Clear Sky Science · en
A biallelic MRPL42 variant causes a combined oxidative phosphorylation deficiency syndrome revealed by multi-omics
When the Cell’s Powerhouses Falter
Mitochondria are often called the power plants of our cells, quietly turning fuel into the energy that keeps hearts beating and brains thinking. This article follows the story of a newborn whose severe illness led scientists to uncover a tiny fault in the cell’s protein-making machinery inside mitochondria. By combining several cutting‑edge laboratory techniques, the researchers show how a single inherited change in one gene can destabilize cellular power production and cause a fatal multisystem disease.
A Newborn in Serious Trouble
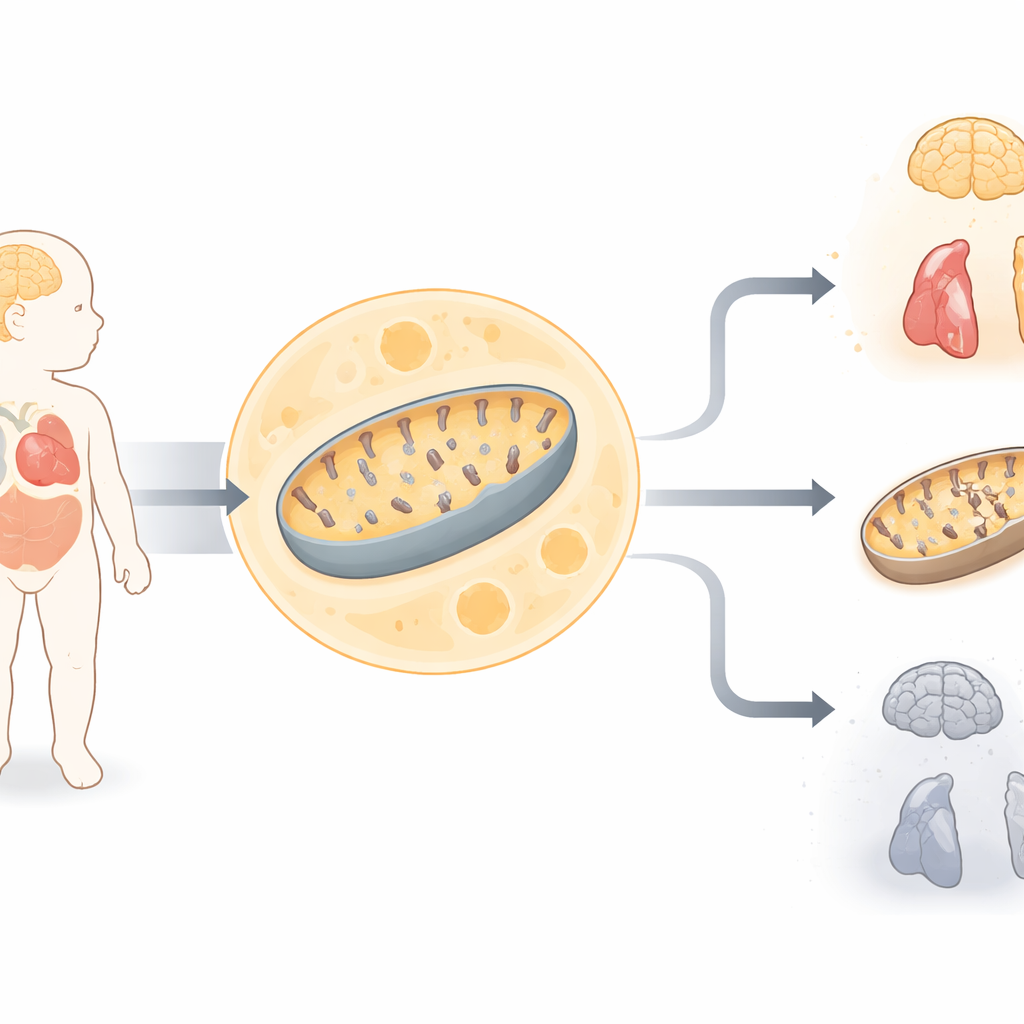
The study centers on a baby girl born to related parents who, at first glance, appeared normal but quickly developed grave medical problems. She had weak muscles, breathing difficulties, seizures, hearing problems, heart changes, and high levels of lactic acid in the blood—a telltale sign that cells are struggling to make energy. Brain scans revealed widespread loss of brain tissue, and tests on skin cells showed that two major energy‑producing enzyme complexes in her mitochondria were underperforming. These clues pointed toward a severe disorder of mitochondrial energy metabolism, but standard genetic tests before birth had failed to find a known cause.
Hunting for a Hidden Gene Error
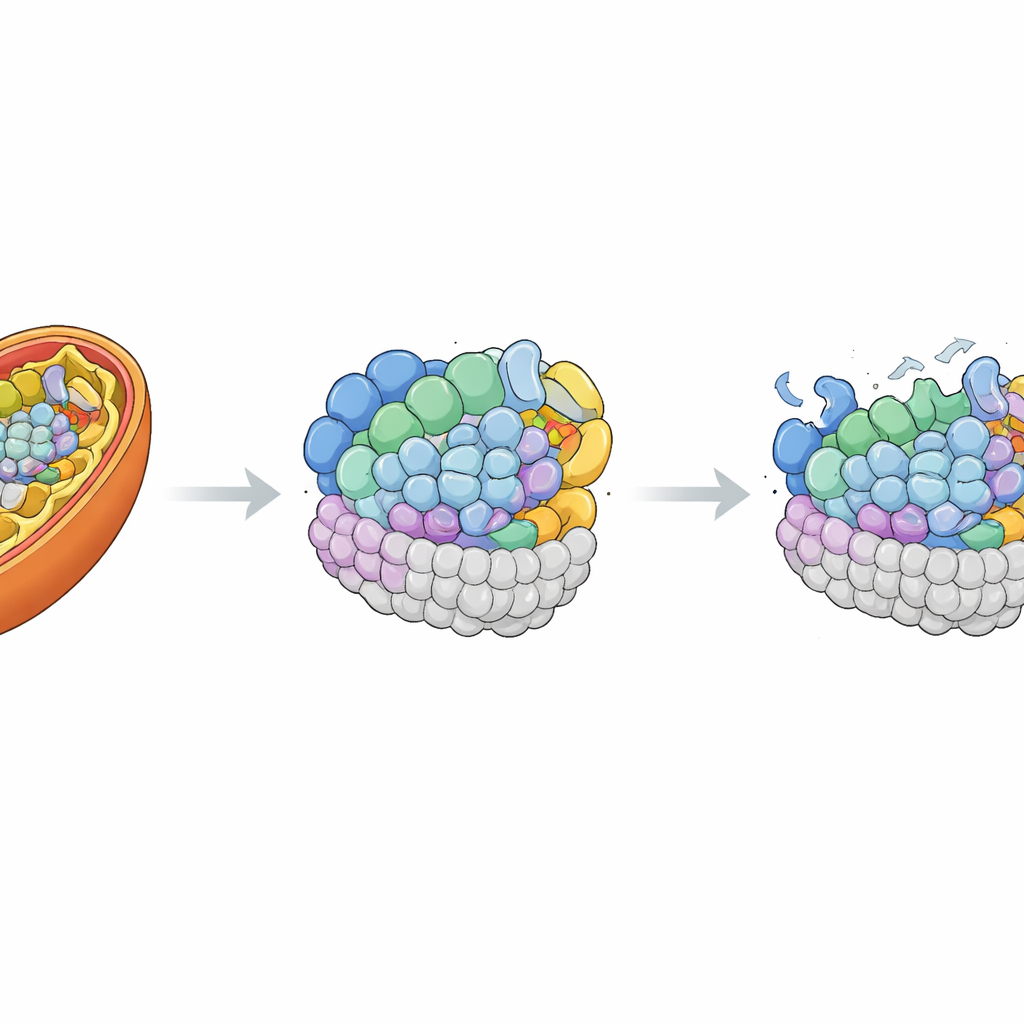
To go beyond routine testing, the team used a “multi‑omics” strategy—reading the DNA, the RNA messages copied from DNA, and the full set of proteins in the child’s cells. Whole‑genome sequencing revealed a rare change in both copies of a gene called MRPL42, which encodes a small piece of the large protein‑making unit (ribosome) inside mitochondria. On its own, this DNA change was classified as uncertain because MRPL42 had not previously been tied to human disease. However, RNA sequencing showed that the altered gene message was mostly misspliced: a critical section was skipped in about four out of five molecules, causing a shift in the reading frame and an early stop signal. Only a small fraction of normal message, and thus some normal protein, was still produced.
How a Missing Piece Weakens the Power Plant
Protein and imaging studies in the patient’s skin cells revealed what this faulty message meant in practice. Levels of MRPL42 protein were reduced to about a quarter of normal, and the mitochondrial network appeared more fragmented than in healthy cells. Detailed protein surveys showed that many building blocks of the mitochondrial ribosome—both its large and small parts—were reduced, suggesting that the entire structure was less stable. In parallel, numerous components of the energy‑producing complexes I and IV, and to a lesser extent complex III, were diminished, while complex II remained unchanged. Measurements of oxygen use confirmed that the cells’ ability to breathe and make ATP, the cell’s energy currency, was markedly impaired.
Putting the Missing Piece Back
To prove that the MRPL42 defect was indeed responsible, the researchers re‑introduced a normal copy of the gene into the patient’s cells using a harmless viral carrier. This boosted MRPL42 levels above those in control cells and partially restored the normal tubular shape of the mitochondrial network. Importantly, many of the lost protein components of the mitochondrial ribosome and of complexes I and IV rebounded toward normal amounts. When the team re‑measured mitochondrial respiration, they found that maximal breathing capacity and spare capacity nearly normalized, and basic energy turnover also improved. These rescue experiments strongly linked the child’s cellular defects to the shortage of functional MRPL42.
What This Means for Patients and Science
Altogether, the study shows that a biallelic (two‑copy) loss‑of‑function change in MRPL42 can cause a severe, often fatal disorder in which multiple mitochondrial energy complexes fail. The gene appears not to be absolutely essential—some protein still forms, allowing development to reach birth—but it is crucial for keeping the mitochondrial ribosome stable enough to support normal energy production. For families, this work provides a molecular explanation for a devastating illness and a route to accurate genetic counseling. More broadly, it highlights how deeply integrated DNA sequencing, RNA analysis, and protein profiling can unmask new disease genes and clarify how tiny faults in the cell’s machinery ripple outward to affect entire organs and, ultimately, the whole body.
Citation: Boschann, F., Kopp, J., Römer, S. et al. A biallelic MRPL42 variant causes a combined oxidative phosphorylation deficiency syndrome revealed by multi-omics. npj Genom. Med. 11, 20 (2026). https://doi.org/10.1038/s41525-026-00564-1
Keywords: mitochondrial disease, oxidative phosphorylation, ribosomal protein, multi-omics, genetic variant