAPL1, that helps cells sense arsenic-induced stress and decide whether to pause protein production and protect their genetic messages. Understanding this hidden safeguard may shed light on how cells cope with pollution, why some cells become more resistant to toxins, and how stress-control pathways can go wrong in disease.
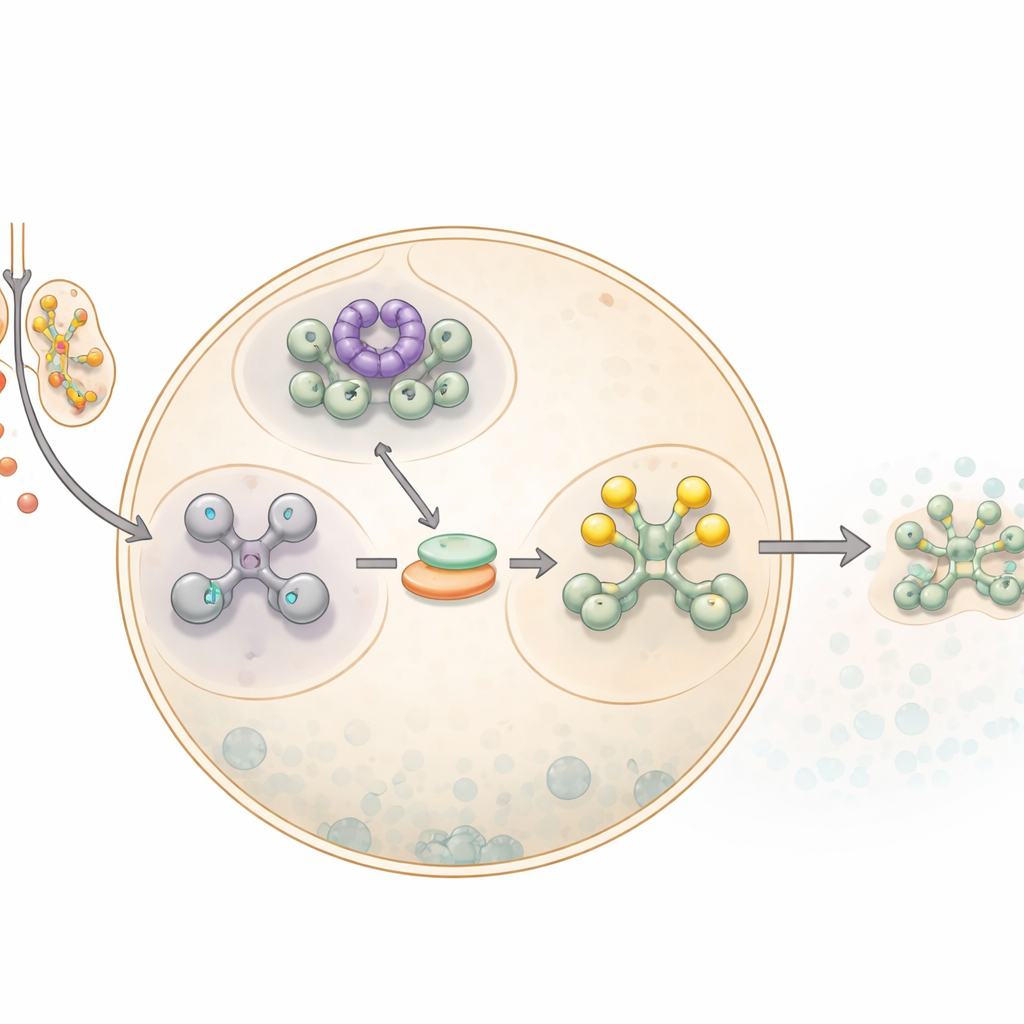
How cells hit the pause button
When cells encounter sodium arsenite, a form of arsenic, they activate a broader alarm network known as the integrated stress response. A key step in this response is shutting down most protein production. This is done by modifying a core translation factor, eIF2α, which normally helps start the process of building proteins from messenger RNAs. Once eIF2α is chemically tagged, or phosphorylated, the cell slows general protein making and funnels unused messenger RNAs into dense droplets called stress granules. These granules act as temporary storage sites, protecting RNAs until the danger has passed and giving the cell time to reprogram which proteins it makes.
A surprising role for a recycling protein
GABARAPL1 belongs to a family of proteins best known for autophagy, the cell’s internal recycling system that breaks down unwanted components. The researchers worked with human lung cancer cells and removed GABARAPL1 using gene editing or silencing. They then exposed cells to arsenite and watched what happened. In normal cells, arsenite rapidly triggered stress granule formation and strong eIF2α phosphorylation, consistent with a robust stress response. In striking contrast, cells lacking GABARAPL1 formed far fewer stress granules and showed only weak phosphorylation of eIF2α, even though arsenite was clearly entering the cells and activating other signaling routes.
Pinpointing the broken link in the alarm chain
To understand where the alarm chain failed, the team focused on HRI, a kinase that sits upstream of eIF2α and is specifically activated by arsenite-related oxidative stress. They found that early activation steps of HRI, involving self-modification of the protein, still occurred in cells without GABARAPL1. However, a later, crucial step was impaired: the efficient partnership between HRI and a chaperone protein called HSP90, which helps HRI reach its fully active form. Using an imaging method that detects when two proteins sit very close together, the researchers saw that, under arsenite stress, HSP90 and HRI re-associated over time in normal cells but failed to do so properly when GABARAPL1 was missing. This pointed to GABARAPL1 acting as a kind of scaffold that brings HSP90 and HRI together at the right moment.
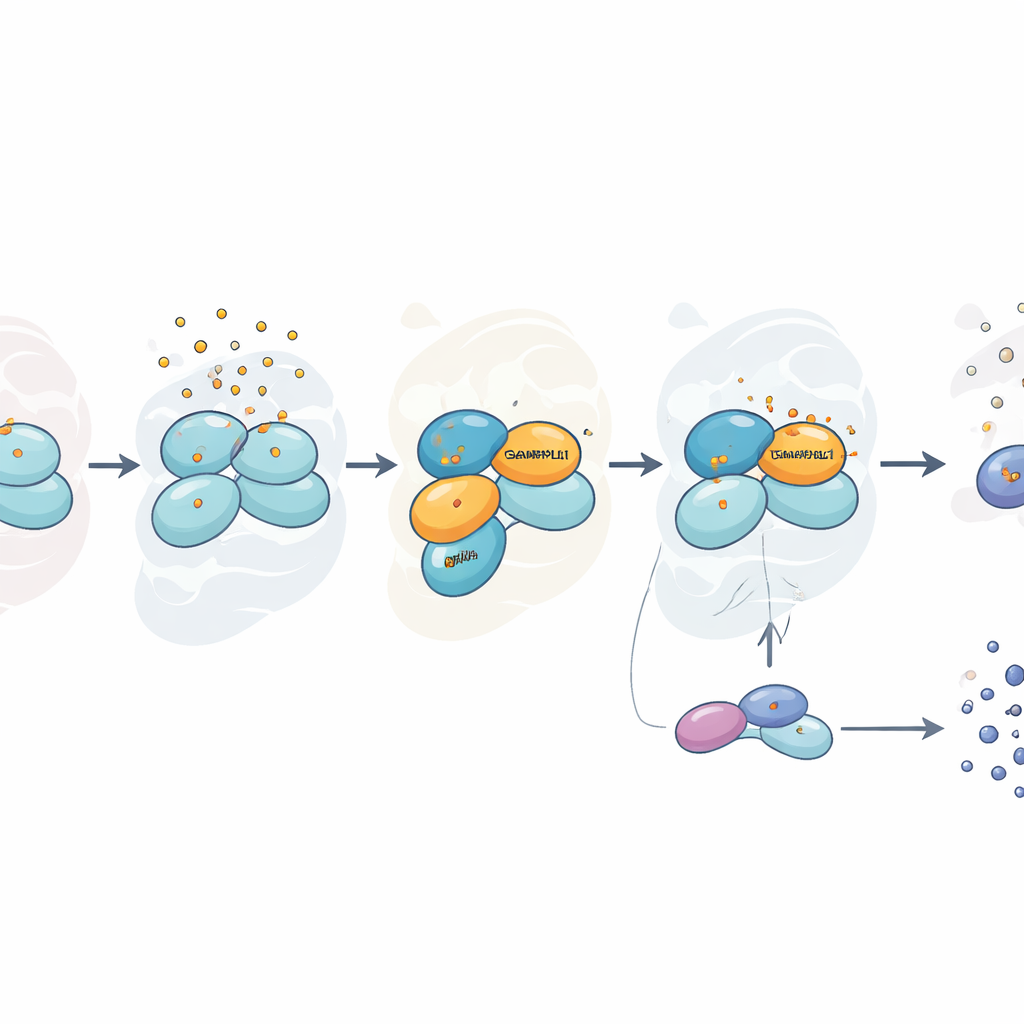
Stress that is selective, not universal
Interestingly, this dependency on GABARAPL1 was specific to arsenite. When the researchers stressed cells with sorbitol, which alters cell water balance, or with hydrogen peroxide, another source of oxidative damage, eIF2α phosphorylation and stress granule formation proceeded normally even without GABARAPL1. Under arsenite, however, cells lacking GABARAPL1 not only mounted a weaker stress response but also appeared more resistant to the toxin in viability tests, likely because they dampened their metabolic activity and avoided some of the damage associated with an intense alarm reaction. Another related protein from the same family, LC3B, showed only a partial effect, hinting that several family members can cooperate but that GABARAPL1 plays a leading role.
What this means for cell protection
Taken together, the study proposes that GABARAPL1 is a previously unrecognized helper in the arsenite stress pathway. By acting as a cofactor for HSP90, it supports the full activation of HRI, which in turn modifies eIF2α, slows protein production, and drives the formation of protective stress granules. Without GABARAPL1, this chain of events is weakened, leaving cells less responsive to arsenite and altering how they balance survival versus damage. These insights deepen our understanding of how cells interpret specific environmental threats and may inform future research on toxic exposure, cancer cell resistance, and therapies that target cellular stress responses.
Citation: Campenet, S., Lamarre, M., Durand, J. et al. GABARAPL1 is important for the activation of HRI during eIF2α phosphorylation-dependent stress response to sodium arsenite. Sci Rep 16, 10818 (2026). https://doi.org/10.1038/s41598-026-44621-2
Keywords: cellular stress response, arsenic toxicity, stress granules, protein quality control, autophagy-related proteins