Clear Sky Science · en
GPR109a-AMPK axis mediates the Attenuation of uric acid-induced M1 macrophage polarization by β-hydroxybutyrate from Lacticaseibacillus rhamnosus M2b
Why gut bugs matter for sore joints
People with high uric acid in their blood often develop painful gout and other inflammatory problems, yet current treatments mostly focus on lowering uric acid rather than calming the immune system it agitates. This study explores how a helpful gut bacterium and one of its small chemical products may gently nudge immune cells away from a harmful, attack mode toward a soothing, repair mode, offering a fresh angle on easing inflammation linked to high uric acid.
From high uric acid to angry immune cells
Hyperuricemia, or high uric acid, is increasingly common worldwide and is tied not only to gout but also to broader chronic inflammation. Uric acid can push certain white blood cells called macrophages into an aggressive state that pours out inflammatory substances, damaging nearby tissues. Researchers know this aggressive state, often called the M1 type, plays a central role in disease, but therapies that directly and safely steer these cells back toward a calmer, healing state are still limited.
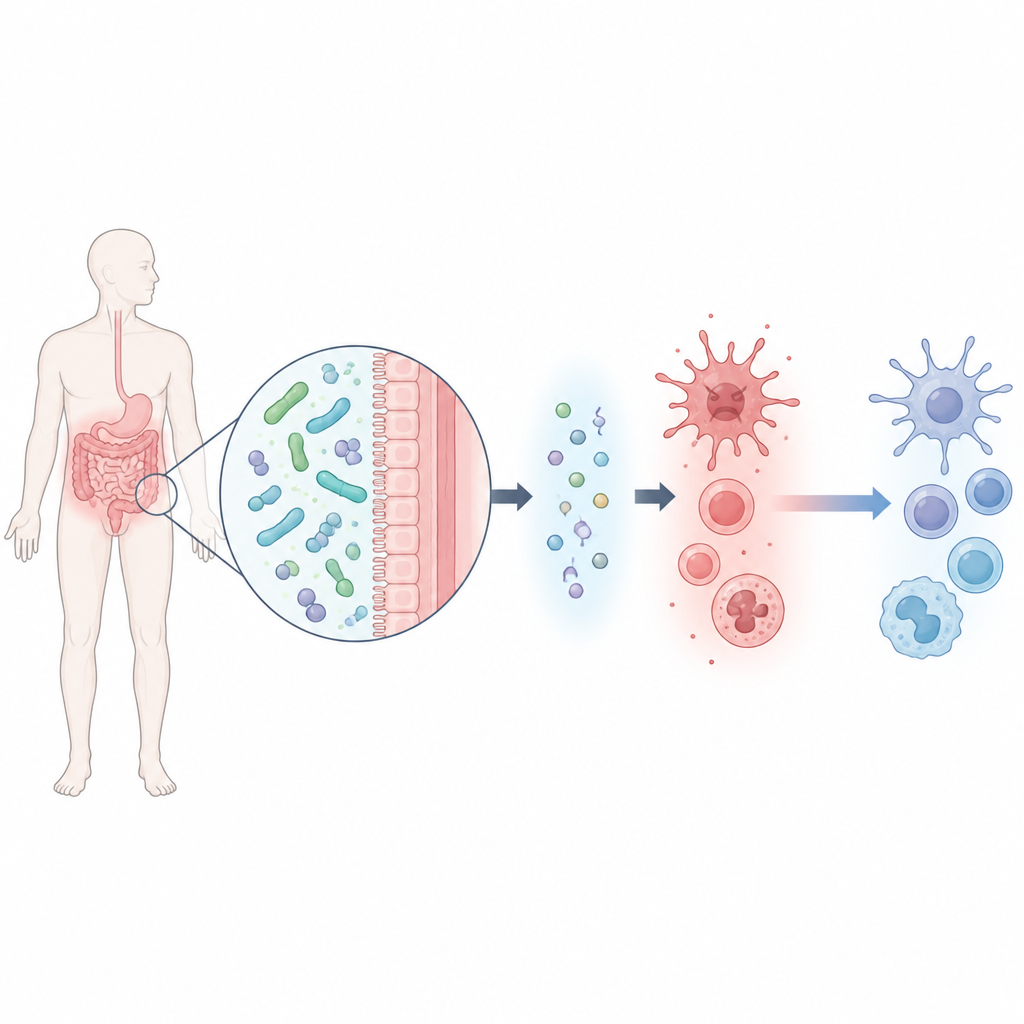
A helpful gut microbe and a small chemical signal
The team began by isolating a strain of Lacticaseibacillus rhamnosus, named M2b, from the stool of healthy men with low uric acid levels. This bacterium excelled at breaking down uric acid in the lab. Using a broad survey of the chemical byproducts the microbe released, the scientists identified six candidate molecules and tested which ones could protect mouse macrophage cells that were stressed by uric acid. One molecule, called beta hydroxybutyrate, stood out: it not only improved cell survival but also sharply reduced several key inflammatory substances while boosting an anti inflammatory signal called IL 10.
Turning attack cells into healing cells
To see how this small molecule reshaped immune behavior, the researchers exposed macrophages to uric acid alone or together with beta hydroxybutyrate or liquid taken from M2b cultures. Uric acid alone drove the cells into a classic attack posture, with high levels of markers linked to the M1 state and low levels of markers tied to a more soothing, M2 like state. When beta hydroxybutyrate or the bacterial culture liquid was added, these patterns flipped: the attack markers dropped, the healing markers rose, and the cells’ shapes under the microscope shifted toward a calmer appearance. This showed that a metabolite made by a gut bacterium could directly tilt immune cells away from a damaging response.
A receptor switch and an energy sensor inside the cell
The story did not end with observing these changes; the scientists wanted to know how they happened. They focused on a protein on the macrophage surface known as GPR109a, which can sense molecules like beta hydroxybutyrate. Computer modeling suggested that beta hydroxybutyrate fits snugly into this receptor. Experiments confirmed that uric acid lowered GPR109a levels, while beta hydroxybutyrate and the M2b culture restored them. When the researchers deliberately reduced GPR109a in the cells, beta hydroxybutyrate lost its calming effects. When they boosted GPR109a, the cells naturally shifted toward the healing state, even under uric acid stress.
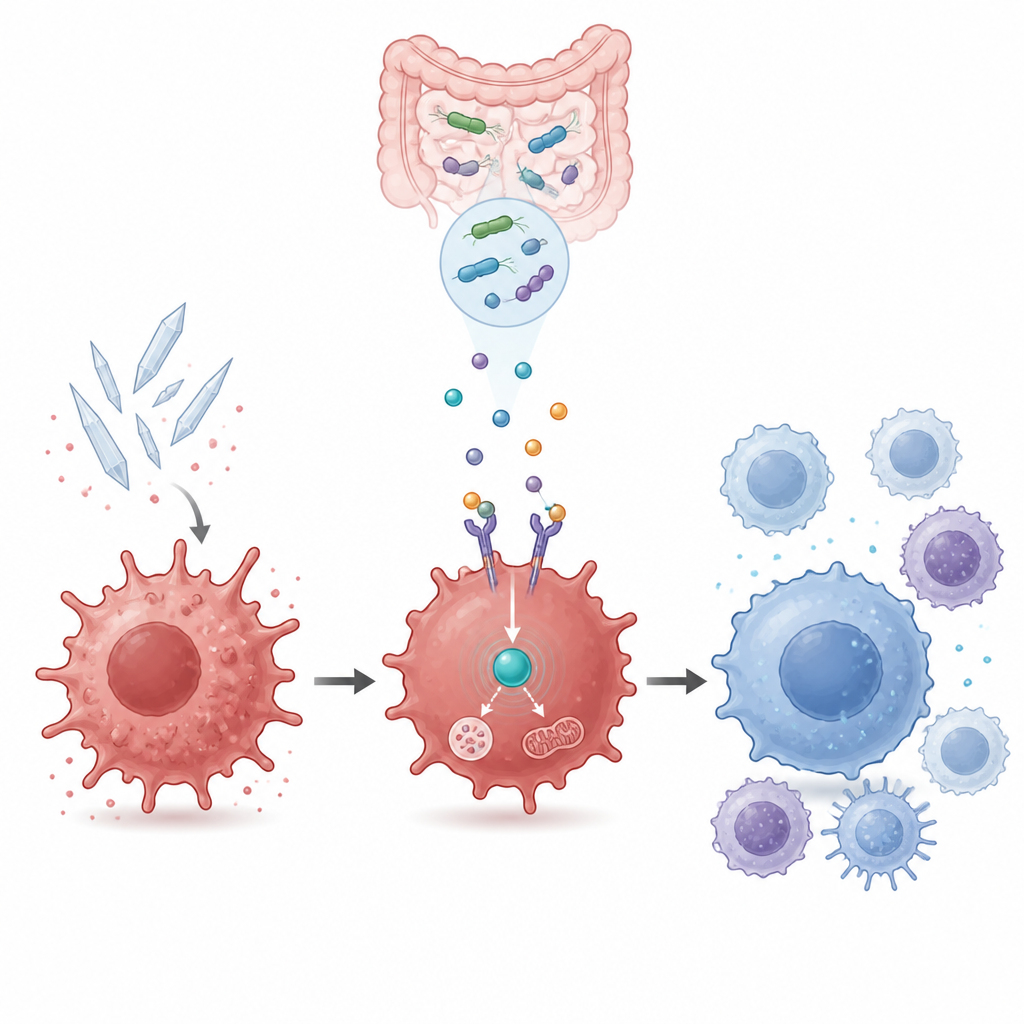
Linking cell energy use to inflammation control
Inside the cell, GPR109a connects to another key player called AMPK, a sensor of the cell’s energy status. Gene activity measurements and protein tests showed that uric acid dulled AMPK activity, while beta hydroxybutyrate revived it in a way that depended on GPR109a. Blocking AMPK with a chemical inhibitor erased the benefits of beta hydroxybutyrate and the M2b culture: inflammatory markers surged again, and healing markers faded. This tied the calming immune shift to an internal energy sensing circuit that must remain active for the bacterial metabolite to work.
What this could mean for people with high uric acid
Taken together, the findings sketch a simple chain of events: a gut bacterium makes beta hydroxybutyrate, this small molecule activates the GPR109a receptor on immune cells, which in turn switches on AMPK and helps move macrophages from a harmful attack mode to a more peaceful, repair focused mode. While the work was done in cultured mouse cells and still needs testing in animals and people, it points to the gut microbe metabolite GPR109a AMPK route as a promising target for easing inflammation in hyperuricemia, potentially complementing standard uric acid lowering drugs with more precise immune calming strategies.
Citation: Du, M., He, Y., Zhu, Y. et al. GPR109a-AMPK axis mediates the Attenuation of uric acid-induced M1 macrophage polarization by β-hydroxybutyrate from Lacticaseibacillus rhamnosus M2b. Sci Rep 16, 14955 (2026). https://doi.org/10.1038/s41598-026-39746-3
Keywords: hyperuricemia, gut microbiota, macrophage polarization, beta hydroxybutyrate, GPR109a AMPK pathway