Clear Sky Science · en
Gut microbiota dysbiosis and metabolic abnormalities promote oxidative stress and fibrosis in idiopathic pulmonary fibrosis
Why Your Gut Could Matter to Your Lungs
Idiopathic pulmonary fibrosis (IPF) is a relentless lung disease that scars the delicate air sacs, making every breath harder. Doctors have long focused on the lungs themselves, but this study asks a surprising question: could microbes and chemicals in our intestines quietly help drive this scarring process? By combining patient samples with animal experiments, the researchers trace a chain of events from gut imbalance to chemical stress in the blood and, ultimately, to stiff, damaged lungs.
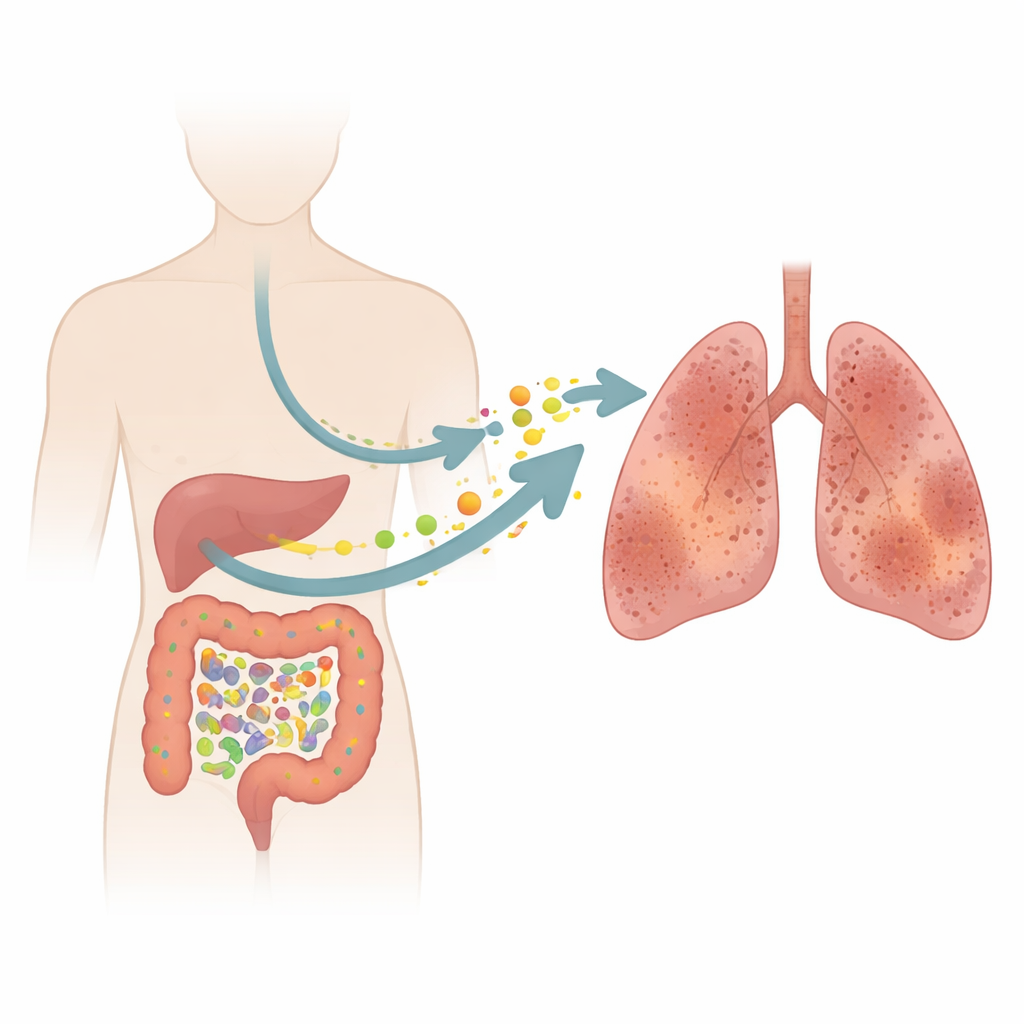
A Silent Illness That Steals Breath
IPF mainly affects older adults and gradually robs them of lung capacity. In this study, 17 people with IPF were compared to 17 healthy peers of similar age and sex. Standard breathing tests showed that people with IPF had markedly reduced lung volumes and, even more dramatically, a poorer ability to move oxygen from air into the blood. These measurements reflect thickened, scarred walls in the lung’s air sacs, which trap air instead of allowing it to flow freely. Despite modern scans and tests, IPF is still hard to diagnose early and current drugs only slow, rather than stop, the disease, underscoring the need to look beyond the lungs for new clues.
The Gut Community and Its Chemical Footprint
The team next turned to the gut. Participants provided stool samples so the researchers could catalog their intestinal microbes and the thousands of small molecules those microbes and the body produce. Overall, the broad structure of the gut community did not look radically different between patients and healthy volunteers. However, more detailed analysis revealed that certain bacterial groups, particularly those in the Ruminococcus family and a species called Ruminococcus torques, were more abundant in people with IPF. At the same time, the chemical “fingerprint” of the stool was clearly altered, showing that even modest shifts in key microbes can have outsized effects on the metabolic environment of the gut.
A Missing Protective Molecule and Rising Chemical Stress
Among many shifted chemicals, one stood out: a small dipeptide called tryptophan–glycine (Trp‑Gly), formed from the amino acids tryptophan and glycine. Levels of this molecule were strongly reduced in IPF patients and tightly linked to the overabundance of Ruminococcus. Blood tests added another piece to the puzzle. People with IPF had higher levels of inducible nitric oxide synthase (iNOS), a marker of oxidative stress—essentially chemical wear and tear on tissues. Their blood contained less tryptophan and glycine overall, but more kynurenine, a breakdown product of tryptophan that is associated with inflammation and oxidative damage. Together, these findings suggest that an imbalanced gut community may consume or reroute tryptophan and glycine, depleting potentially protective molecules like Trp‑Gly while fueling pathways that generate harmful oxidative stress.
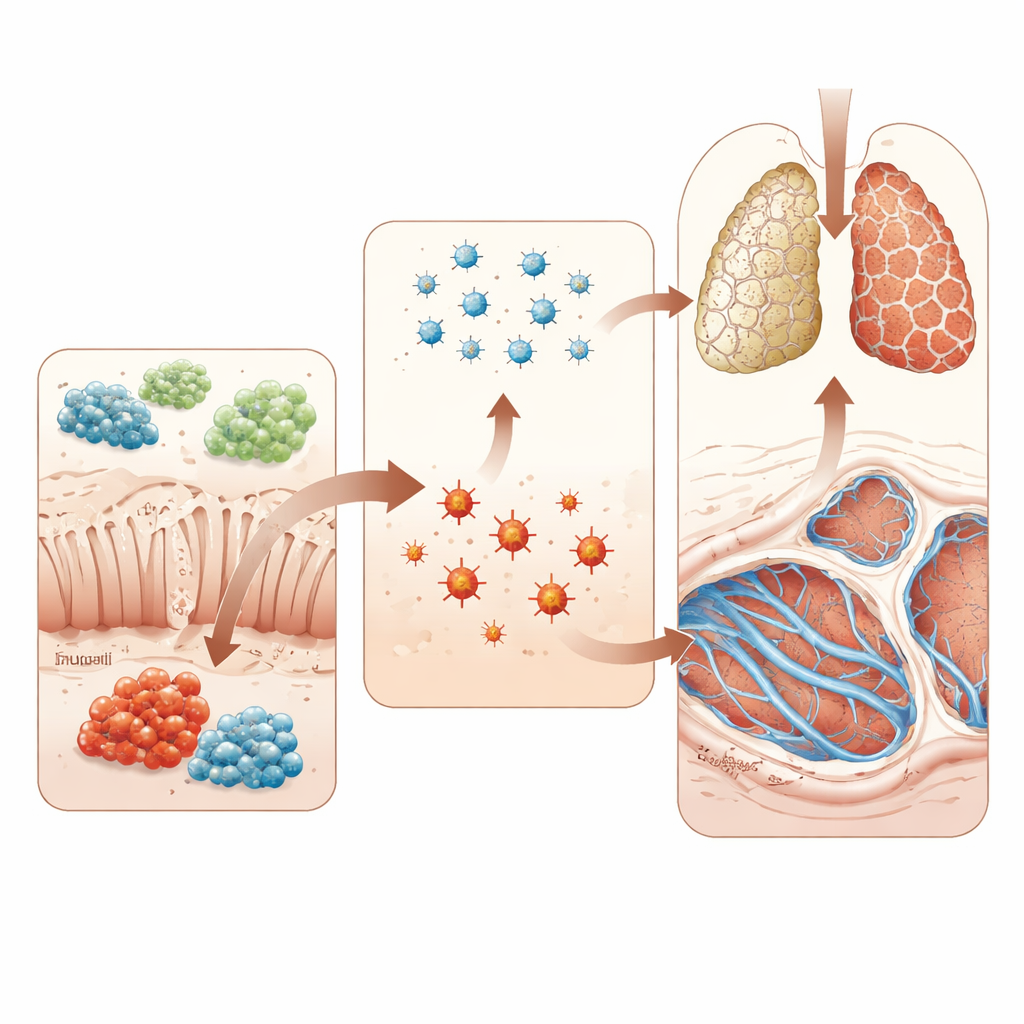
Testing Cause and Effect in Animal Models
To move beyond correlation, the researchers used a standard rat model of lung scarring triggered by the drug bleomycin. Some animals received Trp‑Gly by mouth, while others were given live R. torques bacteria. Trp‑Gly supplementation reduced markers of oxidative stress in the blood, lowered the lung weight relative to body size, and lessened the thick collagen bands and distorted architecture seen under the microscope. It also dampened activity in a key scarring pathway known as TGF‑β/Smad3. In sharp contrast, rats given R. torques had worse lung scarring, higher oxidative stress, and stronger activation of the same fibrotic signaling pathway. These experiments support the idea that certain gut bacteria can push the lungs toward fibrosis, while specific gut‑derived molecules can pull them back.
What This Means for Future Care
In everyday terms, this work proposes a chain reaction: an unhealthy shift in gut microbes, especially expansion of Ruminococcus and R. torques, disrupts the production of small protective molecules like Trp‑Gly, which in turn raises chemical stress in the bloodstream and flips on scarring programs in the lungs. While the study is still early and involved a modest number of volunteers and animals, it points to a new way of thinking about IPF: not just as a disease of the lungs, but as the end result of a gut–lung axis gone awry. If future research confirms these links, doctors may one day diagnose risk earlier using gut or blood markers and treat IPF not only with lung‑targeted drugs, but also with tailored diet, microbial therapies, or designer metabolites that restore this fragile biochemical balance.
Citation: Bai, B., Li, F., Zheng, P. et al. Gut microbiota dysbiosis and metabolic abnormalities promote oxidative stress and fibrosis in idiopathic pulmonary fibrosis. Sci Rep 16, 12305 (2026). https://doi.org/10.1038/s41598-026-43366-2
Keywords: idiopathic pulmonary fibrosis, gut microbiota, oxidative stress, tryptophan metabolism, fibrosis