Clear Sky Science · en
Ablation of prostaglandin E2 signalling through dual receptor knockout in CAR T cells enhances therapeutic efficacy in solid tumours
Why this research matters for future cancer treatments
Many people have heard of CAR T cell therapy, a form of personalized immunotherapy that can eradicate certain blood cancers. Yet the same strategy has struggled in solid tumors like pancreatic or colorectal cancer. This study explores a hidden chemical brake inside solid tumors—mediated by a fatty molecule called prostaglandin E2—and shows how carefully editing two of its docking sites on therapeutic T cells can markedly boost their power without disrupting the rest of the body.
A smart upgrade to living cancer drugs
CAR T cells are ordinary immune cells taken from a patient and reprogrammed to recognize a specific marker on cancer cells. In blood cancers, they can expand vigorously, attack, and sometimes cure disease after just one infusion. Solid tumors are different. They are surrounded by a hostile neighborhood, known as the tumor microenvironment, that is filled with substances that exhaust or paralyze T cells. One of the most influential of these is prostaglandin E2, or PGE2, a lipid signal produced in high amounts by many tumors and associated with poor outcomes. Instead of trying to block PGE2 everywhere in the body—a strategy that has caused serious side effects in past drug trials—the authors asked whether they could make CAR T cells simply ignore this signal.
Disarming a key suppressive signal
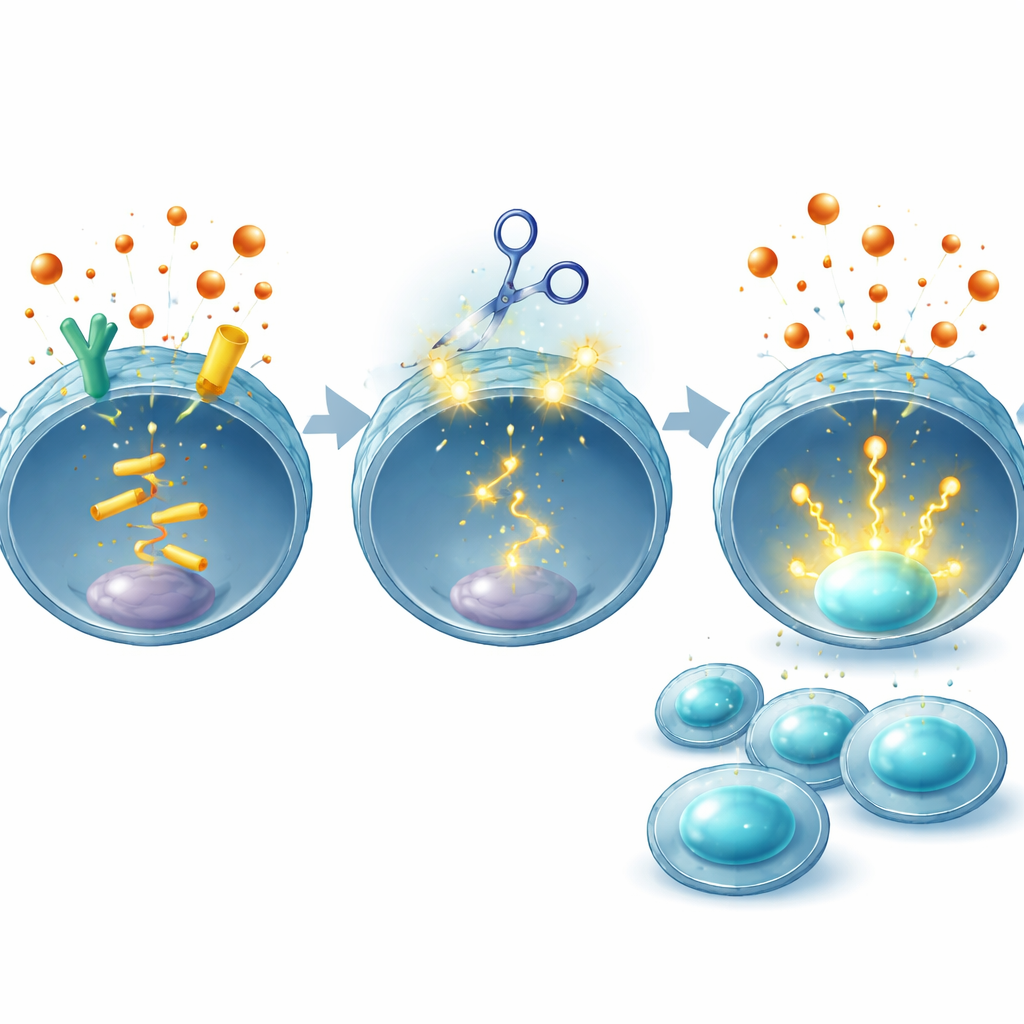
PGE2 works by binding to four receptors on cell surfaces, named EP1 through EP4. Earlier work from this and other groups showed that the EP2 and EP4 receptors on T cells are particularly important for weakening anti-tumor immunity, largely by disrupting the cells’ ability to respond to the growth factor interleukin-2. In this study, the researchers used CRISPR–Cas9 gene editing to remove EP2 and EP4 specifically from T cells that had been engineered as CAR T cells. Because these receptors are difficult to detect with standard laboratory antibodies, the team validated the edits indirectly: they measured downstream chemical messengers, such as the signaling molecule cAMP and the activated form of a protein called CREB, after adding high doses of PGE2. Double-edited cells showed a near-complete loss of PGE2 signaling, confirming that the receptors had effectively been disabled.
From lab dishes to mice and patient tumors
The team first tested their edited CAR T cells in cell cultures. When ordinary CAR T cells were pre-exposed to PGE2, their ability to multiply and clear cancer cells dropped sharply, even though each cell remained capable of killing once activated. In contrast, CAR T cells lacking both EP2 and EP4 kept expanding and fully eliminated tumor cells despite the presence of PGE2. This pattern held true in both mouse and human systems using targets relevant to pancreatic cancer and other solid tumors. The key difference was not that edited cells were more aggressive per cell, but that more of them survived and divided under suppressive conditions.
Stronger and longer-lasting responses in living models
Next, the researchers moved into animal models. In mice bearing PGE2-rich tumors, T cells or CAR T cells without EP2 and EP4 accumulated in tumors in much higher numbers than their unedited counterparts, while numbers in lymph nodes and blood stayed similar. Tumor growth slowed, survival improved, and a subset of mice experienced complete tumor clearance—an outcome rarely seen with conventional CAR T cells in solid cancers. Imaging studies using a bioluminescent reporter confirmed that edited CAR T cells expanded more robustly inside tumors before eventually declining as tumors regrew, directly tying tumor control to T cell persistence. Finally, in three-dimensional cultures made from patient tumor samples of colorectal, pancreatic, and neuroendocrine cancers, dual-edited CAR T cells consistently killed more tumor cells than standard CAR T cells, underscoring the clinical relevance of the approach.
Balancing power and safety
Because any gene-editing strategy raises concerns about unintended DNA changes, the team performed extensive checks. Using computational tools and whole-genome sequencing, they searched for off-target edits in human CAR T cells and found none beyond the intended EP2 and EP4 genes in the donor they examined. They also noted that CRISPR-edited CAR T cells are already being tested in patients for other targets without clear added toxicity so far. Importantly, by changing only the T cells and not blocking PGE2 production throughout the body, this strategy aims to preserve the hormone’s normal roles in tissues such as the gut and kidneys, avoiding side effects that have plagued systemic drugs.
What this could mean for patients
In simple terms, this work shows that many CAR T cells fail in solid tumors not because they cannot recognize their targets, but because the tumor bathes them in a chemical fog that stops them from multiplying. By removing two tiny docking sites for that fog—EP2 and EP4—from the therapeutic cells, scientists can allow them to keep dividing and working even in a hostile environment. The study provides a blueprint for upgrading CAR T cells so they are selectively deaf to one of cancer’s key silencing signals, potentially making cell therapies more effective against some of the deadliest solid tumors while minimizing harm to the rest of the body.
Citation: Dörr, J., Gregor, L., Lacher, S.B. et al. Ablation of prostaglandin E2 signalling through dual receptor knockout in CAR T cells enhances therapeutic efficacy in solid tumours. Nat. Biomed. Eng 10, 697–710 (2026). https://doi.org/10.1038/s41551-025-01610-6
Keywords: CAR T cell therapy, solid tumours, prostaglandin E2, CRISPR gene editing, tumour microenvironment