Clear Sky Science · en
Discovery of an Endonuclease G-inhibitory Ku80-peptide protecting against leukemogenic rearrangements at the MLL breakpoint cluster
Protecting Patients from a Hidden Side Effect
Chemotherapy can save lives, but for a small number of patients it has a dangerous late consequence: years after treatment, they develop aggressive blood cancers triggered by damage at a tiny vulnerable stretch of DNA. This study uncovers how a natural DNA-cutting enzyme contributes to that damage and introduces a small designer peptide that shields the risky DNA segment without weakening the cancer‑killing power of chemotherapy.
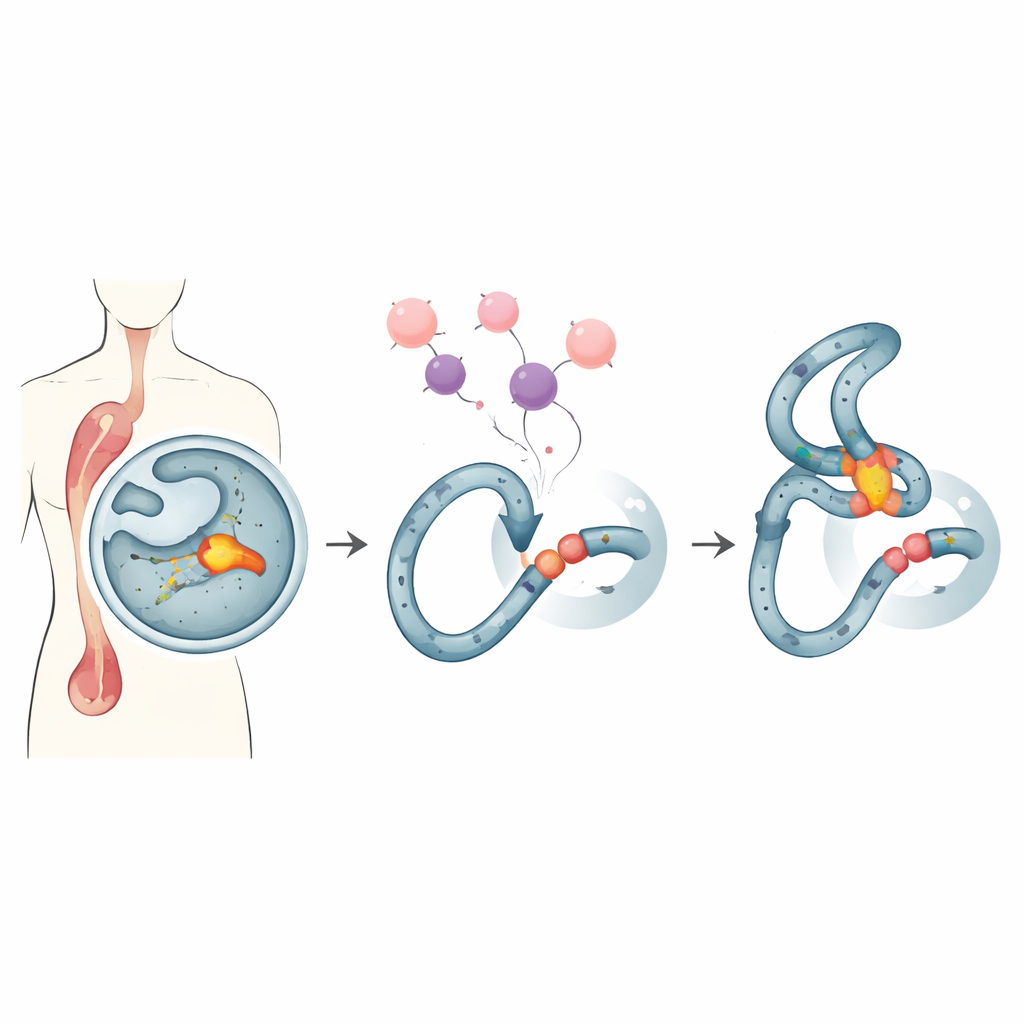
A Fragile Spot in Our Blood‑Making Cells
Some people treated with powerful cancer drugs later develop “therapy‑related” leukemia, a hard‑to‑treat blood cancer. Many of these cases share the same hallmark: breaks and rearrangements in a narrow hotspot of the MLL gene, a region known as the MLL breakpoint cluster. This hotspot sits in a stretch of DNA that tends to form unusual shapes and is especially sensitive to certain drugs. When standard treatments such as the widely used drug doxorubicin stress dividing cells, that fragile region can crack and rejoin incorrectly, creating cancer‑driving DNA fusions.
An Ancient Enzyme Becomes a Risk Factor
The team focuses on Endonuclease G, or EndoG, an evolutionarily ancient enzyme that normally resides in mitochondria but can move to the nucleus. EndoG is known for chopping DNA during cell death, yet at lower, non‑lethal levels it can also nick the genome in ways that foster mutations. Earlier work showed that under replication stress—conditions created by drugs like doxorubicin—EndoG is recruited to the MLL hotspot and helps initiate the breaks that precede leukemic rearrangements. Because of this dual nature, scientists have sought a way to restrain EndoG’s harmful activity at this hotspot without blocking its useful roles in killing cancer cells.
Turning a Repair Protein into a Shield
In flies, EndoG is kept in check by a natural inhibitor protein. No such inhibitor was known in humans, but the researchers noticed that part of a human DNA repair factor called Ku80 structurally resembles the fly inhibitor. Ku80 usually teams up with Ku70 to seal broken DNA ends, yet its loose tail region appeared capable of binding EndoG. Using biochemical tests and structural modeling, the authors showed that EndoG indeed attaches to Ku80’s tail and that a specific stretch of Ku80, called the C‑terminal domain, can physically interact with EndoG. When they expressed just this tail fragment in human cells, it cut the frequency of therapy‑like rearrangements at the MLL hotspot in half, without altering normal DNA repair or cell survival after chemotherapy.
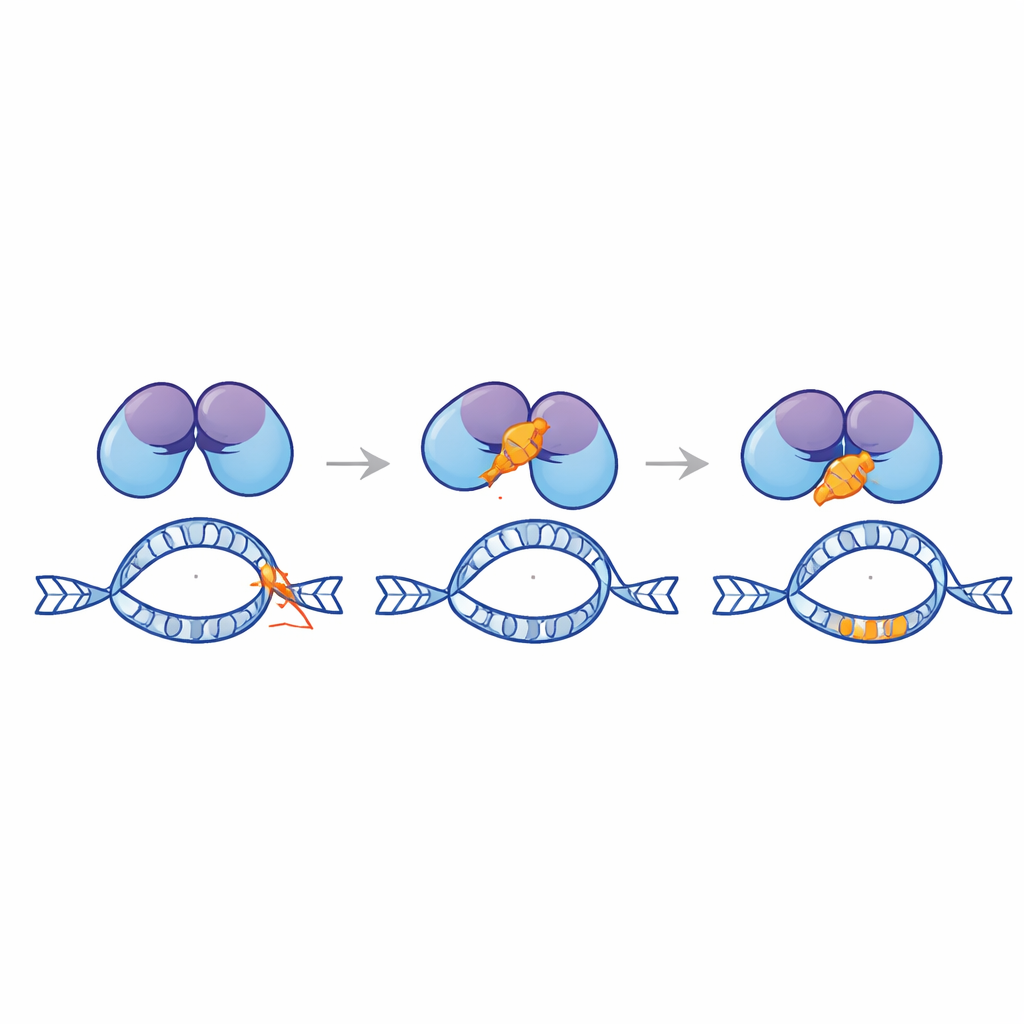
Designing a Protective Peptide
Guided by computer modeling, the team carved Ku80’s tail into thousands of candidate short peptides and screened them for strong predicted binding to EndoG. From this library they singled out a 28‑amino‑acid peptide, dubbed Ku3, that docked tightly onto EndoG in simulations. When Ku3 was delivered into leukemia and cervical cancer cell lines carrying an engineered MLL reporter, it reduced doxorubicin‑induced rearrangements and protected the integrity of the hotspot, again without reducing the drug’s ability to kill cells. Microscopy‑based proximity assays showed that Ku3 disrupts the formation of complexes between EndoG and Ku80 in the nucleus. Single‑molecule tracking further revealed that Ku3 changes how EndoG interacts with chromatin, consistent with a direct physical brake on EndoG at vulnerable DNA sites.
From Mechanism to Future Therapy
This work proposes a precise way to blunt a dangerous side effect of chemotherapy: a tailor‑made peptide derived from a natural repair protein locks onto EndoG, prevents it from attacking a fragile DNA hotspot, and thereby lowers the chance of leukemogenic rearrangements, all while leaving the cancer‑killing damage intact. In the long term, such peptides—or improved versions based on the same motif—could be co‑administered with standard genotoxic drugs to protect patients’ blood‑forming cells and reduce the risk of secondary leukemias that currently shadow otherwise successful cancer treatment.
Citation: Eberle, J., Salem, A., Hofmann, M. et al. Discovery of an Endonuclease G-inhibitory Ku80-peptide protecting against leukemogenic rearrangements at the MLL breakpoint cluster. Nat Commun 17, 3562 (2026). https://doi.org/10.1038/s41467-026-72034-2
Keywords: therapy-related leukemia, DNA break hotspots, endonuclease G, Ku80 peptide inhibitor, chemotherapy side effects