Clear Sky Science · en
Rational design of k-casein peptides to modulate GSK-3B dynamics for Alzheimer’s therapy
Milk Molecules and Memory
Alzheimer’s disease slowly robs people of their memories and independence, and today’s drugs do little to stop the underlying damage in the brain. This study explores an unexpectedly familiar source for new treatment ideas: milk. The authors used advanced computer simulations to redesign a tiny fragment of a milk protein so that it can latch onto a key brain enzyme linked to Alzheimer’s, potentially calming some of the harmful processes that drive the disease.
The Enzyme That Misguides the Brain
In Alzheimer’s, brain cells are harmed by tangled tau fibers, sticky amyloid plaques, and failing connections between neurons. A single enzyme, called GSK-3β, helps drive several of these problems at once. It adds chemical tags to the tau protein, encouraging it to clump into tangles, and it also influences how amyloid is formed, as well as how well nerve cells communicate and respond to insulin-like signals. Because of this central role, many researchers see GSK-3β as a master switch for disease progression and a prime target for new therapies.
Why Look at Milk Proteins?
Milk is more than just fat, sugar, and calcium: it also contains proteins that can be chopped into short fragments, or peptides, with surprisingly strong biological effects. Earlier work suggested that dairy intake might protect against cognitive decline, and that specific milk-derived peptides can shield the brain, reduce harmful enzyme activity, and improve memory in animal models. One of milk’s main proteins, casein, includes a variety of subtypes; among them, κ-casein has been relatively unexplored as a source of brain-protective fragments. The authors set out to see whether a short stretch of κ-casein could be shaped into a potent blocker of GSK-3β.
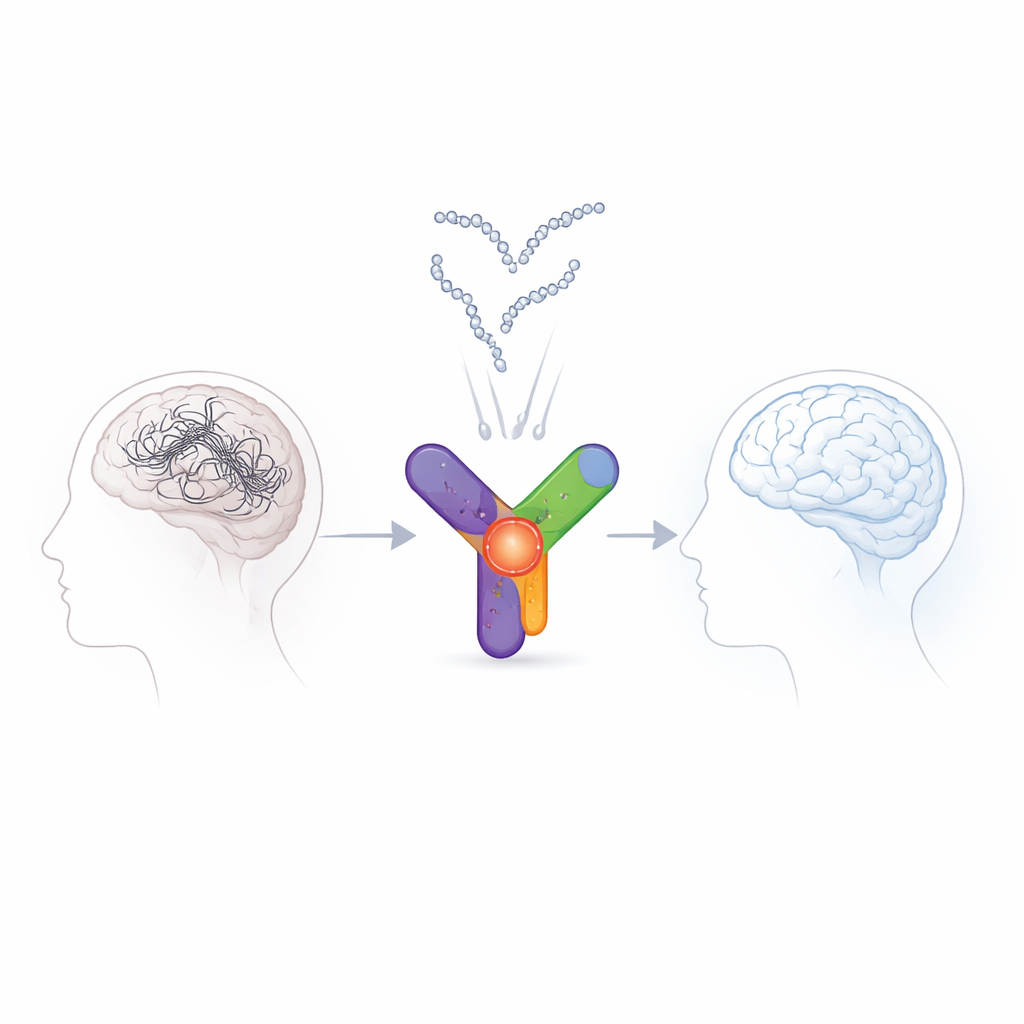
Designing Smarter Peptides on a Computer
The team began by predicting the three-dimensional structure of κ-casein and docking it against GSK-3β to find a portion that nestles into the enzyme’s pocket where energy-carrying molecules normally bind. They found a 10–amino acid segment that already showed decent grip. Using detailed energy calculations, they pinpointed four positions within this segment that contributed weakly or even unfavorably to binding. Then, with a mutation-prediction tool, they systematically swapped these four building blocks for alternatives likely to strengthen the interaction, building a virtual library of 48 new peptide variants. Safety checks removed any candidates predicted to be toxic or allergenic, leaving 22 that were then re-docked onto GSK-3β.
Narrowing Down the Most Promising Candidates
Docking scores and interaction maps highlighted four standout peptides, labeled PEP8, PEP36, PEP40, and PEP44. All four shared two key changes that replaced a neutral, oily residue near one end with a more interactive one, helping them anchor more firmly in the enzyme’s pocket. PEP8 and PEP44 went further, adding a negatively charged residue at another position, which boosted their grip. Computer simulations run over hundreds of nanoseconds showed that when PEP8 or PEP44 is bound, the enzyme’s structure becomes slightly more compact and less flexible, especially around the pocket where its natural targets would usually sit. These peptides made more contact points with the enzyme than the original milk fragment, suggesting higher strength and better selectivity. Energy analyses confirmed that PEP8 and PEP44 formed the tightest complexes, driven mainly by close-range attractions and electrical forces between the peptide and the enzyme.
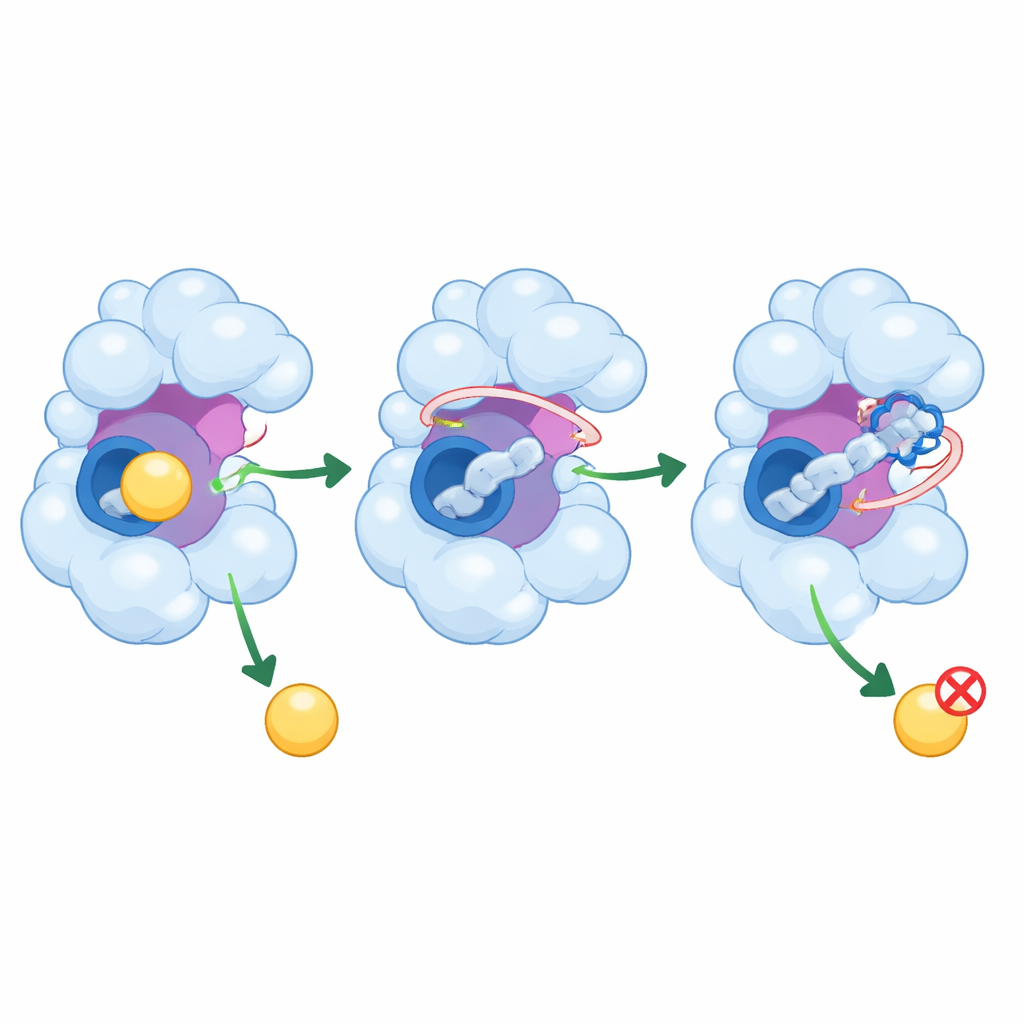
How These Peptides Might Calm a Runaway Enzyme
Beyond simply plugging the central pocket, PEP8 and PEP44 reach into neighboring regions of GSK-3β that help control its activity. They interact with a flexible loop that guides incoming molecules and, in the case of PEP44, with a catalytic residue that is crucial for the enzyme’s chemistry. The simulations show that these interactions reduce the wiggling of important parts of the protein, shrinking the range of shapes the enzyme can adopt. With its moving parts partly locked down and its pocket more crowded, GSK-3β is less able to welcome its usual partners, which could translate into reduced production of harmful tau modifications and amyloid-related changes in real cells.
What This Could Mean for Future Treatments
While this work was done entirely in silico—inside computers rather than in living brains—it outlines a clear path from a natural food protein to carefully tuned peptide candidates that could, in principle, dial down a major Alzheimer’s driver. PEP8 and PEP44 stand out as the strongest binders, reshaping the enzyme’s dynamics in ways that should hinder its damaging actions. The next steps will require laboratory testing to confirm whether these designer milk fragments can reach the brain, remain stable in the body, and truly protect nerve cells. If they do, they could form the basis of a new class of gentle, multitasking treatments inspired by everyday nutrition.
Citation: Moghaddam, N., Ramazani, A. & Zarei, A. Rational design of k-casein peptides to modulate GSK-3B dynamics for Alzheimer’s therapy. Sci Rep 16, 12768 (2026). https://doi.org/10.1038/s41598-026-42103-z
Keywords: Alzheimer’s disease, GSK-3β inhibition, milk peptides, κ-casein, computational drug design