Clear Sky Science · en
Extracellular K+ modulates the pore conformations of Cys-loop receptor anion channels
How Cells Listen to Potassium Outside
Every thought, movement, and heartbeat in our bodies depends on tiny charged atoms, or ions, moving across cell membranes. Potassium is one of the most important of these ions, and its levels outside nerve cells are kept within a very tight range. This study reveals an unexpected way that brain cells can directly sense changes in potassium outside the cell and translate them into changes in the flow of chloride, another key ion. The work uncovers a hidden sensing mechanism in a family of nerve receptors and suggests new ways the brain might monitor and respond to its own chemical environment.
A New Kind of Potassium Sensor
Until now, most known sensors of potassium in animals were channels and pumps that use potassium as fuel or cargo, not as a signal in its own right. The authors focused on a little-studied protein in the fruit fly Drosophila, called DmAlka, which belongs to the Cys-loop receptor family. Members of this family typically form channels that open when they bind neurotransmitters and then let chloride ions cross the membrane to dampen electrical activity. Surprisingly, earlier work showed that DmAlka does not respond to the usual neurotransmitter glycine but is activated by alkaline (basic) conditions. Here, the researchers show that DmAlka is also finely tuned to the normal range of potassium concentrations outside cells, and that this tuning strongly shapes how chloride flows through the channel.
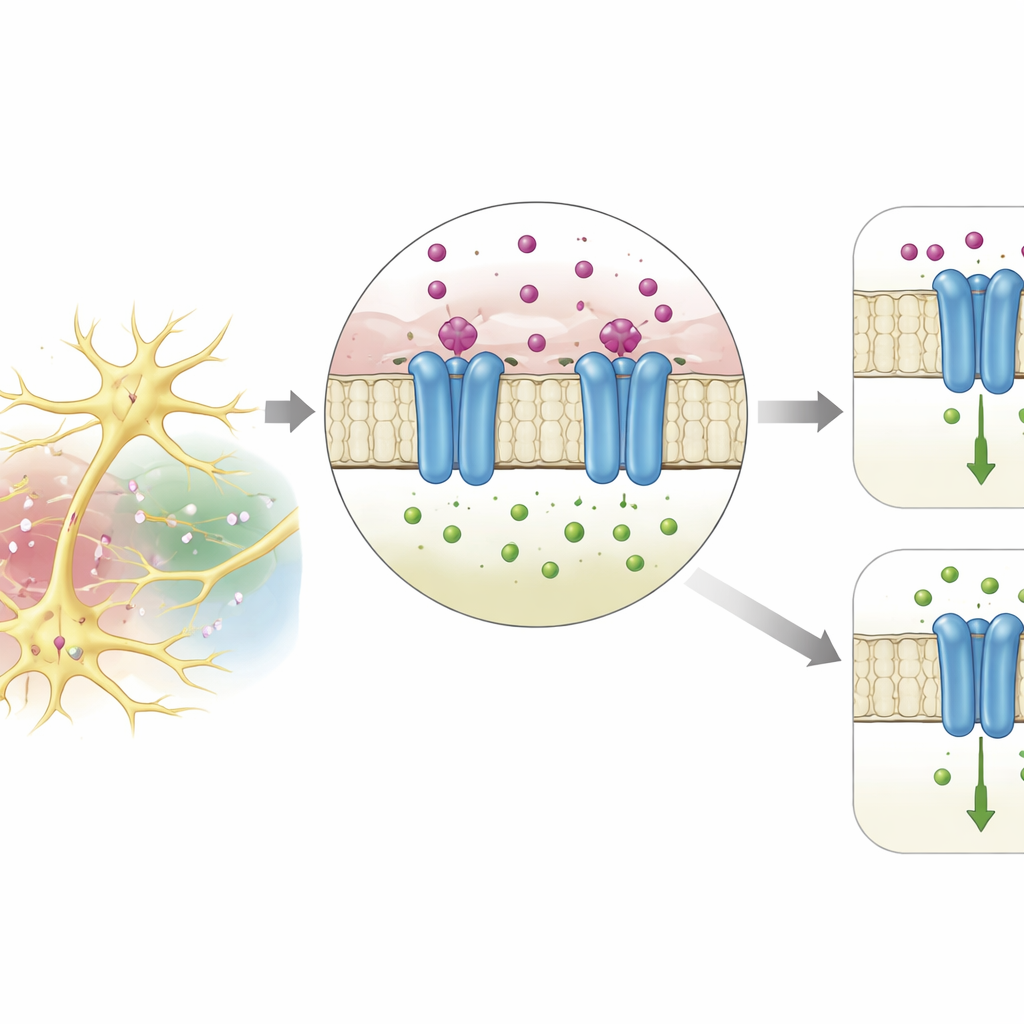
Where Potassium Grabs Hold
Using a powerful structure-prediction program, the team modeled the three-dimensional shape of DmAlka. The model revealed a small pocket at the junction between the outer, signal-sensing part of the protein and the membrane-spanning pore that carries ions. In this pocket, a potassium ion is cradled by four oxygen atoms from nearby amino acids, in an arrangement that closely mimics the way potassium is held in classic potassium channels or by water molecules in solution. By altering individual building blocks that help form this pocket, the researchers could weaken or abolish the channel’s sensitivity to potassium, confirming that this site is the key docking spot. Similar features were found in related proteins across many arthropod species, suggesting that this potassium recognition strategy is widely conserved in invertebrates.
Two Modes, Switched by Potassium
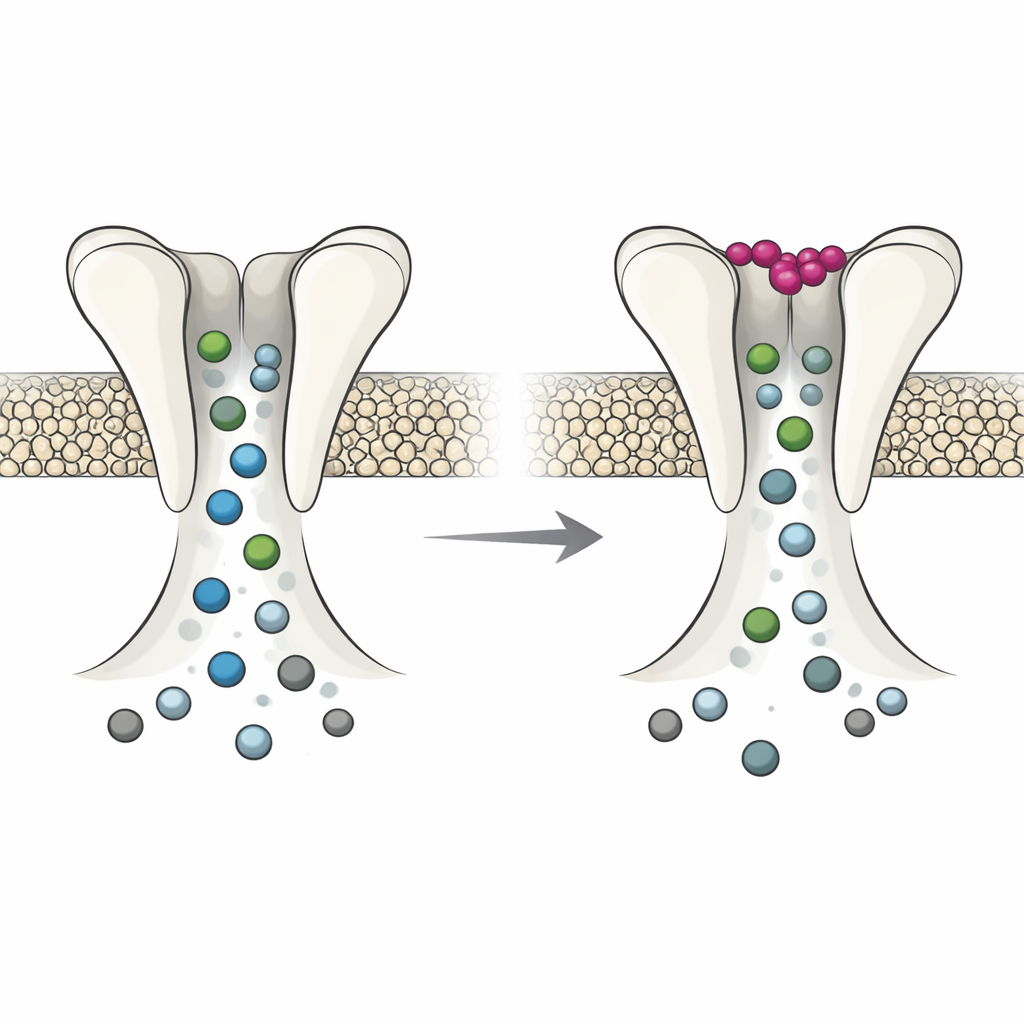
Electrophysiology experiments in frog eggs, which can be used as miniature factories to produce channels, revealed that DmAlka behaves like a switch with two modes. When potassium outside the cell is low, the channel is more likely to stay open, responds differently to alkaline conditions, and desensitizes less over time. In this mode, the pore is relatively loose: it favors chloride but also allows other negative ions, such as bicarbonate, to pass more freely, and it becomes less vulnerable to blockage by certain drugs. When potassium binds at its special site, the whole protein subtly shifts. The pore narrows and becomes more selective for chloride, conducts other anions less well, and shows a stronger response to pore-blocking molecules. In effect, extracellular potassium toggles the channel between a flexible, broadly permeable state and a tighter, more chloride-focused state.
Echoes in the Human Brain
Intriguingly, the same general mechanism appears to exist, at least in latent form, in humans. A human glycine receptor subtype, GlyR α2, normally responds to the neurotransmitter glycine and not to potassium. By transplanting key features of the DmAlka potassium pocket into the human receptor, the authors created a mutant that gained potassium sensitivity: high external potassium boosted baseline currents even without glycine present. They also showed that naturally occurring human variants of GlyR α2, some associated with epilepsy-related changes in the brain, can acquire potassium responsiveness. In these human receptors, as in DmAlka, potassium binding shifts pore properties, changing how easily chloride and bicarbonate pass and how well certain blockers work.
Why This Matters for Brain Health
The fruit fly version of this channel is enriched in glial cells, the support cells that help maintain potassium balance and pH around neurons. The newly uncovered mechanism suggests a simple logic: when extracellular potassium drops, DmAlka opens in a way that lets more chloride enter glia and more bicarbonate leave, potentially helping restore both potassium levels and acidity in the surrounding space. In humans, similar potassium-tuned behavior in glycine receptors could become important under extreme conditions, such as stroke or epileptic seizures, when potassium outside neurons can soar far above normal. Overall, this work reveals that some Cys-loop receptors are not just passive responders to neurotransmitters; they can also act as direct sensors of the ionic environment, linking changes in extracellular potassium to shifts in chloride signaling across species.
Citation: Shimomura, T., Kubo, Y., Saitoe, M. et al. Extracellular K+ modulates the pore conformations of Cys-loop receptor anion channels. Nat Commun 17, 3453 (2026). https://doi.org/10.1038/s41467-026-71629-z
Keywords: extracellular potassium sensing, chloride channels, Cys-loop receptors, glycine receptor variants, glial ion homeostasis