Clear Sky Science · en
The association between early pregnancy infection with SARS-CoV-2 and fetal birth defects: a prospective study
Why this study matters for expectant families
Many people who are pregnant, or hope to be, still wonder what a COVID-19 infection might mean for their baby. This study followed more than a thousand women in China who were in the very earliest weeks of pregnancy during a major wave of SARS-CoV-2 infections. By carefully tracking their health and their babies’ development, the researchers set out to answer a pressing question: does catching the virus in the first trimester raise the chance of birth defects?
Who was studied and how
The research team worked with three large hospitals and invited women who were in their first 13 weeks of pregnancy between late 2022 and early 2023 to take part. All had been vaccinated against COVID-19 before becoming pregnant. Using PCR tests, the doctors divided participants into two groups: 535 women who tested positive for SARS-CoV-2 during early pregnancy, and 592 who remained virus‑negative throughout. Women with known genetic conditions, serious illnesses, or exposure to other causes of birth defects (such as certain drugs or toxins) were excluded, so that the focus stayed on the effects of the virus itself. The team then followed the pregnancies by reviewing medical records and making follow‑up phone calls after birth.
What COVID-19 was like for these pregnant women
Among the infected women, illness was generally mild. Most had short‑lived fevers, cough, sore throat, or muscle aches, and none required hospital care specifically for COVID-19. Many took common fever and pain medicines that are considered acceptable in pregnancy under Chinese guidelines, and some used traditional remedies. The researchers compared women whose babies had birth defects with those whose babies developed typically and found no meaningful differences in how high their fevers were, how long they lasted, or whether they used medications. This suggests that the infection itself, rather than fever or treatment, is the more likely driver of any increased risk.
What happened to the babies
When the pregnancies ended in birth, miscarriage, or stillbirth, doctors assessed the babies for structural or chromosomal problems. Overall, birth defects were more frequent among babies whose mothers had been infected in early pregnancy. In singleton pregnancies (one baby, not twins), about 9 in 100 babies in the infected group had a birth defect, compared with about 5 in 100 in the non‑infected group. The most common problems involved the heart and major blood vessels: these cardiovascular abnormalities appeared roughly twice as often after early infection. Other defects, such as those affecting the urinary system, face, skeleton, or nervous system, were less frequent and did not clearly differ between groups. Twin pregnancies themselves carried a higher risk of defects, regardless of infection, which is consistent with previous medical knowledge.
Clues about how the virus might affect development
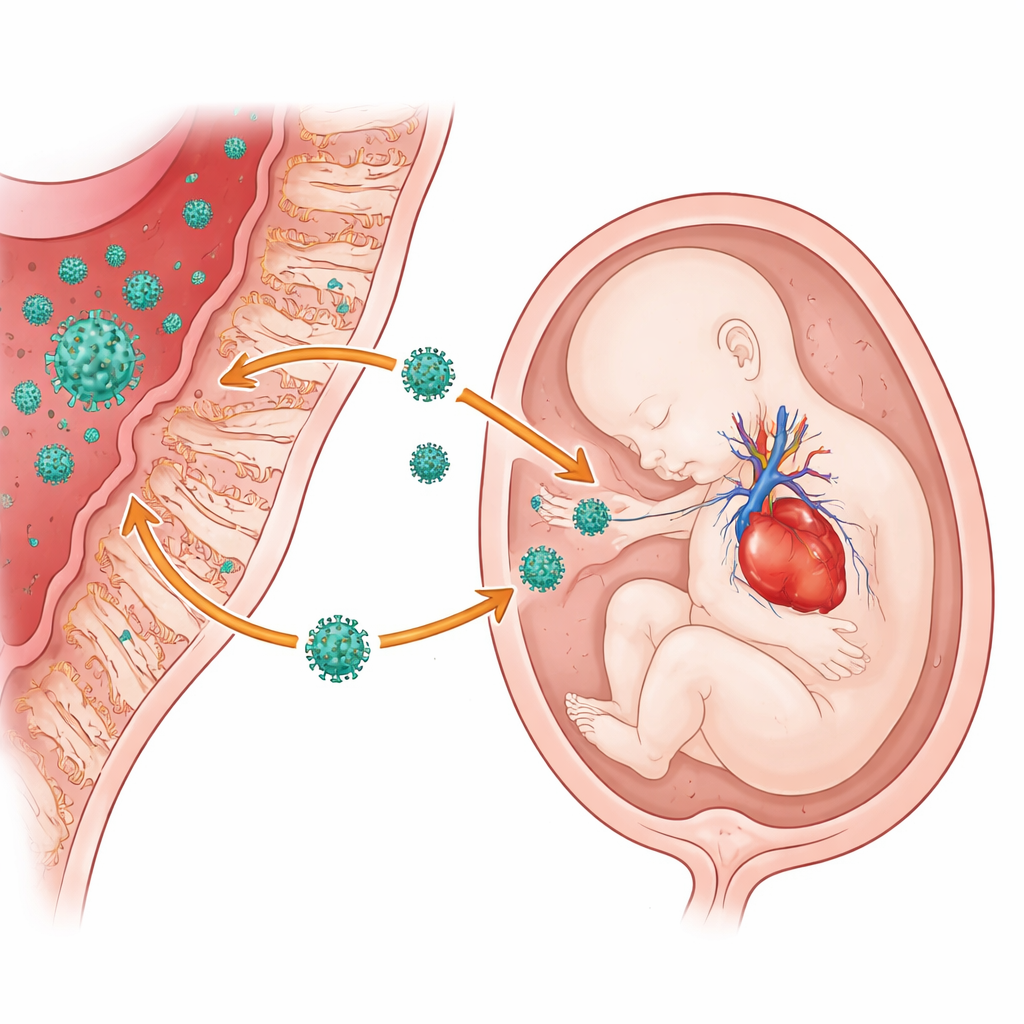
The study was not designed to prove exactly how SARS-CoV-2 might interfere with a developing fetus, but the authors discuss several plausible pathways based on earlier work. Viral infections can inflame the placenta, disturbing the hormone and signaling environment that guides early brain and organ formation. The virus may also cross the placenta and infect embryonic tissues directly; other research has detected viral material in amniotic fluid, umbilical cord tissue, and even in early embryonic cells. One protein the virus uses to enter cells is abundant in developing heart muscle, suggesting a possible reason why heart defects stood out in this study. The researchers also note that subtle changes in gene regulation, triggered by viral competition with the fetus’s own RNA‑binding proteins, could disturb organ development in more complex ways that are not yet fully visible at birth.
What this means for pregnancy care
To analyze risk more precisely, the team used statistical models that accounted for maternal age and whether a pregnancy involved twins. After these adjustments, early SARS-CoV-2 infection remained linked to more than double the risk of birth defects, while twin pregnancy carried an even higher independent risk. Importantly, there was no evidence that having both factors together multiplied the danger beyond what each contributed on its own. The study did not find clear increases in preterm birth, cesarean delivery, or miscarriage tied specifically to early infection, suggesting that its main impact may be on organ formation rather than on the timing or mode of delivery.
Take‑home message for non‑specialists
This work supports a cautious but not alarmist view. For an individual pregnant woman, the chance of a baby being born with a defect after early COVID-19 infection is still relatively low; most babies in the infected group were born healthy. At the same time, the link to a higher rate of birth defects—especially those involving the heart—means that early pregnancy remains a period when avoiding infection is particularly important. For those who do become infected in the first trimester, the findings argue for careful prenatal monitoring, including detailed ultrasound checks of the baby’s heart and other organs. The authors call for longer follow‑up and broader studies, but their results already offer practical guidance: early protection against viral infection and attentive prenatal care may help reduce avoidable risks to the next generation.
Citation: Luo, J., Liu, P., chen, P. et al. The association between early pregnancy infection with SARS-CoV-2 and fetal birth defects: a prospective study. Sci Rep 16, 10001 (2026). https://doi.org/10.1038/s41598-026-39855-z
Keywords: early pregnancy, COVID-19, SARS-CoV-2, birth defects, fetal heart development