Clear Sky Science · en
Epigenetic regulation of the glucocorticoid receptor gene through methylation is linked to post-traumatic stress disorder
Why some minds bend but do not break
Not everyone who lives through war, assault, or disaster develops post-traumatic stress disorder (PTSD). This uneven toll of trauma raises a basic question: what makes one brain vulnerable while another proves resilient? In this rat study, researchers looked beneath the surface of behavior to the level of chemical tags on DNA, exploring how subtle switches in the brain’s stress circuitry may tilt some individuals toward long‑lasting fear, anxiety, and low mood after trauma.
From one traumatic event to very different outcomes
To mimic trauma, the team exposed rats to an intense sequence of stressors—restraint, forced swimming, exposure to anesthetic fumes, and mild electric shock—known as the single prolonged stress model. Afterwards, the animals were left undisturbed for a week and then examined with a battery of standard tests that measure despair‑like behavior, anxiety, and loss of pleasure. Even though all rats had faced the same ordeal, their responses diverged. Some showed marked immobility in the forced swim test, reduced exploration of open spaces, and less interest in sweetened water; these were labeled high‑susceptibility animals. Others behaved much like unstressed rats and were considered low‑susceptibility, a proxy for resilience. 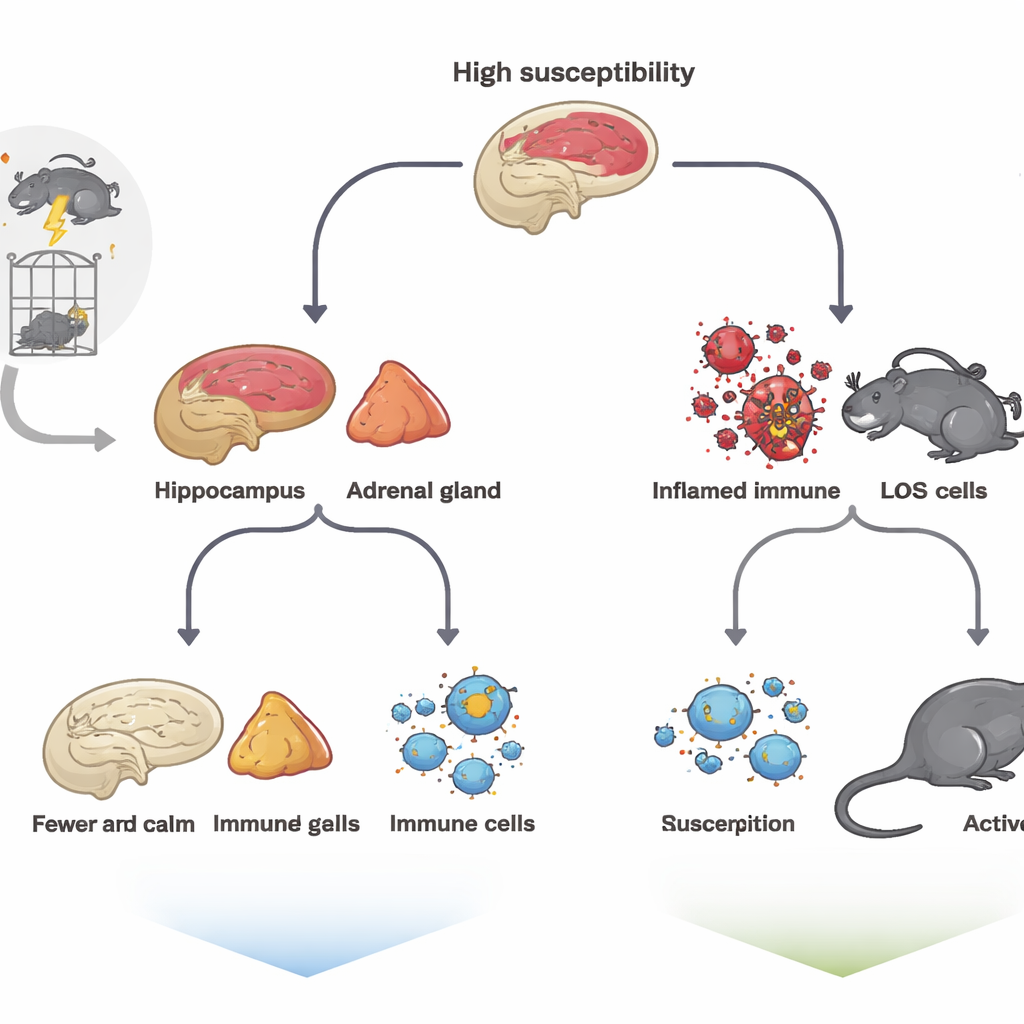
A chemical dimmer switch on a key brain receptor
The researchers focused on a receptor in the brain called mGluR5, which helps nerve cells adjust their connections and is involved in fear learning and emotional regulation. Rather than looking for mutations in the receptor’s gene, they examined DNA methylation—small chemical tags that act like dimmer switches, usually turning genes down when they accumulate on certain spots. In the hippocampus, a region critical for memory and stress control, high‑susceptibility rats had both lower activity of the mGluR5 gene and higher methylation at specific sites in the gene’s control region. Low‑susceptibility rats did not show this pattern. This pairing of heavier chemical tagging with reduced gene activity suggests that the trauma experience had effectively “turned down” a receptor that normally helps the brain adapt to stress.
Stress hormones, inflammation, and cellular wear and tear
Changes in gene control did not occur in isolation. High‑susceptibility rats showed a spike in the body’s main stress hormone system. Cells in a deep brain region that releases corticotropin‑releasing factor—a key trigger of the stress cascade—were more active, and blood levels of corticosterone, the rodent counterpart of cortisol, were elevated. At the same time, their immune systems tilted toward a more hostile state. Levels of a pro‑inflammatory signal, interleukin‑1β, were higher, while levels of the calming signal interleukin‑10 were lower. Microglia, the brain’s resident immune cells, were more activated. These animals also showed clear signs of oxidative stress: more chemical by‑products of damage to fats in cell membranes, and reduced levels of natural antioxidants such as glutathione and superoxide dismutase. Low‑susceptibility rats, in contrast, maintained more balanced hormone, immune, and antioxidant profiles.
Linking brain switches to whole‑body stress responses
By combining behavior, brain tissue analysis, and blood measurements, the study paints a coordinated picture. Animals that succumbed to PTSD‑like symptoms were the ones in which DNA methylation suppressed mGluR5 in the hippocampus, while stress hormones surged, inflammation flared, and cellular defenses against oxidative damage faltered. Those that stayed resilient, despite facing the same ordeal, avoided this full cascade of changes. Although the work was done in rats and focused on one brain region and one form of epigenetic control, it highlights how a small chemical change on DNA can ripple outward to reshape stress networks across the brain and body. 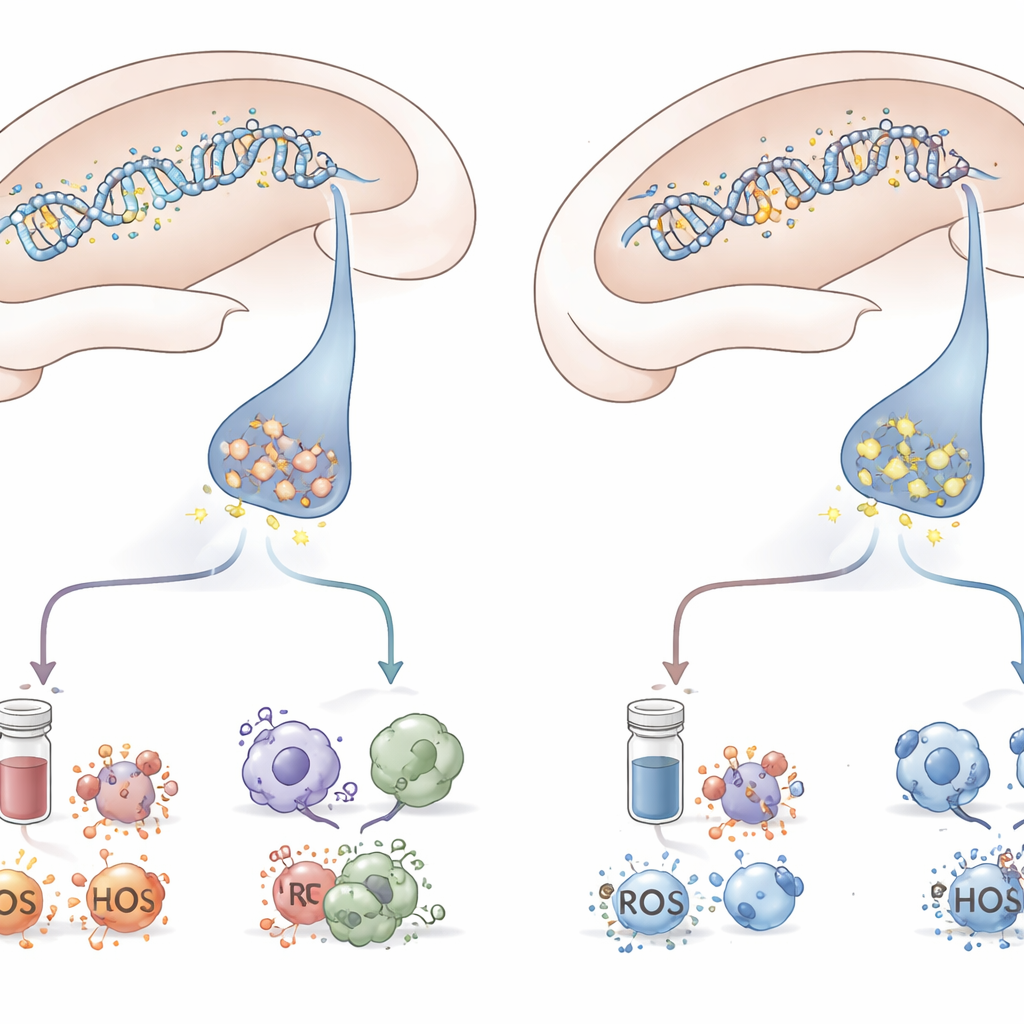
What this means for people living with trauma
For a lay reader, the core message is that vulnerability to PTSD may depend not only on life events but also on how our genes are chemically tuned after trauma. In this study, heavier methylation on the mGluR5 gene acted like a brake on a helpful brain receptor and went hand in hand with overactive stress hormones, simmering inflammation, and greater cellular strain. These findings suggest that similar epigenetic marks in humans could someday help identify who is at greatest risk after trauma, guiding early support and treatment. They also hint that therapies aimed at gently re‑setting these molecular switches—whether through drugs, lifestyle changes, or future epigenetic tools—might one day help shift the balance from susceptibility toward resilience.
Citation: Ye, M., Lee, Hj. & Shim, I. Epigenetic regulation of the glucocorticoid receptor gene through methylation is linked to post-traumatic stress disorder. Sci Rep 16, 10635 (2026). https://doi.org/10.1038/s41598-026-43615-4
Keywords: post-traumatic stress disorder, epigenetics, DNA methylation, stress resilience, hippocampus