Clear Sky Science · en
Synthesis and characterization of NIR-sensitive curcumin-gelatin nanoparticles for targeted drug delivery in 3D colon cancer
Turning a Spice into a Smart Cancer Fighter
Many people know curcumin as the bright yellow compound in turmeric, a common kitchen spice. Beyond its culinary fame, curcumin can slow the growth of cancer cells in the lab. The problem is that our bodies do not absorb it well, and it breaks down quickly. This study shows how scientists turned curcumin into a "smart" treatment for colon cancer by packaging it inside tiny gelatin particles that release their drug cargo when warmed gently with near‑infrared light.
Why Colon Tumors Are Hard to Treat
Colon cancer remains one of the most common and deadly cancers worldwide. Traditional treatments like surgery, chemotherapy and radiation can save lives but often damage healthy tissues and may fail when tumors become resistant. Researchers are therefore seeking therapies that concentrate their punch inside tumors while sparing the rest of the body. The team behind this paper focused on building a drug system that responds to an external trigger—light—so that most of the action happens only where it is needed, inside or near the tumor in the colon.
Designing Tiny Gelatin Carriers
The scientists built their delivery system from gelatin, a familiar food and medical material that the body can safely break down. They mixed dissolved gelatin and curcumin, then used brief bursts of ultrasound to form nanoparticles—spheres thousands of times smaller than a grain of sand. By carefully tuning the mixing time and the ratio of curcumin to gelatin, they obtained stable particles about 20–40 nanometers across that trapped roughly two‑thirds of the available curcumin. A battery of physical tests confirmed the particles’ size, structure and stability, and suggested that curcumin was held mainly by weak physical forces rather than permanent chemical bonds, making it easier to release on demand. 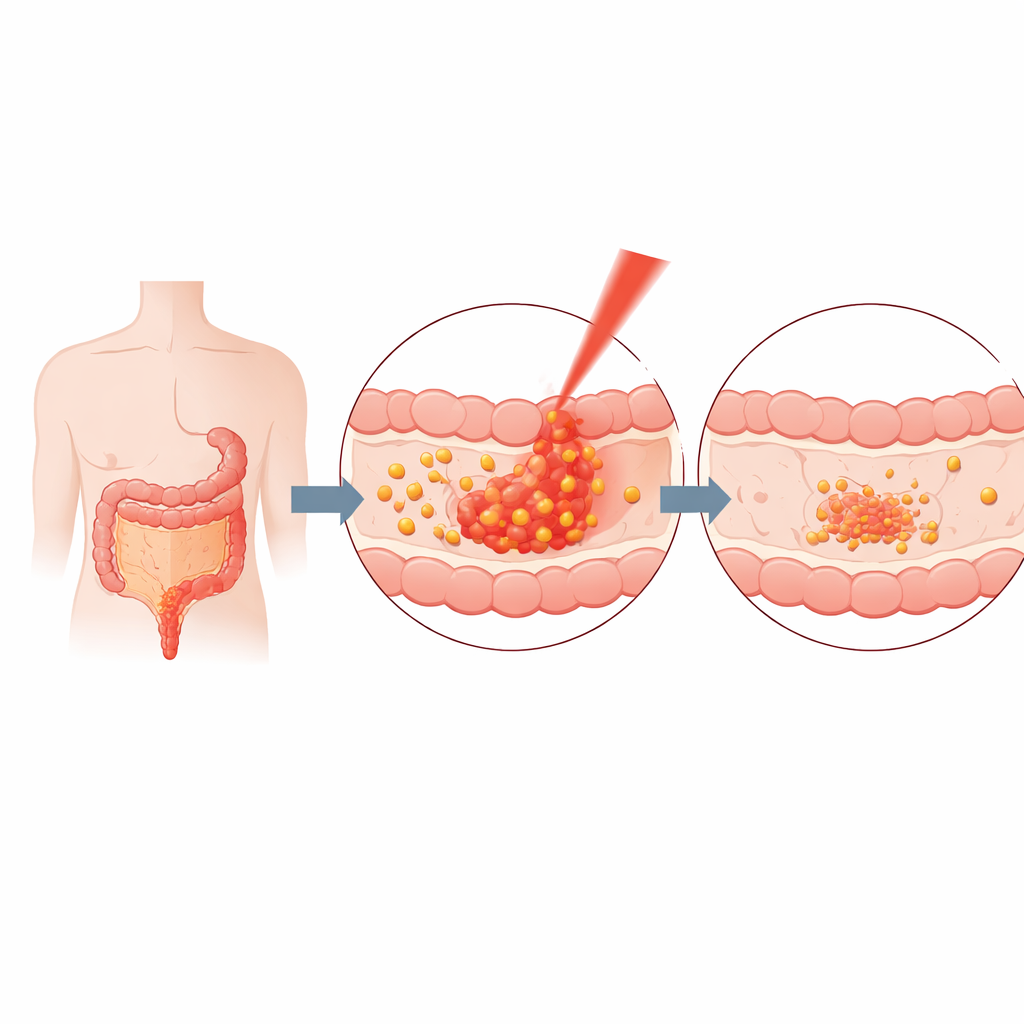
Using Gentle Heat as an On–Off Switch
Next, the team explored how near‑infrared (NIR) light could act as a remote control for drug release. NIR light, which is invisible to the eye, can penetrate tissue more deeply than visible light and is already used in some medical devices. When the curcumin–gelatin particles were exposed to an infrared lamp that warmed them to around 38 °C—just a bit above normal body temperature—their size swelled and they started to leak curcumin more quickly. At this mild temperature, about half of the trapped drug was released, and the overall release over time was up to one‑third faster than without light. Importantly, this effect could be achieved in just 30 seconds of illumination, demonstrating a rapid, temperature‑sensitive switch for turning drug release on. 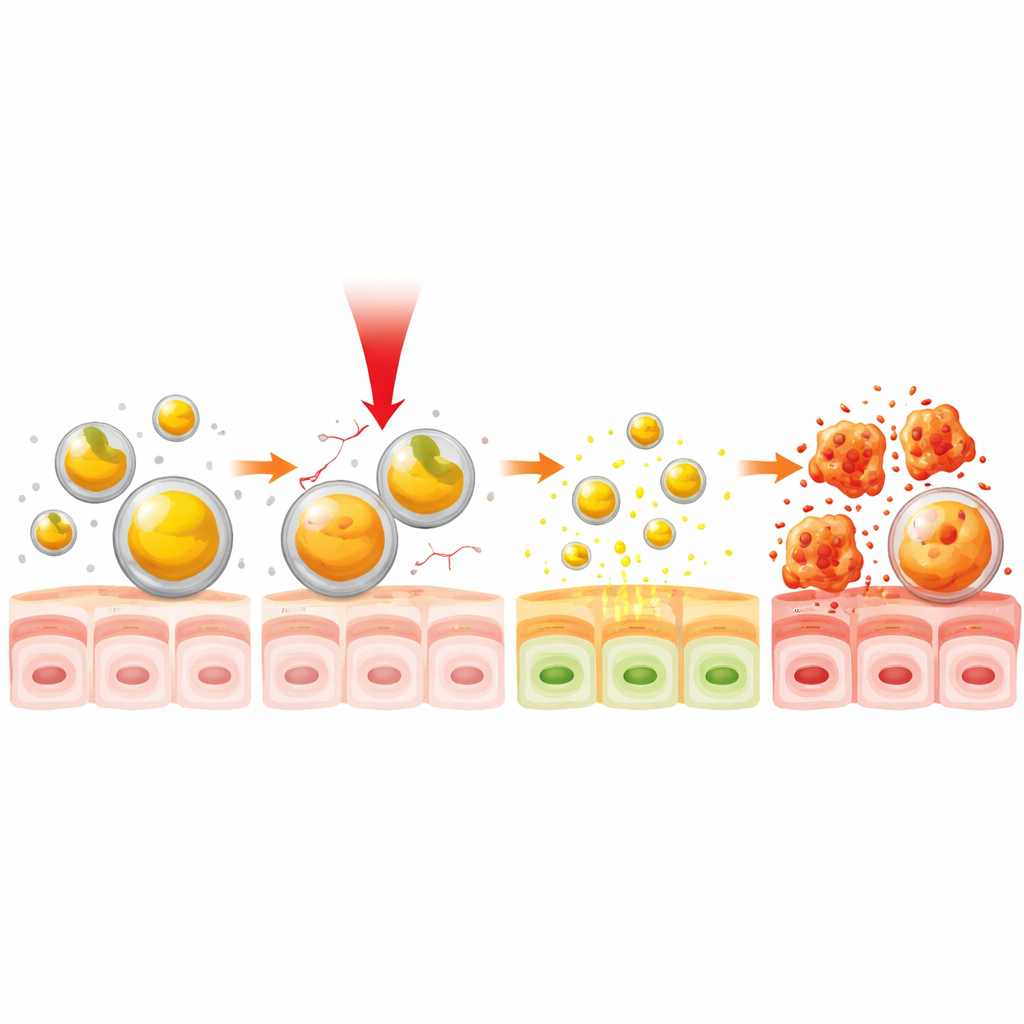
Putting the System to the Test in 2D and 3D Tumor Models
To see whether this light‑activated system actually harmed cancer cells, the researchers tested it on two human colon cancer cell lines and on healthy blood vessel cells grown in dishes. They also built a more realistic three‑dimensional model in which colon cancer cells grew together with endothelial cells, mimicking a mini‑tumor with its supporting blood vessels. Because curcumin glows naturally under certain light, the team could watch the nanoparticles enter cells and accumulate in the cell fluid. At a curcumin‑equivalent dose of 25 micrograms per milliliter, the particles significantly reduced cancer cell survival, movement and invasion, especially when combined with NIR warming. In contrast, healthy cells were much less affected at the same or lower doses, suggesting a degree of selectivity for tumor tissue.
How the Particles Push Cancer Cells Toward Death
Beyond simply counting living cells, the researchers examined how the treatment changed cell behavior. Cancer cells exposed to NIR‑activated curcumin particles showed classic signs of controlled cell death, or apoptosis: their DNA condensed, their nuclei fragmented, and their energy‑producing mitochondria lost their normal electrical charge and shrank. In wound‑healing and Transwell tests that measure how fast cells spread and invade, NIR‑treated particles slowed cancer cell migration and invasion far more than particles without light exposure. These changes are consistent with curcumin’s known ability to lower harmful reactive oxygen molecules and calm inflammatory signals, pathways that help tumors grow and spread.
What This Could Mean for Future Cancer Care
In plain terms, this work turns turmeric’s active ingredient into a targeted, "remote‑controlled" cancer treatment. By hiding curcumin inside biodegradable gelatin spheres and using gentle near‑infrared warming as an on–off switch, the researchers achieved rapid, controlled drug release and strong anti‑tumor effects in realistic lab models, while largely sparing healthy cells. Although much more testing in animals and humans is needed, the study suggests a path toward colon cancer therapies that are more precise, less toxic and potentially easier to produce, opening the door to smarter, light‑guided treatments based on safe, natural compounds.
Citation: Özerkan, D., Danışman-Kalındemirtaş, F. & Kariper, İ.A. Synthesis and characterization of NIR-sensitive curcumin-gelatin nanoparticles for targeted drug delivery in 3D colon cancer. Sci Rep 16, 12167 (2026). https://doi.org/10.1038/s41598-026-42199-3
Keywords: colon cancer, curcumin, nanoparticles, near infrared light, targeted drug delivery