Clear Sky Science · en
Generation of marmoset monkeys with a non-mosaic disruption of the OTOF gene as a model of human deafness
Why Tiny Monkeys Matter for Hearing Loss
Hearing loss caused by faulty genes is common, and for one important gene called OTOF, gene therapy in children is already beginning. Yet scientists still lack an animal model that closely mirrors the human ear to test how safe, precise, and long-lasting such treatments really are. This study creates just that: a line of small marmoset monkeys engineered to have the same type of deafness seen in many children, offering a powerful bridge between mouse experiments and human clinical trials. 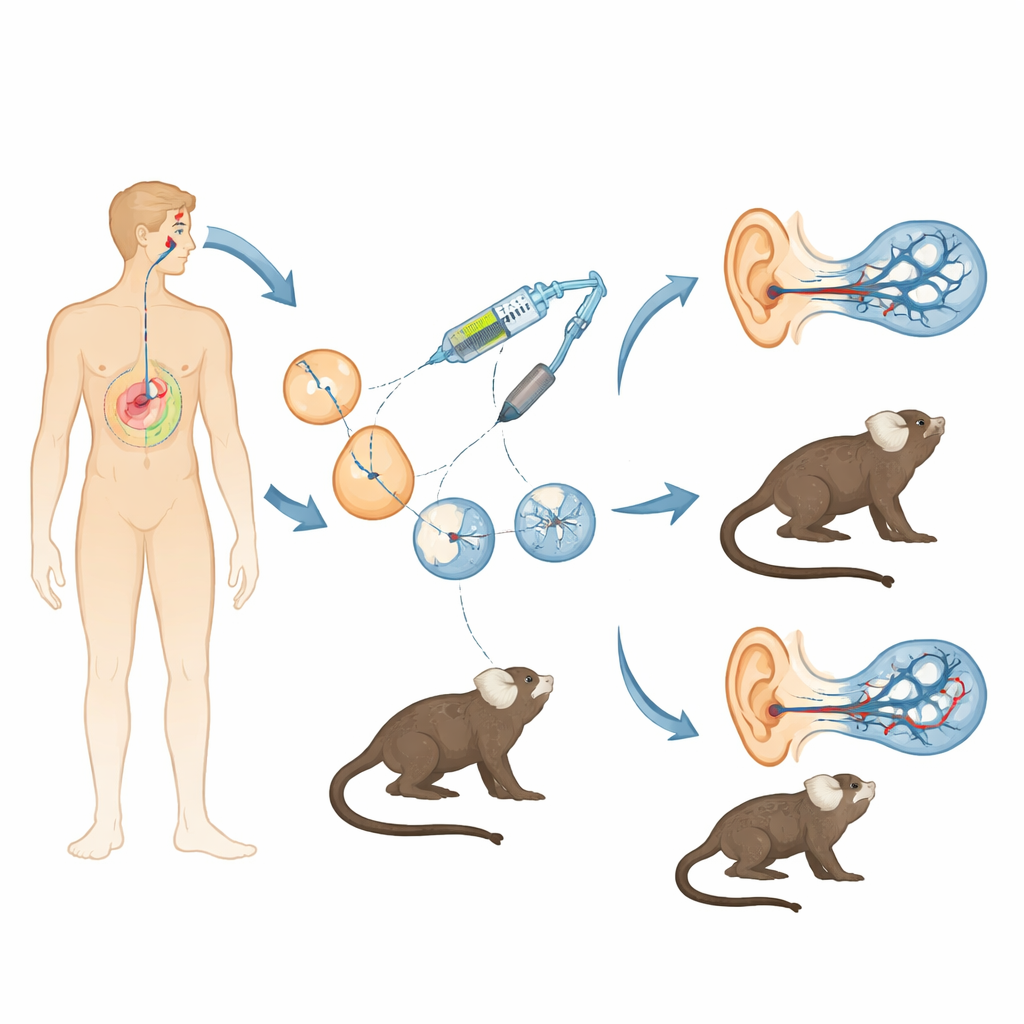
A Silent Synapse at the Heart of Deafness
Many people with OTOF-related deafness have ears that look normal and even outer ear cells that still respond to sound. The problem lies deeper, at the microscopic junctions where inner hair cells in the cochlea pass sound information to the hearing nerve. The OTOF gene provides the blueprint for a protein called otoferlin, which is crucial for releasing tiny packets of chemical signals at these synapses. When otoferlin is missing, the sound signal simply does not get relayed, a condition known as auditory synaptopathy. Because the rest of the ear can remain structurally healthy for years, this form of deafness is especially promising for one-time gene therapies that aim to restore missing otoferlin.
Engineering a Primate Model of Human Deafness
Mice have been invaluable for understanding hearing, but their genes, development, and hearing onset differ in key ways from ours. Marmoset monkeys, by contrast, are small non-human primates that share more of our DNA, start hearing before birth, and have rich social vocal communication. The researchers combined in vitro fertilization with the CRISPR/Cas9 gene-editing system to disrupt a crucial exon in the marmoset OTOF gene. By injecting carefully optimized mixtures of CRISPR components into early embryos, they sought to delete this segment in both gene copies while avoiding a major pitfall of primate editing: mosaicism, where different cells in the same animal carry different mutations. After many rounds of oocyte collection, fertilization, and embryo transfer into surrogate mothers, they obtained two surviving monkeys with biallelic, non-mosaic OTOF disruptions—meaning every cell carried the intended knockout.
Checking Hearing, Brain Signals, and Behavior
To find out whether these engineered monkeys truly modeled the human condition, the team tested their hearing starting at six months of age. Electrical recordings from the brainstem, which normally show clear waves when sound reaches the auditory pathway, were flat even at very loud sound levels in the knockout animals. At the same time, tests of otoacoustic emissions—tiny echoes generated by outer hair cells—were robust and similar to those of normal monkeys. This combination of absent brain responses but preserved cochlear mechanics closely matches human OTOF-related auditory synaptopathy. Microscopic examination of one animal’s inner ears confirmed that inner hair cells were present but lacked otoferlin protein, while overall cochlear structure remained intact. 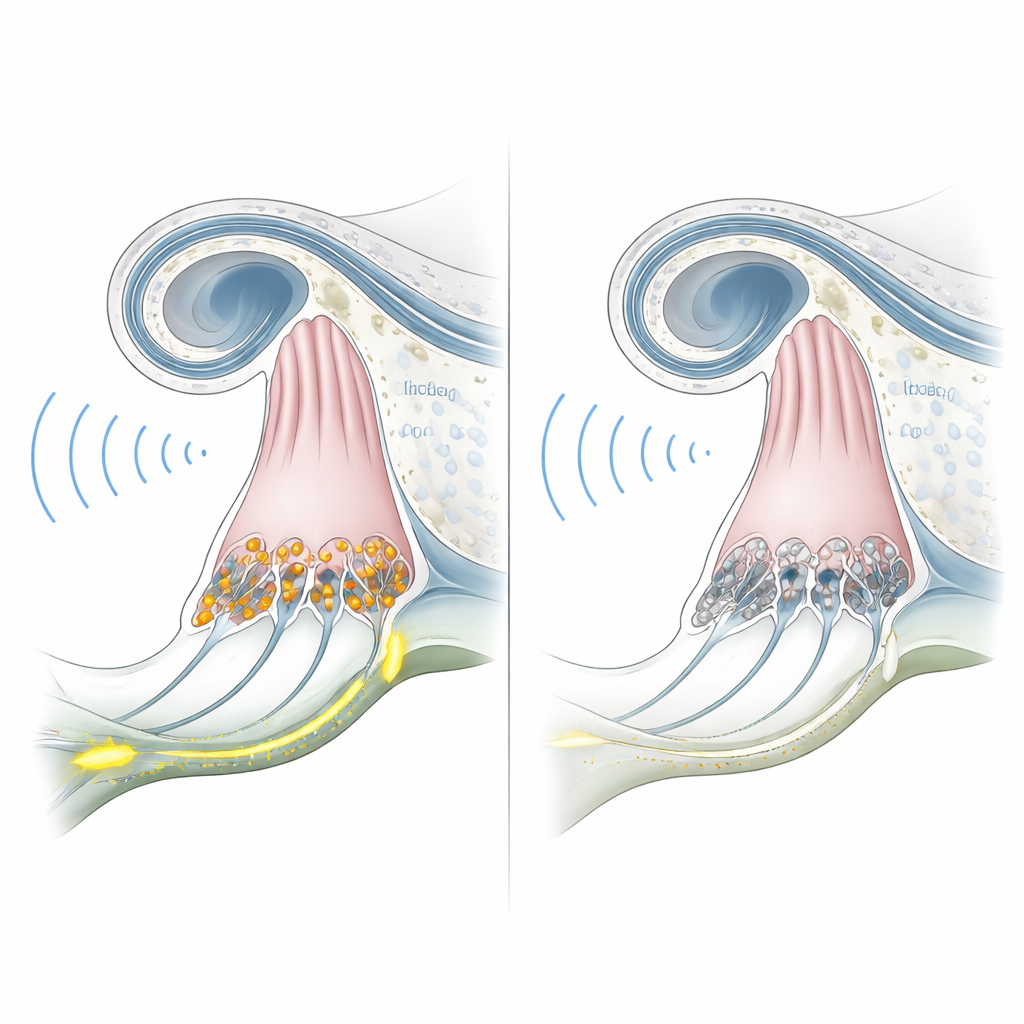
Voices Without Hearing
Because marmosets are famously talkative, the scientists also asked how deafness affected early vocal development. When infants were briefly recorded away from their families, both normal and knockout monkeys produced thousands of calls, including all major call types that healthy infants usually make. Calling rates were similar across groups, suggesting that the basic drive and repertoire for vocalizations are largely built-in and do not require hearing, at least early in life. However, more detailed analysis revealed that the longest call types did not lengthen with age as much in deaf animals as in hearing ones, hinting that auditory feedback still shapes finer aspects of vocal maturation over time. These subtle differences make the model valuable not only for hearing research but also for understanding how sound experience sculpts communication.
New Paths Toward Better Hearing Treatments
By producing marmosets with a clean, non-mosaic knockout of the OTOF gene, this work delivers a primate model that closely tracks a human form of inherited deafness. The animals show the hallmark features of auditory synaptopathy: normal outer ear mechanics, missing otoferlin in inner hair cells, and a failure to send sound information to the brain. This model will allow researchers to fine-tune gene therapy strategies—testing different viral delivery systems, doses, and timing, and linking long-term hearing outcomes to precise measurements of otoferlin restoration in the inner ear. Although creating such primate models is slow, costly, and ethically demanding, the authors argue that the potential to improve life-changing therapies for children with genetic deafness makes this carefully controlled approach both scientifically and medically worthwhile.
Citation: Kahland, T., Lindenwald, D.L., Jeschke, M. et al. Generation of marmoset monkeys with a non-mosaic disruption of the OTOF gene as a model of human deafness. Nat Commun 17, 3033 (2026). https://doi.org/10.1038/s41467-026-71047-1
Keywords: genetic deafness, marmoset model, gene therapy, auditory synaptopathy, CRISPR editing