Clear Sky Science · en
Biallelic variants in the noncoding RNA gene RNU4-2 cause a recessive neurodevelopmental syndrome with distinct white matter changes
Why this rare genetic story matters
Most of us think of genes as blueprints for proteins, but some genes work quietly behind the scenes, helping cells edit other genes’ messages. This paper uncovers a new childhood brain disorder caused not by a broken protein, but by tiny changes in a noncoding RNA gene called RNU4-2. By following families around the world, the researchers show how inherited changes in this gene disrupt brain wiring, produce a recognizable MRI pattern, and differ from a better-known, related condition. The work illustrates how subtle faults in the cell’s RNA editing machinery can reshape brain development—and how new genomic tools are finally revealing these hidden diseases.
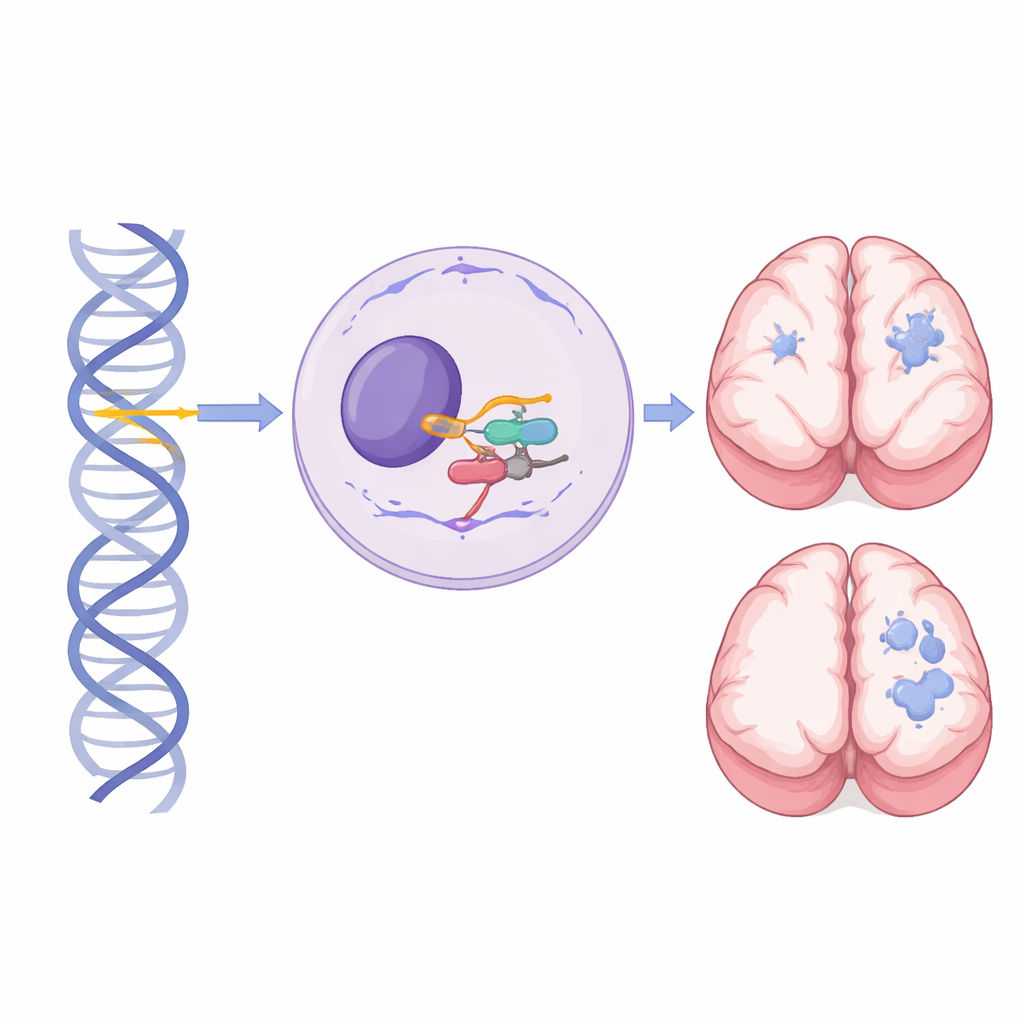
A small gene with a big role in brain development
Inside every cell, a large machine called the spliceosome cuts and pastes RNA messages so that genes can be turned into working molecules. RNU4-2 encodes U4 small nuclear RNA, a critical component of the major spliceosome, which handles nearly all introns in our genes. Previously, single new mutations in a short, central stretch of RNU4-2 were shown to cause a common dominant neurodevelopmental condition known as ReNU syndrome: a child is affected even if only one copy of the gene is altered. In contrast, this study focuses on people who inherited damaging variants in both copies of RNU4-2—one from each parent—revealing a distinct, recessive syndrome.
Finding families and a shared clinical picture
The team searched large rare-disease sequencing projects and biobanks to find individuals with two RNU4-2 variants and neurodevelopmental problems. They ultimately identified 38 people from 28 families, and gathered detailed clinical data for 31 of them. Nearly all had global developmental delay or intellectual disability, often in the moderate to severe range, with marked speech delay; about half had no meaningful spoken language. Many children walked late, and more than a quarter never walked independently by age five. Low muscle tone in infancy, seizures, movement and coordination problems, and behavioral challenges such as aggression or obsessive traits were common. Skin changes, eye problems, short stature, and genital anomalies in males appeared in a substantial minority, forming a multi-system picture rather than a purely brain-limited disease.
Brain scans reveal a striking white matter signature
Magnetic resonance imaging provided one of the clearest clues that this recessive condition is distinct from dominant ReNU syndrome. Among 27 individuals with scans or reports, 24 had abnormalities, and a pediatric neuroradiologist reviewed 13 scans in depth. Every reviewed scan showed white matter involvement, most characteristically an expansion of the fluid-filled spaces that surround small blood vessels (perivascular spaces) in the deep and periventricular white matter. In the most severe cases, these spaces coalesced into tightly packed, microcyst-like clusters, often accompanied by thinning of the corpus callosum and shrinkage of the cerebellum. This dramatic pattern had not been reported in ReNU syndrome and was rarely seen in people with RNU4-2 variants from the general population, making it a powerful radiologic signature that can prompt targeted genetic testing.
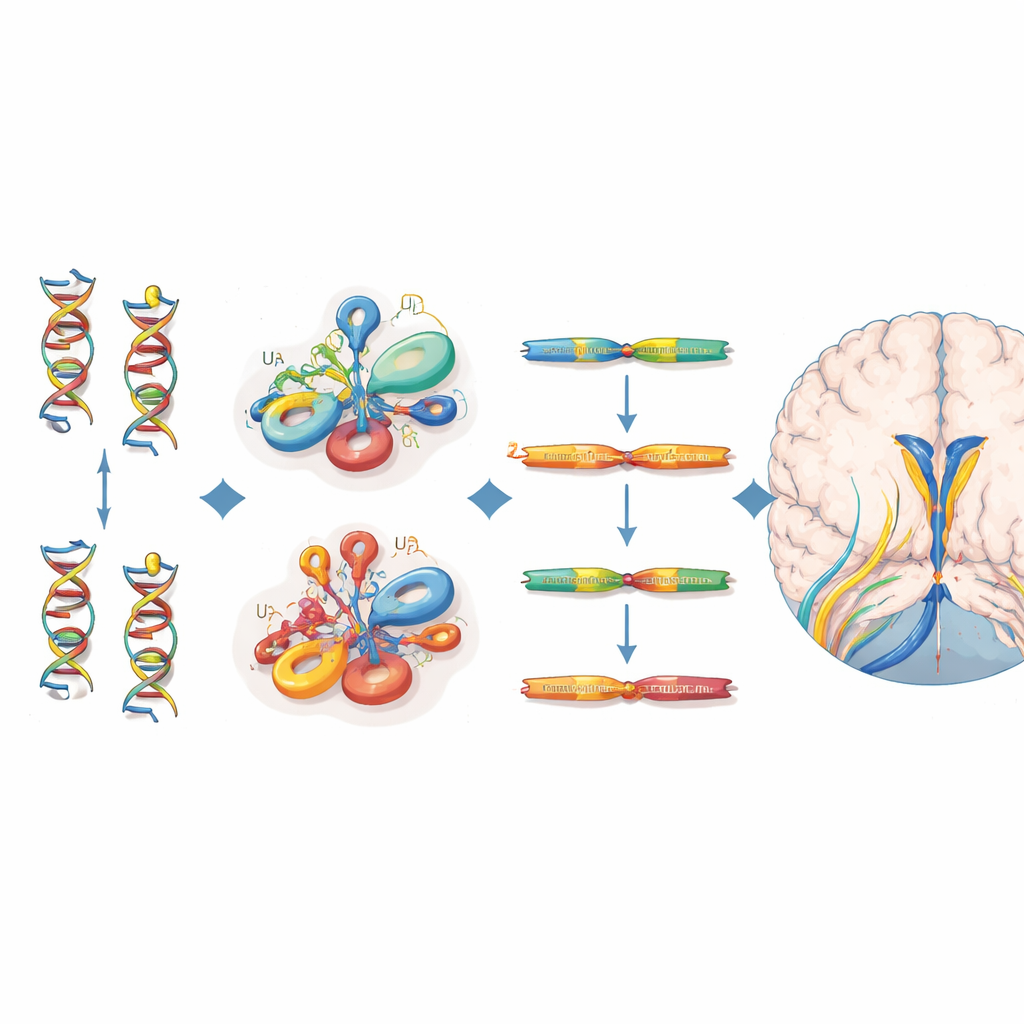
Pinpointing the mutations and their molecular impact
All 38 affected individuals carried biallelic variants that clustered in specific structural regions of the U4 RNA—particularly the Stem II region that binds to U6 RNA, a sharp bend called the k-turn that recruits proteins, and a stretch required for binding stabilizing Sm proteins. Many of these positions line up with equivalent sites in a related RNA, RNU4ATAC, where mutations are already known to cause other recessive disorders. To gauge how harmful each variant is, the authors drew on a saturation genome editing experiment that had measured how hundreds of possible RNU4-2 changes affect cell survival. Variants in patients with neurodevelopmental disease tended to have lower function scores than those seen in healthy volunteers from the UK Biobank, supporting their role in disease, even though not every clinically important variant met a strict cutoff in this cell-based assay.
Different disease mechanism from the dominant sister syndrome
By analyzing RNA sequencing data, the researchers showed that children with the recessive RNU4-2 condition have markedly reduced levels of RNU4-2 RNA, together with a sharp rise in a closely related RNA, RNU4-1. The ratio of RNU4-2 to RNU4-1 was far lower than in thousands of other patients’ samples and could reliably distinguish affected individuals, suggesting a loss-of-function mechanism partly countered—but not fully rescued—by RNU4-1 upregulation. In contrast, people with dominant ReNU syndrome actually had increased RNU4-2 levels and a characteristic shift in how certain splice sites are used, pointing to a qualitatively different molecular disturbance. Notably, despite the central role of U4 in the major spliceosome, the team did not detect the broad intron-retention pattern seen in related disorders of the minor spliceosome, implying a subtler or tissue-specific defect in RNA processing.
What this means for families and future diagnoses
For non-specialists, the key message is that this study defines a new inherited brain disorder caused by having two faulty copies of a small, noncoding RNA gene. Children with this condition typically show significant developmental delay, frequent seizures, and a characteristic pattern of white matter change on MRI, but they are clinically distinct from patients with the more common, dominantly acting ReNU syndrome. The discovery expands the known spectrum of diseases tied to the spliceosome’s RNA components, underscores the importance of including small RNA genes in genetic testing panels, and points to simple biomarkers—such as the RNU4-2 to RNU4-1 expression ratio and the presence of extreme perivascular space dilation—that can help end the diagnostic odyssey for affected families.
Citation: Rius, R., Blakes, A.J.M., Chen, Y. et al. Biallelic variants in the noncoding RNA gene RNU4-2 cause a recessive neurodevelopmental syndrome with distinct white matter changes. Nat Genet 58, 761–773 (2026). https://doi.org/10.1038/s41588-026-02554-6
Keywords: neurodevelopmental disorder, spliceosome, noncoding RNA, white matter changes, RNU4-2