Clear Sky Science · en
CAF-enriched COL4A6 promotes gastric cancer progression and stromal remodeling despite an inverse association in bulk transcriptomes
Why the Tumor’s Surroundings Matter
Most people think of cancer as a disease of rogue cells, but this study shows that the “soil” around the tumor can be just as important as the cancer cells themselves. In gastric (stomach) cancer, the tissue scaffolding and support cells surrounding the tumor can either slow the disease or push it to grow, spread, and resist treatment. This research uncovers how a particular scaffold protein, called COL4A6, behaves very differently depending on where it is located, and how a specific group of support cells, cancer-associated fibroblasts, can turn this protein into a powerful accomplice of tumor progression.
A Closer Look at the Tumor Neighborhood
The authors began by mapping the “landscape” of the extracellular matrix—the mesh of proteins and fibers that surrounds cells—in hundreds of human stomach tumors. They found that nearly half of the core matrix genes were altered in cancer tissue compared with nearby normal tissue, confirming that the physical environment of gastric tumors is heavily remodeled. Using these genes, they built a Matrisome Risk Score that could reliably distinguish tumor from normal tissue and separate patients into two main groups: one with a dense, highly remodeled matrix and worse survival, and another with a looser matrix and better outcomes. The high-matrix group also showed signs of an immune “cold” tumor, with many suppressive cells and few active fighter T cells, as well as reduced sensitivity to standard chemotherapy.
A Protein with a Split Personality
One matrix component, COL4A6, stood out because it appeared to behave like a “protective” factor when the researchers looked only at bulk gene readouts from whole tumor samples. Statistically, higher COL4A6 gene levels in these bulk datasets were linked to lower apparent risk, which had led some to suspect it might help restrain cancer. But this study suspected a hidden twist: bulk measurements mix signals from many different cell types. When the team examined actual tumor tissue slices from patients, they found that COL4A6 protein was in fact strongly increased in cancers compared with normal stomach tissue, and that collagen fibers became thicker and more abundant as tumors advanced.
Fibroblasts as Hidden Drivers
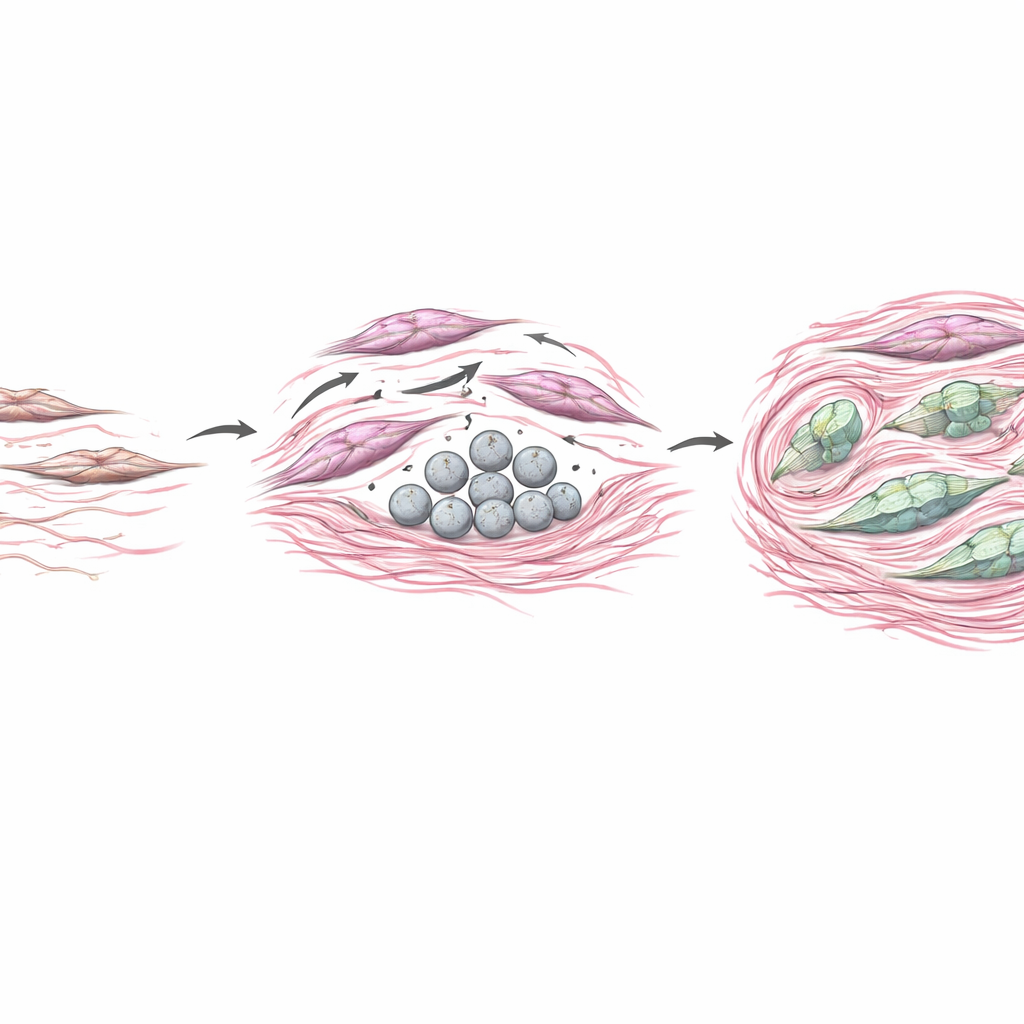
Using fluorescent imaging and single-cell sequencing, the researchers discovered where COL4A6 was really coming from. It was not the cancer cells themselves, but primarily a type of support cell called cancer-associated fibroblasts, or CAFs, nestled in the tumor’s connective tissue. COL4A6 signals overlapped tightly with a CAF marker, revealing that these fibroblasts were the main source. This explained the earlier confusion: while cancerous epithelial cells may lose COL4A6 expression at the gene level, the fibroblast compartment ramps up production and deposits the protein into the surrounding matrix, a pattern that bulk tests blur together.
How COL4A6 Fuels Growth and Spread
To test how CAF-derived COL4A6 affects tumors, the team grew gastric cancer cells together with fibroblast cultures engineered to either produce more COL4A6 or less of it. When fibroblasts overproduced COL4A6, nearby cancer cells became more mobile and invasive, and they shifted into a more aggressive, shape-shifting state known as epithelial–mesenchymal transition. In mice, tumors formed with COL4A6-rich fibroblasts grew larger, had denser collagen deposits, and showed strong COL4A6 signals woven through the stroma. At the same time, COL4A6 appeared to act back on the fibroblasts themselves, reorganizing their internal scaffolding into thick stress fibers and boosting classic activation markers. In essence, COL4A6 helped convert fibroblasts into powerful remodelers that stiffen the tissue, build physical barriers, and create a niche that supports tumor invasion and drug resistance.
What This Means for Patients
For non-specialists, the main message is that the same molecule can look helpful or harmful depending on how and where it is measured. COL4A6 may seem “protective” when averaged across all cells in a tumor, but when viewed in the right compartment it emerges as a key promoter of disease, produced by activated fibroblasts that build a dense, hostile environment. By defining matrix-based tumor subtypes and exposing this CAF–COL4A6 axis as a driver of immune evasion and chemotherapy resistance, the study suggests new ways to stratify gastric cancer patients and highlights the tumor stroma as a promising target. Therapies that disrupt COL4A6 in fibroblasts or soften the surrounding matrix might one day help turn cold, treatment-resistant stomach tumors into ones that are more responsive to drugs and immunotherapy.
Citation: Sun, Q., Wei, S., Yao, J. et al. CAF-enriched COL4A6 promotes gastric cancer progression and stromal remodeling despite an inverse association in bulk transcriptomes. Sci Rep 16, 10399 (2026). https://doi.org/10.1038/s41598-026-38120-7
Keywords: gastric cancer, tumor microenvironment, cancer-associated fibroblasts, extracellular matrix, COL4A6