Clear Sky Science · en
Phage homing endonuclease amplifies anti-defense genes to evade bacterial immunity
How viruses outsmart bacterial defenses
Bacteria and the viruses that infect them, called phages, are locked in a microscopic arms race. Bacteria evolve new ways to detect and block invading phages, while phages evolve counter-moves to slip past these defenses. This study uncovers a surprising viral trick: some phages can rapidly copy and amplify specific chunks of their own DNA that help them beat different bacterial immune systems, effectively turning up the volume on their anti-defense tools just when they need them most.
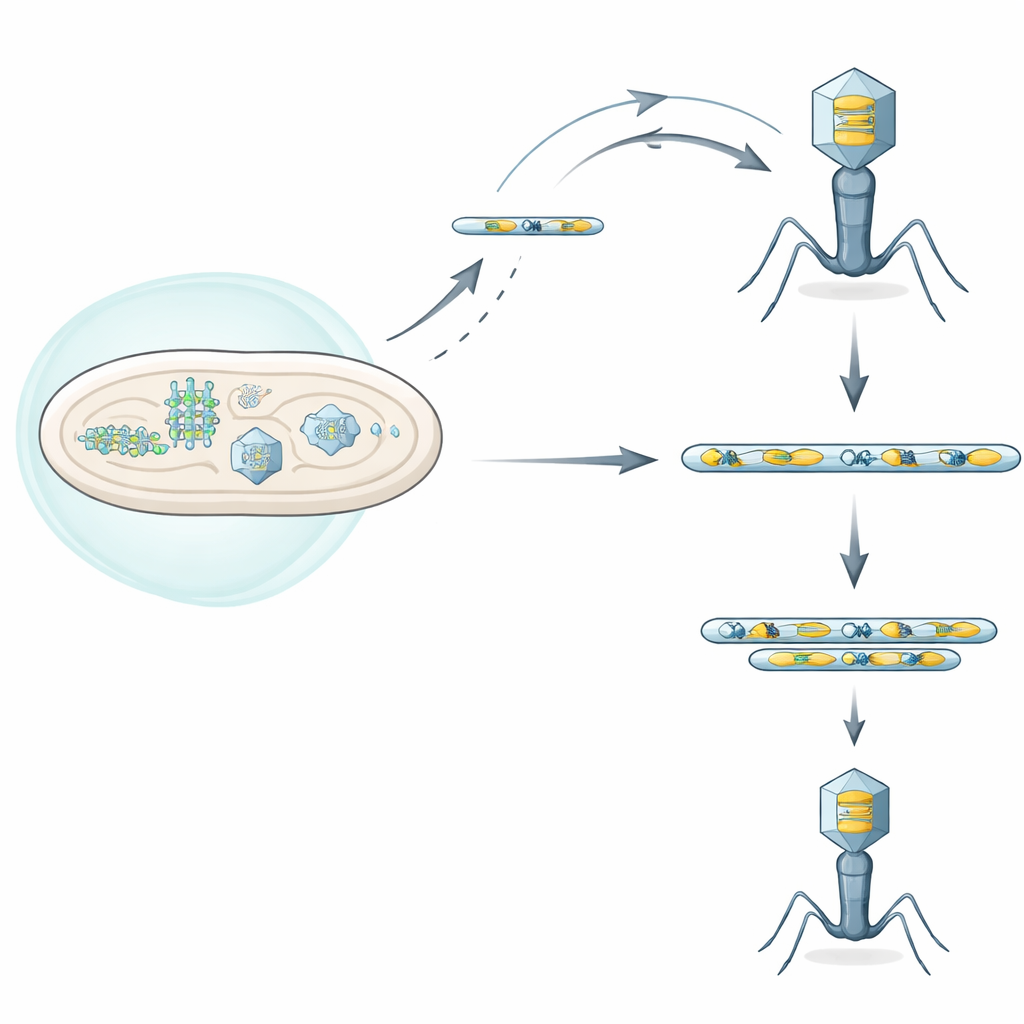
A duel between bacterial shields and viral attackers
Bacteria carry many defense systems that sense viral infection and shut it down, sometimes even killing the infected cell to protect the wider population. The authors focused on one such system, called Septu, found in a clinical strain of Escherichia coli. Septu attacks a small but vital molecule, a transfer RNA (tRNA) needed to translate genetic code into proteins. By cutting this tRNATyr, Septu starves both the cell and the invading phage of a key ingredient for protein synthesis, halting viral replication. Yet some phages still manage to grow despite Septu, hinting that they have evolved clever escape routes.
Extra tRNA copies as viral backup supply
Working with a T-even phage known as T6, the researchers isolated rare “escape” mutants that could replicate in the presence of Septu. When they sequenced these viral genomes, they did not find a single, neat mutation shared by all escapees. Instead, they discovered that each escape phage had massively amplified a short DNA segment containing two features: the full tRNATyr gene and a nearby gene called segB, which encodes a homing endonuclease, a DNA-cutting protein. These amplified segments formed tandem repeats, like the same paragraph pasted many times in a row. The extra copies boosted tRNATyr production, providing enough tRNA to overcome Septu’s cutting activity and allowing viral replication to resume.
A DNA-cutting enzyme that drives genome expansion
The team then asked whether SegB was merely a passenger in this amplified region or the driver of the process. By evolving phages lacking SegB, or carrying a disabled version of the protein, they showed that only phages with a working SegB could build these DNA repeats under immune pressure. Biochemical tests confirmed that SegB cuts specific spots in the phage genome, often near tRNA genes. After SegB introduces breaks, the viral recombination machinery can rejoin DNA in ways that accidentally duplicate nearby segments. When Septu is present, variants that happen to gain more tRNATyr copies have a strong survival advantage and quickly take over the phage population. Once the defense pressure is removed, these extra copies tend to collapse back down, showing that the amplifications are dynamic and reversible.
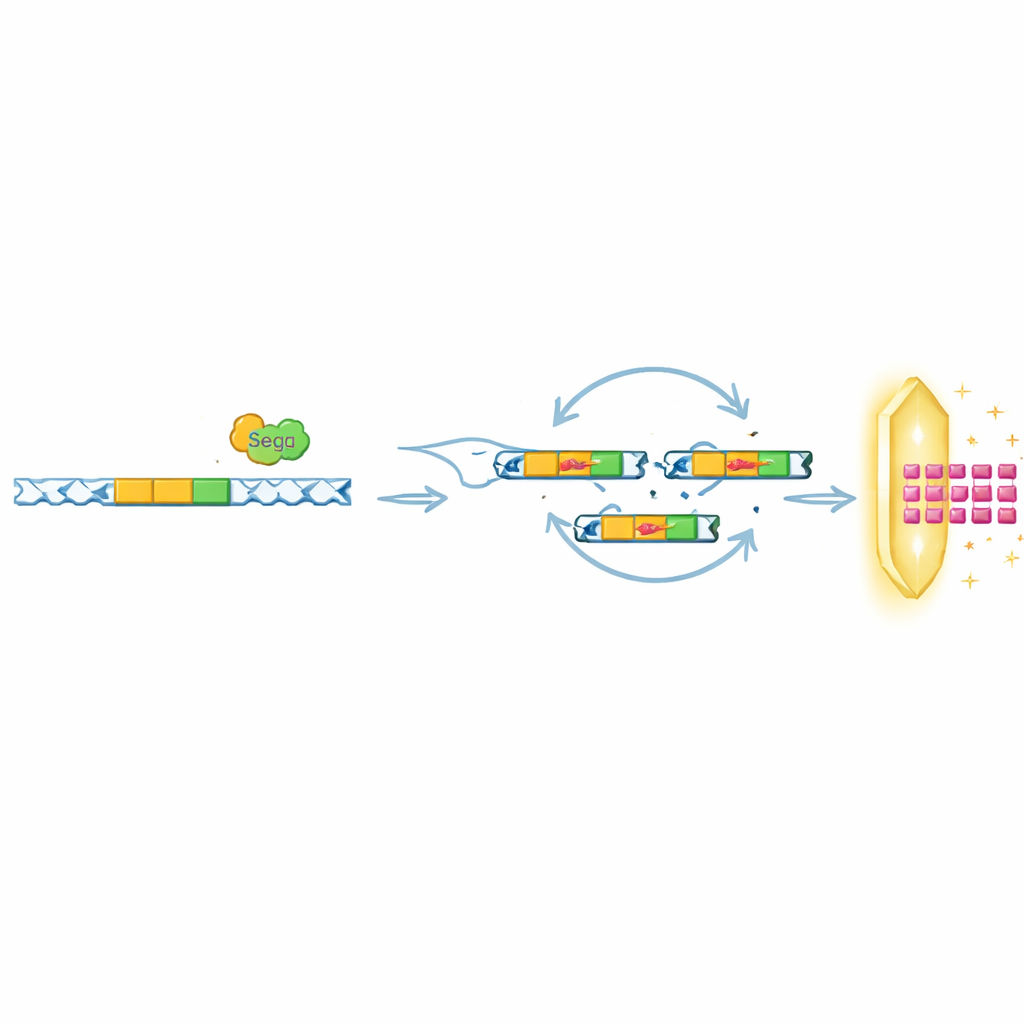
Recycling the same trick against different defenses
Intriguingly, the researchers found that SegB’s role goes beyond helping T6 outwit Septu. In other experiments, they challenged phages with different bacterial systems, including OLD and ToxIN, which also block infection but in very different ways. Under OLD pressure, SegB drove amplification of a distant DNA region that encodes a small viral protein, Gp49.2, which physically binds and inhibits the OLD defense protein. Under ToxIN pressure in a related phage, SegB was required for amplifying yet another anti-defense gene, tifA. Across many phage genomes in public databases, genes like SegB were frequently found close to tRNA clusters, suggesting that this strategy of coupling DNA cutting with local or long-range gene amplification may be widespread in nature.
What this means for the virus–bacteria arms race
To a non-specialist, the key message is that some viruses don’t just wait for slow, single-letter mutations in their DNA to adapt. Instead, they can rapidly “stretch” parts of their genome like an accordion, making multiple extra copies of helpful genes when under attack and shrinking back when those copies are no longer needed. In this case, a DNA-cutting enzyme called SegB sparks genome rearrangements that amplify tRNA genes and other anti-defense factors, letting phages overpower several different bacterial immune systems. This reveals a flexible, fast-response strategy that helps viruses stay one step ahead in their battle with bacteria.
Citation: Chihara, K., Azam, A.H., Egorov, A.A. et al. Phage homing endonuclease amplifies anti-defense genes to evade bacterial immunity. Nat Commun 17, 3468 (2026). https://doi.org/10.1038/s41467-026-71036-4
Keywords: bacteriophage immunity, bacterial antiviral defense, genome amplification, homing endonuclease, phage–bacteria arms race