Clear Sky Science · en
Dynamic optical coherence microscope integrated with cell-cultivation chamber enabled longitudinal and early-stage assessment of tumor spheroid-drug interaction
Watching Cancer Cells Respond in Real Time
Cancer drugs are usually judged by how much they shrink a tumor, but by the time a mass visibly changes size, precious hours or days have passed. This study introduces a way to watch tiny 3D clusters of breast cancer cells—called tumor spheroids—respond to treatment almost as soon as a drug is added. By combining an advanced light-based microscope with a miniaturized cell-culture chamber, the researchers track how living cancer cells behave deep inside these spheroids without dyes, labels, or destroying the sample.
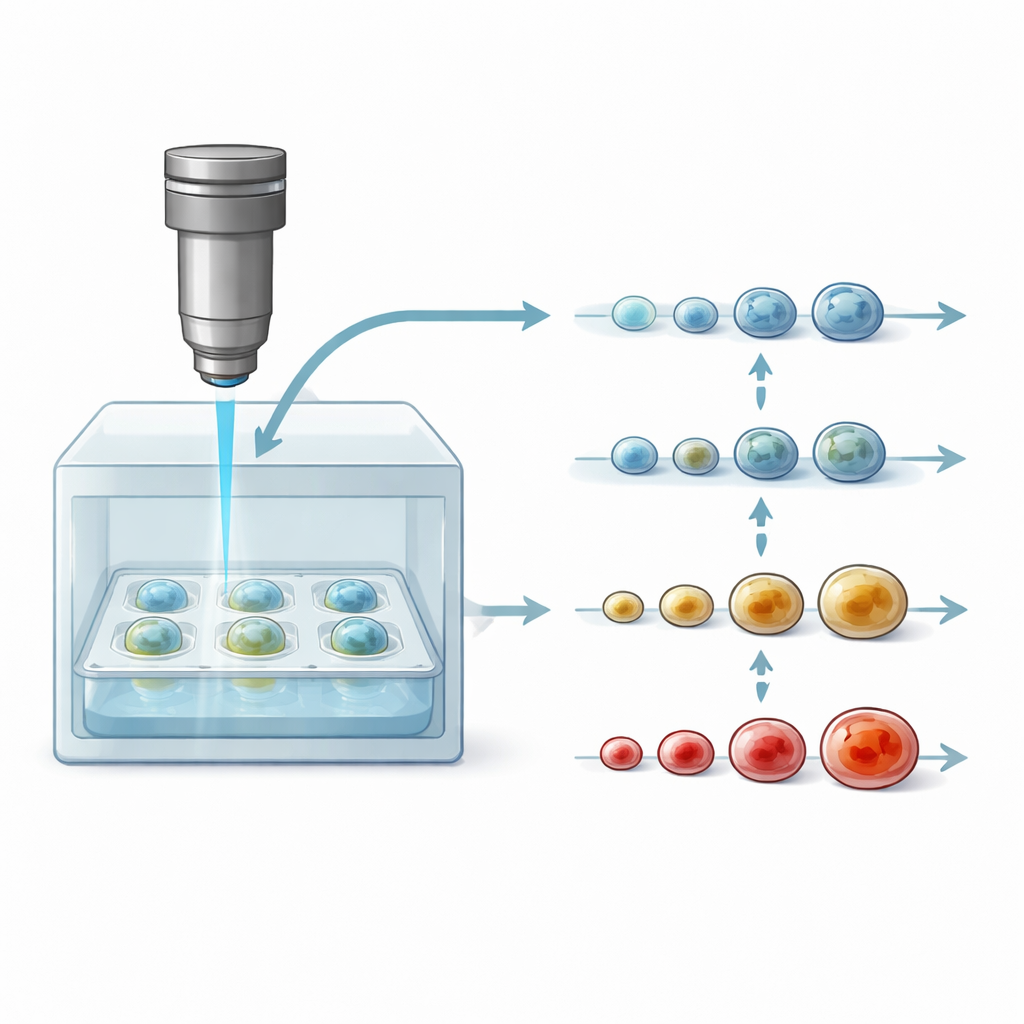
A New Window into Living Cancer Models
Researchers increasingly grow tumor spheroids because they mimic many features of real tumors better than flat cell layers or some animal models. However, standard tools to examine them, such as staining and fluorescence imaging, often require slicing or chemically altering the tissue. That means each spheroid can be tested only once, and fine-grained tracking over time is impossible. Traditional optical coherence tomography (OCT)—a non-invasive, 3D imaging method used in eye clinics—can see inside spheroids, but it mostly shows shape and size. Those structural changes tend to appear late, long after a drug has begun to affect cell health.
Turning Motion into a Vital Sign
The team built on OCT by using a dynamic version, called DOCT, that doesn’t just map structures; it measures subtle fluctuations in the light signal caused by tiny motions inside cells. Two analysis methods, one tracking how much the signal varies over time and another tracking how quickly it decorrelates, act together like a motion-sensitive stethoscope. When cells are healthy and active, their internal components move and rearrange, creating a lively DOCT signal. When cells slow down, become stressed, or die, that internal bustle decreases. By quantifying “high-dynamics” and “low-dynamics” regions inside each spheroid, the system estimates where living and non-viable cells lie in 3D and how those patterns evolve.
Keeping Cells Comfortable While We Watch
To follow the same spheroid for days, the researchers integrated their DOCT microscope with a compact cultivation chamber that maintains body-like conditions: 37°C and controlled carbon dioxide. A standard 96-well plate holding many spheroids sits beneath the imaging head, protected by a lid to avoid contamination. The light beam passes through thin glass layers to reach the sample, and the scanning is fast and gentle enough that each point in the tissue is only briefly exposed. This setup allowed two styles of experiment: one where dozens of spheroids, treated with different doses of three common breast-cancer drugs, were imaged every four hours for about four days, and another where individual spheroids were followed automatically every 30 minutes over the same period.
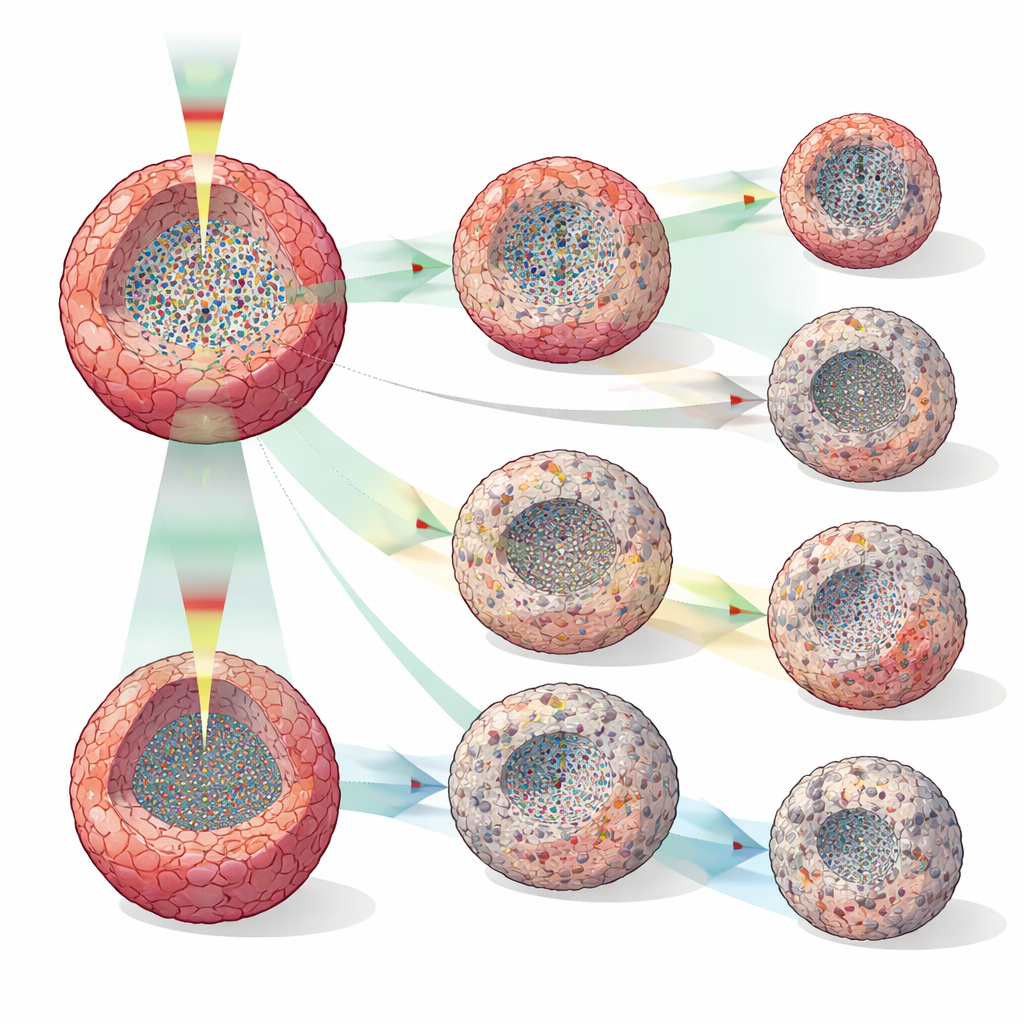
Seeing Drug Effects Long Before Size Changes
The group treated human breast cancer spheroids (MCF-7) with doxorubicin, tamoxifen, or paclitaxel at several concentrations and compared DOCT readouts with simple volume measurements. While spheroid size often looked similar across doses for many hours, DOCT metrics diverged much sooner—sometimes just two hours after treatment, and consistently by 12 hours. Different drugs produced distinct internal patterns: high-dose doxorubicin led to shrinking spheroids and central zones of reduced activity suggestive of cell death; tamoxifen mainly slowed growth, sometimes forming a quiet outer shell that may reflect growth arrest or apoptosis at the periphery; and paclitaxel produced scattered low-activity patches, consistent with its known disruption of cell division and intracellular transport. High-temporal-resolution runs at 30-minute intervals revealed transition points—when quiet cores disappeared then reappeared, or when low-activity layers formed—that would be invisible in sparse, destructive tests.
What This Means for Future Cancer Drug Testing
For a non-specialist, the key message is that this integrated DOCT–chamber system can “listen” to how tumor-like cell clusters react to drugs from the inside out, hours before they noticeably grow or shrink. Instead of waiting days to see whether a treatment works, researchers can detect early, dose-dependent changes in cell activity without adding dyes or sacrificing samples. That makes it easier to compare drugs, tune doses, and study why certain tumors resist therapy. While still a laboratory tool, this approach points toward faster, more informative preclinical drug testing platforms that could ultimately help identify promising cancer treatments more efficiently and reduce reliance on animal experiments.
Citation: Abd El-Sadek, I., Morishita, R., Guo, Y. et al. Dynamic optical coherence microscope integrated with cell-cultivation chamber enabled longitudinal and early-stage assessment of tumor spheroid-drug interaction. Sci Rep 16, 14254 (2026). https://doi.org/10.1038/s41598-026-44296-9
Keywords: dynamic optical coherence tomography, tumor spheroids, cancer drug response, label-free imaging, longitudinal cell culture