Clear Sky Science · en
Boundary-guided cell alignment drives mouse epiblast maturation
How Tissues Find Their Shape
Before a mouse embryo takes on any recognizable form, its cells must first decide how to line up and where to carve out the very first internal cavity. This paper explores a deceptively simple question with big implications: how does a seemingly featureless ball of cells self-organize into a well-ordered structure that can later build organs and body axes? By watching early mouse embryos in three dimensions and blending biology with physics, the authors uncover how the edges of a tissue quietly choreograph this transformation.
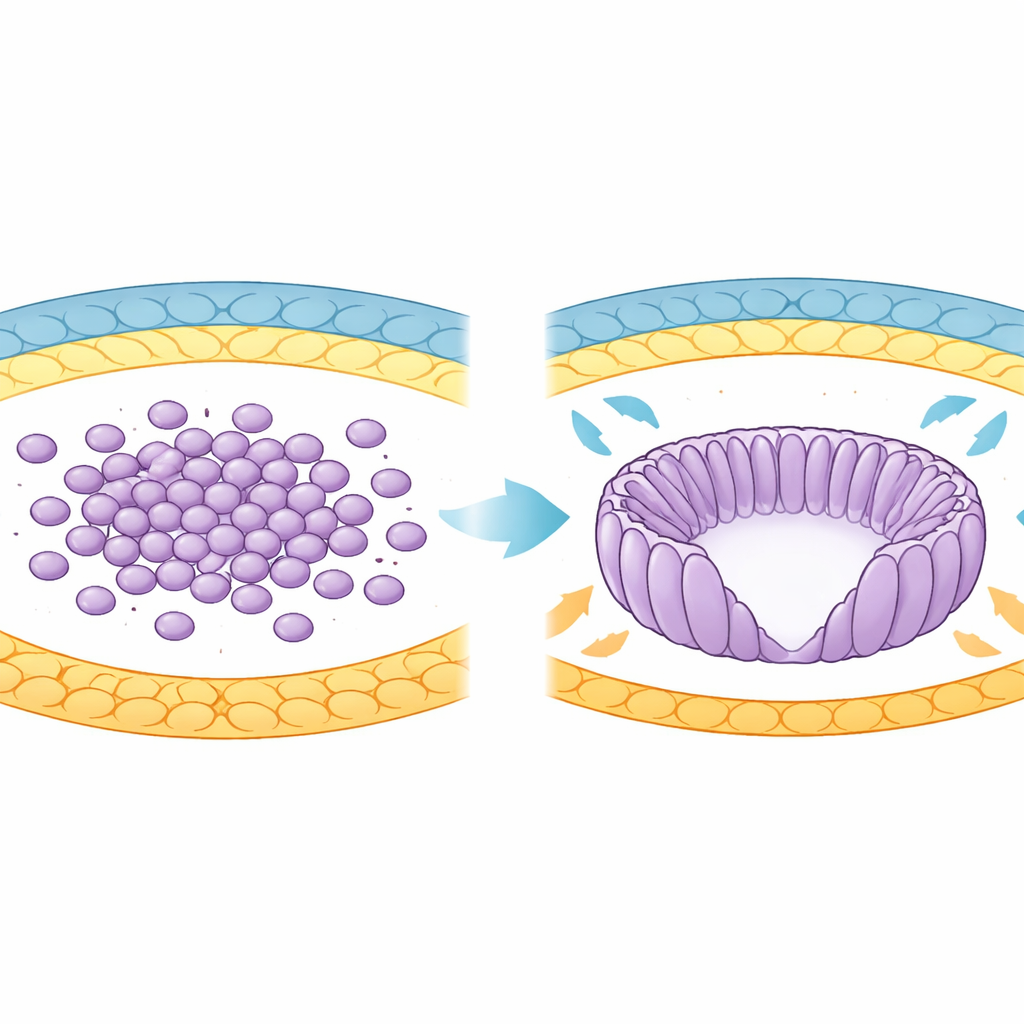
From Cell Cluster to Organized Cup
In early development, the future body of the mouse comes from a group of cells called the epiblast. Initially these cells are round and loosely organized inside the blastocyst. As the embryo implants into the uterus, the epiblast reshapes into a cup-like structure called the egg cylinder. During this transition, cells stretch into elongated forms, line up like spokes in a wheel, and surround a central cavity known as the proamniotic cavity. The authors used advanced 3D imaging and computational analysis to track thousands of cells as this shape change unfolded between specific developmental days, revealing a progressive increase in how similarly neighboring cells were oriented.
The Power of Edges and Borders
The epiblast does not exist in isolation. It is wrapped by two different tissues that form distinct boundaries: one that will become the visceral endoderm below, and another that will become extraembryonic ectoderm above. The study shows that epiblast cells gradually orient perpendicular to the visceral endoderm side but parallel to the extraembryonic side. This means that where the epiblast touches different neighbors, cells adopt different preferred directions. When the researchers mapped cell orientations across entire embryos, they found that alignment was always strongest near these boundaries and weaker in the center. This pattern suggested that the edges of the tissue act as guiding rails, telling cells which way to point and ultimately shaping the whole structure.
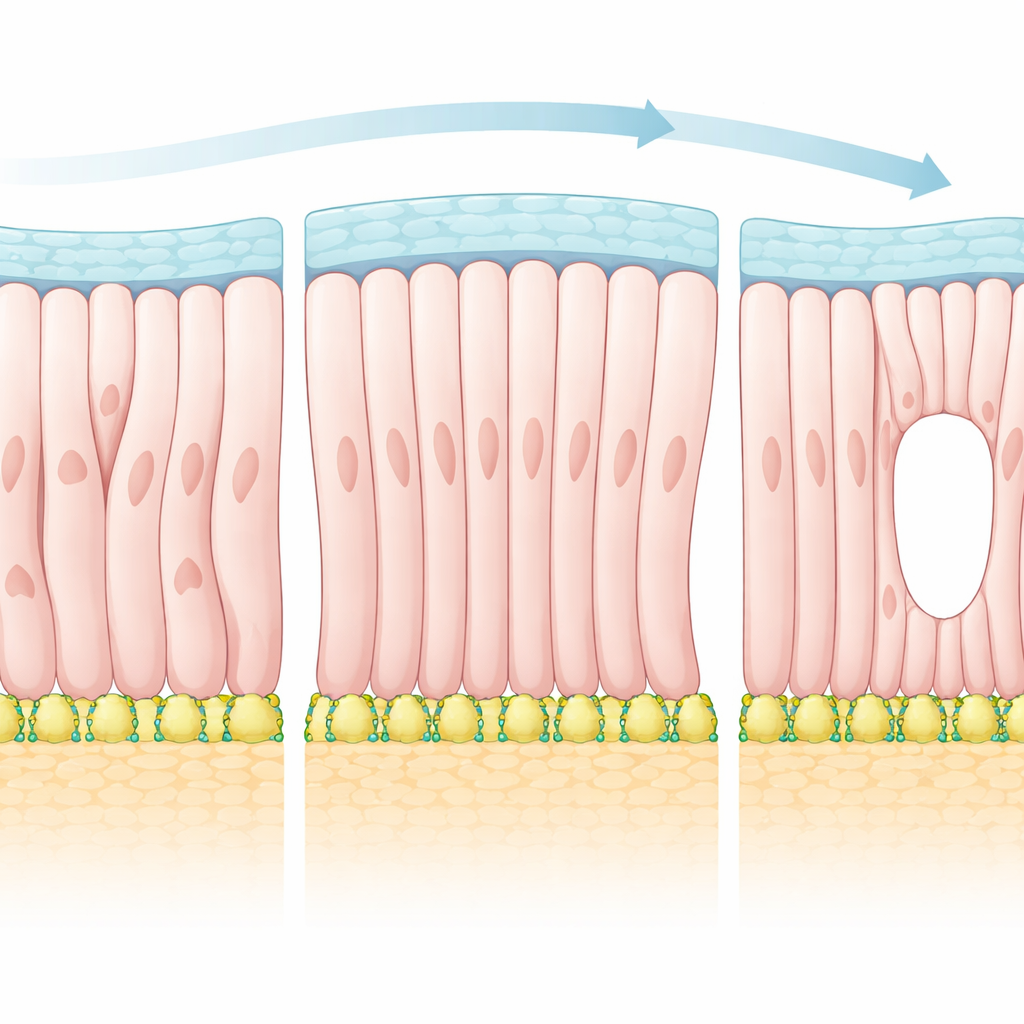
Borrowing Ideas from Liquid Crystals
To explain these observations, the authors turned to a framework more commonly used to describe liquid crystals in displays. In such materials, rod-like molecules tend to line up with each other, but their exact pattern can be strongly influenced by the surfaces that confine them. The team treated epiblast cells as polar “particles” that prefer to align both with their neighbors and with directions imposed by the surrounding boundaries. Using this theory, they predicted that as the influence of the boundaries strengthens, the tissue should undergo a transition into a more ordered state that contains special points of disruption in the alignment field—so-called topological defects. In the embryo, these defects correspond to places where cells’ apical sides converge and where the future cavity first opens.
Molecular Anchors at the Bottom Edge
What, at the molecular level, gives the visceral endoderm boundary its strong influence? The authors combined single-cell gene data and protein staining to show that this lower boundary becomes enriched in specific extracellular matrix components, especially laminin, and in the active form of its cell-surface partner, integrin β1. Together these act like tiny molecular anchors for the basal side of epiblast cells. As laminin–integrin adhesion intensifies over time at this boundary, cell alignment becomes more pronounced and a robust pattern emerges. In computer simulations, weakening this anchoring erased the ordered pattern, hinting that these molecules are essential for the tissue to “feel” and respond to its edge.
When Anchors Fail, Shape and Signals Falter
To test this directly, the researchers examined embryos lacking laminin γ1 or integrin β1. In these mutants, epiblast cells no longer pointed perpendicularly into the visceral endoderm boundary but instead lay tangentially, as they do near the upper boundary. The tissue lost its characteristic radial alignment, rosette-like clusters of apical surfaces failed to form properly, and lumen initiation sites were disrupted. Yet the basic extent of cell elongation still increased, indicating that the main defect was in how cells were oriented rather than whether they could stretch. In a separate manipulation, enzymatically weakening the matrix at the lower boundary reduced activity of ERK, a signaling pathway important for cell growth and differentiation, suggesting that correct tissue patterning helps trigger the molecular programs that drive epiblast maturation.
Why This Matters for Building Bodies
Put simply, the study shows that the way early embryonic cells line up is not random; it is steered by differences at their boundaries, especially by laminin–integrin adhesion at the visceral endoderm side. This boundary-guided alignment is enough to explain where the first cavity appears and how signals like ERK are activated to move the embryo forward in development. For a lay reader, the key message is that edges are not just passive borders: they are active organizers that help a shapeless clump of cells become an ordered, functional tissue, laying down the blueprint for the rest of the body.
Citation: Ichikawa, T., Guruciaga, P.C., Hu, S. et al. Boundary-guided cell alignment drives mouse epiblast maturation. Nat. Phys. 22, 461–473 (2026). https://doi.org/10.1038/s41567-026-03176-9
Keywords: embryonic development, cell alignment, extracellular matrix, tissue patterning, lumen formation