Clear Sky Science · en
Harnessing a bispecific αGD2 × αCD3 protein engager to target GD2-overexpressing lung tumors
Why turning the immune system against lung cancer matters
Lung cancer remains one of the deadliest cancers worldwide, in part because it is often diagnosed late and can resist standard treatments like surgery, chemotherapy, and radiation. In recent years, scientists have begun teaching the immune system to recognize and destroy cancer cells more precisely. This study explores a new way to do that by designing a custom-made protein that physically links immune cells to lung cancer cells that carry a particular surface marker, potentially opening a path to more targeted and less toxic therapies.
A special flag on lung cancer cells
Cancer cells often display unusual molecules on their surface that can serve as flags for targeted therapies. One such flag is GD2, a fatty-sugar molecule found at high levels on certain tumors but only sparsely on most normal tissues. The authors first asked how common GD2 is on lung cancer cells. Using standard lab cell lines, they found that an adenocarcinoma line called A549 carried much higher amounts of GD2 than another non-small-cell lung cancer line, NCI-H460. This confirmed that at least some lung cancers strongly display GD2 and could therefore be singled out based on this marker.
Building a molecular matchmaker
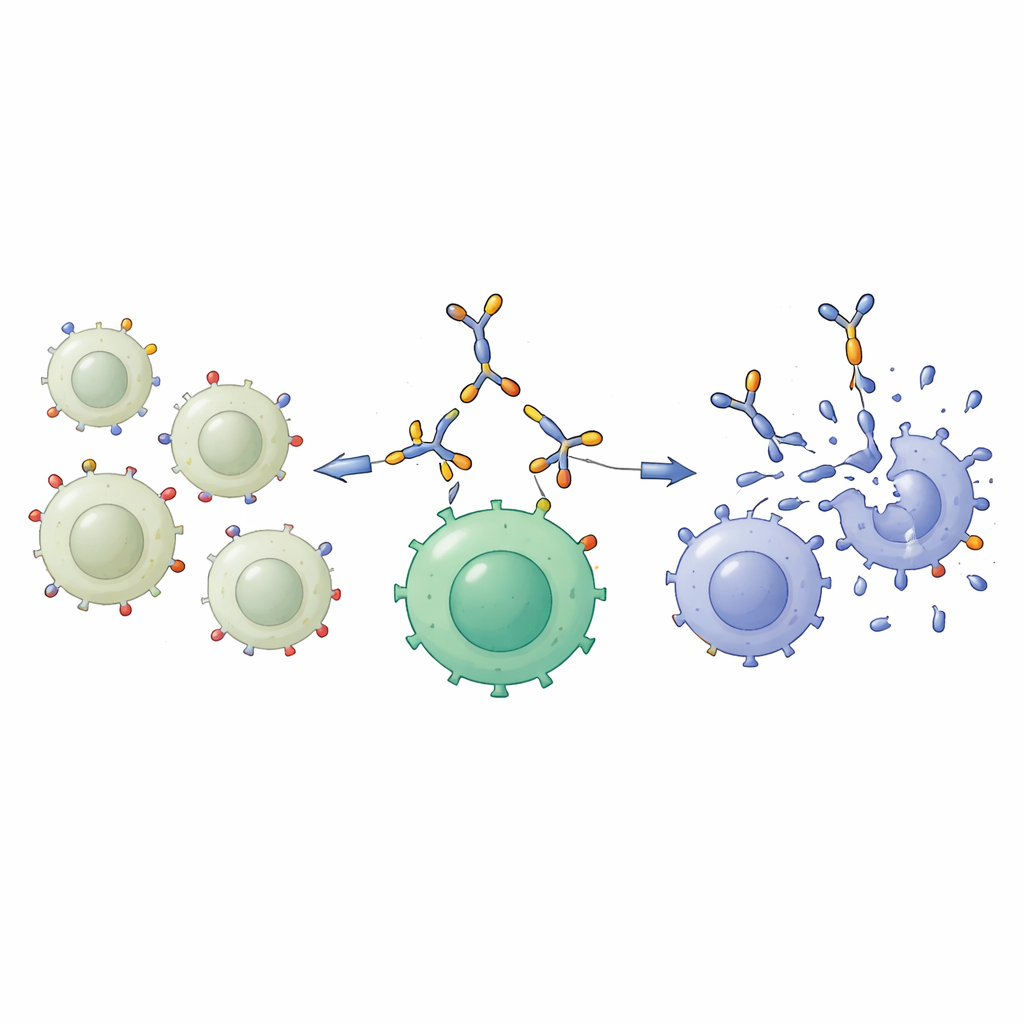
To exploit this difference, the team engineered a “bispecific protein engager,” a compact fusion protein designed to grab two different cell types at once. One end recognizes GD2 on the tumor cell surface; the other end recognizes CD3, a key molecule on T cells, the immune system’s main attack force. Produced in human-derived cells and purified from the culture liquid, this 55-kilodalton protein reliably kept its structure and could be detected with standard biochemical methods. Binding studies showed that it attached strongly to cells with abundant GD2, like A549 and a high-GD2 bone cancer line, and to T cells through CD3, but barely bound to cells with little or no GD2 or CD3. In other words, it acted as a selective molecular bridge between GD2-rich cancer cells and T cells.
Gently waking up immune cells
Next, the researchers checked whether this bridge could activate T cells without harming them. When normal immune cells from healthy donors were exposed to the new protein in the lab, levels of cell death stayed similar to untreated controls and much lower than with a strong non-specific activator. At the same time, the bispecific protein nudged T cells into action: more of them began to divide, and they started expressing surface markers associated with activation. The increase was modest compared to a powerful laboratory stimulant, but clearly above baseline, showing that the engager could awaken T cells in a controlled way.
Forcing a close encounter with lung tumors
The most important test was whether this approach could help T cells kill lung cancer cells more efficiently. The scientists first “armed” T cells by saturating them with the bispecific protein, creating so-called engager-armed T cells. These armed cells and unarmed T cells were then mixed with lung cancer cells showing either low or high GD2 levels. Against the low-GD2 NCI-H460 cells, there was little difference: both types of T cells killed only a limited number of targets. But against the high-GD2 A549 cells, the armed T cells were much more effective, leaving far fewer cancer cells alive after two days of co-culture. Microscopy confirmed that the armed T cells clustered around and destroyed these GD2-rich targets, consistent with the idea that the molecular bridge was pulling the two cell types together.
What this could mean for patients
Overall, the study shows that a carefully designed bispecific protein can safely connect T cells to GD2-rich lung cancer cells, modestly boost T-cell activity, and sharply increase their ability to kill those tumor cells in the lab, especially when the GD2 flag is abundant. While this work is still at a preclinical stage and was done in cell cultures rather than patients, it suggests a new strategy for treating lung cancers that overexpress GD2. In the future, measuring a tumor’s GD2 level could help identify patients most likely to benefit from such targeted immune “matchmakers,” and further animal and clinical studies will be needed to see whether this promise translates into real-world benefit.
Citation: Sawasdee, N., Panya, A., Sujjitjoon, J. et al. Harnessing a bispecific αGD2 × αCD3 protein engager to target GD2-overexpressing lung tumors. Sci Rep 16, 12920 (2026). https://doi.org/10.1038/s41598-026-42885-2
Keywords: lung cancer immunotherapy, GD2 targeting, bispecific engager, T cell redirection, non-small-cell lung cancer