Clear Sky Science · en
Modular engineering of thermoresponsive allosteric proteins
Turning Heat into a Gentle On–Off Switch
Imagine being able to control the activity of almost any protein in a living cell simply by warming it a couple of degrees—no drugs, no strong light, just a tiny change in temperature. This study shows how researchers can build such heat-sensitive protein "switches" in a modular way, using parts that can be plugged into many different proteins. The result is a new toolkit that could one day help guide gene therapies, fine‑tune microbial treatments, or explore how cells work, all by exploiting the narrow temperature range around normal body heat.
Why Temperature Is a Powerful Invisible Remote
Scientists already use light and chemicals to turn proteins on and off inside cells, but both have practical drawbacks. Light often does not penetrate tissues deeply, and chemicals can spread slowly and affect many parts of the body at once. Temperature, by contrast, penetrates tissues well and can be focused very precisely in space and time. The challenge has been that most existing thermal tools either work only at extreme temperatures or mainly control gene expression indirectly. The authors set out to create a general strategy that would let them take an ordinary protein and give it a sharp response to small temperature shifts in the range of 37–41 °C—roughly the difference between normal body temperature and a mild fever.
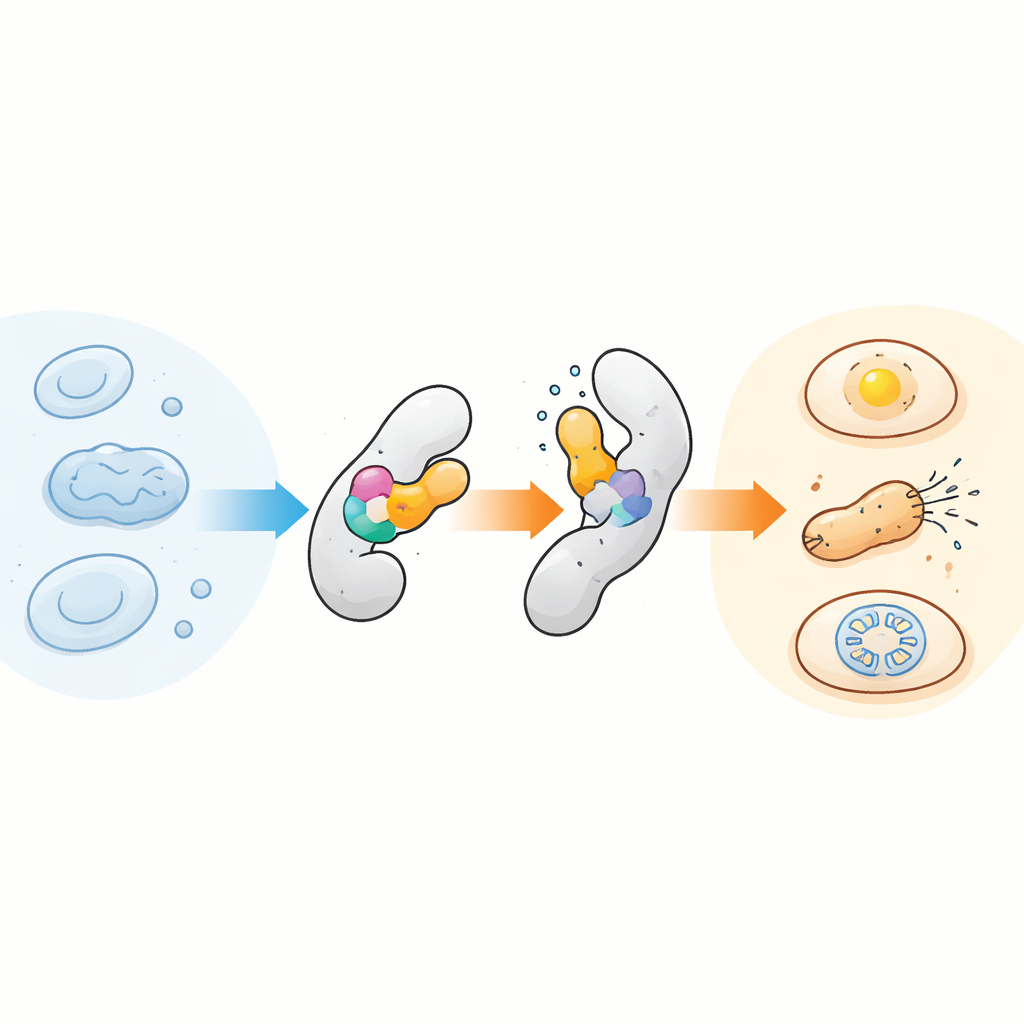
Plugging a Tiny Sensor into Many Proteins
The core idea is deceptively simple: insert a compact temperature‑sensitive sensor domain into another protein so that a small structural change in the sensor is transmitted to the host protein, acting like an internal hinge. The team focused on a well‑studied module called AsLOV2, originally a light sensor from oats, because it naturally flips between different shapes. They discovered that this domain is also surprisingly sensitive to temperature, especially when slightly destabilized by specific mutations. By inserting tailored versions of AsLOV2 into different positions of target proteins, they created chimeric proteins whose activity sharply switched on or off over just a few degrees. In bacteria, they first showed this with AraC, a transcription factor, and then evolved AsLOV2 variants that made the temperature response steeper, tunable, and even reversible over time.
From Bacterial Kill Switches to Molecular Thermostats
Once the approach worked for one protein, the authors applied it to others with very different jobs. Inserting the sensor into an antibiotic resistance enzyme turned it into a heat‑triggered kill switch: bacteria grew at 37 °C but died at 41 °C when exposed to the drug. A similar strategy attached the sensor to a component of a CRISPR‑based gene activator in bacteria, allowing gene expression to be dialed up or down by gentle heating. The team also explored a related trick in which short signaling peptides are hidden or exposed by heat‑induced movements of the sensor’s tail, enabling temperature‑dependent protein degradation in bacteria and modest control of protein trafficking between the nucleus and cytoplasm in human cells.
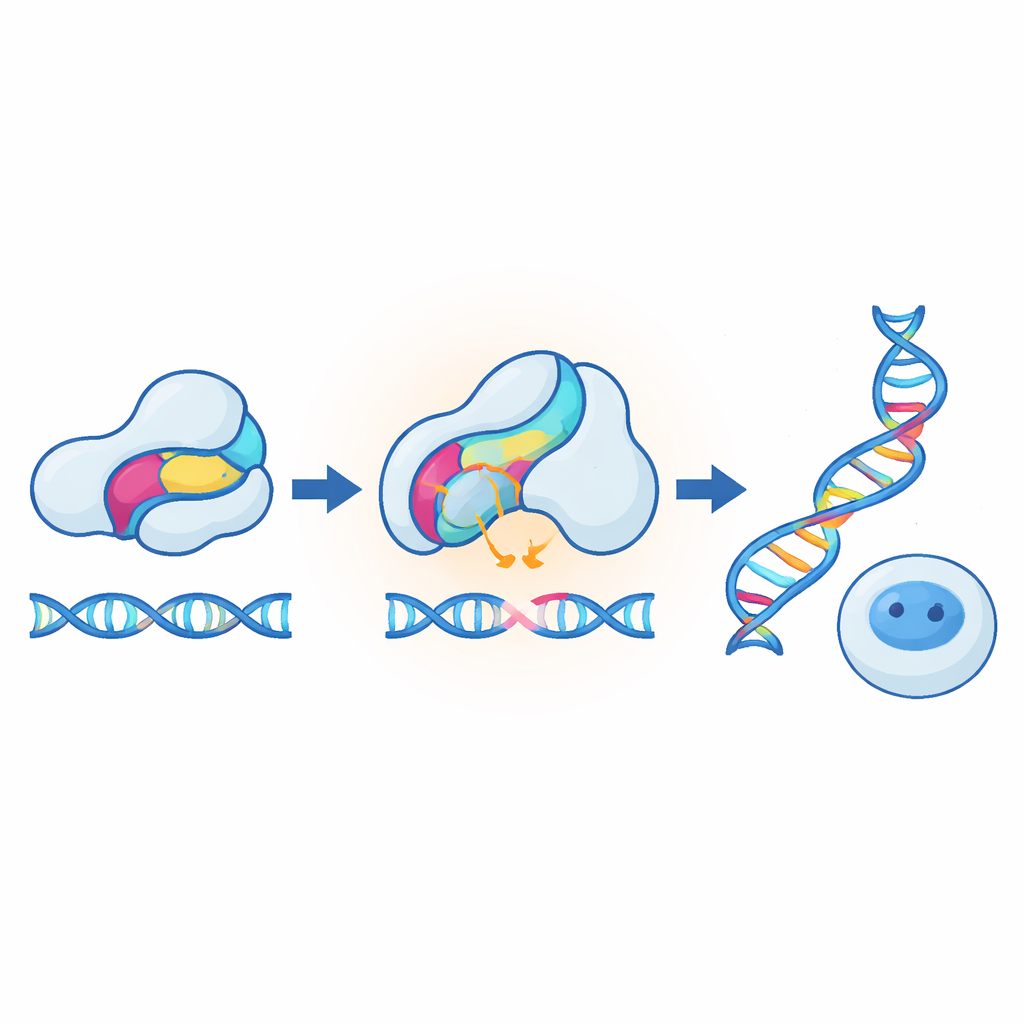
Heat‑Tuned Control of Genome Editing in Human Cells
The most striking demonstrations came from adapting the method to CRISPR tools in mammalian cells. In one design, the sensor was inserted into an anti‑CRISPR protein that normally blocks a genome editor; warming the cells weakened the inhibitor, allowing editing to rise several‑fold between 37 °C and 40 °C. In another design, the sensor was built directly into Cas9 itself, so that heating turned editing down instead of up. Both modes worked on multiple target genes, with little impact on cell health, and could also be transferred to CRISPR‑based transcriptional activators. The researchers then showed that a completely different receptor module—a cortisol‑sensing domain from a hormone receptor—could serve as an alternative temperature‑sensitive insert, especially when the hormone was present. This suggests that many natural sensor domains, not just light receptors, may become unstable and switch‑like when pushed slightly above their usual operating temperatures.
What This Means for Future Therapies and Tools
To a layperson, the takeaway is that the authors have built a general recipe for turning ordinary proteins into precise biological thermostats. By inserting small sensor domains and fine‑tuning their stability, they can make protein activity rise or fall abruptly at chosen temperatures within the normal human range. Because the strategy is modular, it should be compatible with many existing optogenetic and chemogenetic designs, and it can work directly at the level of protein function rather than just gene expression. In the long run, such temperature‑responsive switches could help make gene editing safer and more controllable, enable heat‑guided bacteria that self‑destruct outside desired conditions, and provide biologists with subtle new ways to probe how cells respond to the slightest change in warmth.
Citation: Kroell, AS., Hoffmann, K.H., Motzkus, N.A. et al. Modular engineering of thermoresponsive allosteric proteins. Nat Chem Biol 22, 751–758 (2026). https://doi.org/10.1038/s41589-026-02151-y
Keywords: thermogenetics, protein switches, CRISPR control, allosteric regulation, temperature sensing