Clear Sky Science · en
A bridge-like lipid transfer protein is critical for generation of invasive stages in malaria parasites
How Malaria Parasites Build Their Invasion Gear
Malaria parasites survive by endlessly invading new red blood cells. To do this, each parasite growing inside a cell must build dozens of tiny offspring that are ready to burst out and attack fresh cells. This construction project requires huge amounts of new membrane, made mostly from fatty molecules called lipids. The study summarized here uncovers a key "bridge" protein that ferries lipids inside the parasite, allowing it to assemble the invasion machinery. Understanding this hidden supply line could open new ways to block malaria.
The Hidden Architecture Inside an Infected Cell
Once inside a red blood cell, the malaria parasite multiplies rapidly. Late in its cycle, it turns into a large cell packed with many nuclei. Around each future daughter cell, the parasite builds a supportive shell called the inner membrane complex, or IMC. This shell sits just beneath the outer surface and is essential both for shaping the offspring and for powering their entry into the next red blood cell. Building the IMC from scratch demands a steady stream of lipids from the parasite’s internal factory, the endoplasmic reticulum (ER). The big question the researchers asked was how these lipids move so quickly and efficiently from the ER to the growing IMC.
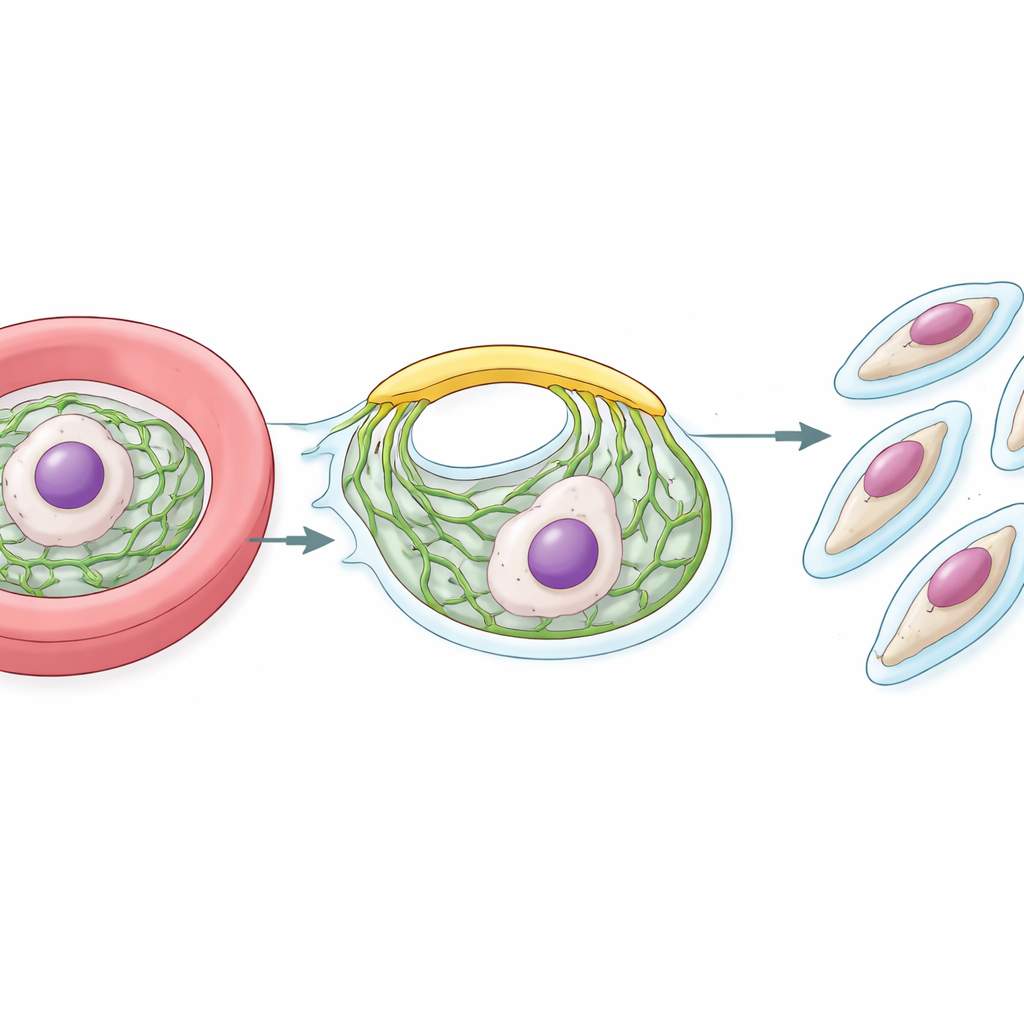
Finding the Lipid Bridge
In many organisms, lipids can move directly between neighboring membranes at close contact sites, helped by special carrier proteins. The team started from a known ER anchor protein called VAP, which grabs partners that contain short "FFAT" motifs. Using a proximity-labeling method, they cataloged proteins that sit near VAP on the ER surface in malaria parasites. Among many candidates, they identified several lipid-handling proteins and focused on a giant one that resembled VPS13, a family of "bridge-like" lipid transporters known from yeast and human cells. They named this parasite protein PfVPS13L1.
How the Bridge Connects Two Membranes
Computer-based structural predictions revealed that PfVPS13L1 forms a long, rod-shaped molecule with an internal groove that can hold many lipid molecules at once. One end of the rod carries an FFAT motif that binds to VAP at the ER, fixing that end to the lipid source. The other end contains adapter regions that latch onto the IMC. The researchers showed that this tip interacts with an IMC protein they termed PfAegerolysin, indicating that PfVPS13L1 physically bridges the ER and the growing IMC. High-resolution microscopy confirmed that PfVPS13L1 clusters precisely where new IMC membrane is forming around future daughter cells.
What Happens When the Bridge Breaks
To test what the bridge does, the scientists used a rapid "mislocalization" trick: they chemically dragged PfVPS13L1 away from its normal position to the parasite’s outer surface, effectively disabling it. When this was done just before the stage where the IMC must grow, parasites could no longer produce normal offspring. The IMC began to form but then stalled; instead of wrapping fully around each nucleus, it remained small and incomplete. As a result, many tiny, shell-wrapped bodies lacked nuclei, while extra genetic material and cytoplasm were left behind in a large residual blob. Other structures, such as specialized secretory organelles, still formed, underscoring that the main failure lay in supplying membrane for the IMC.
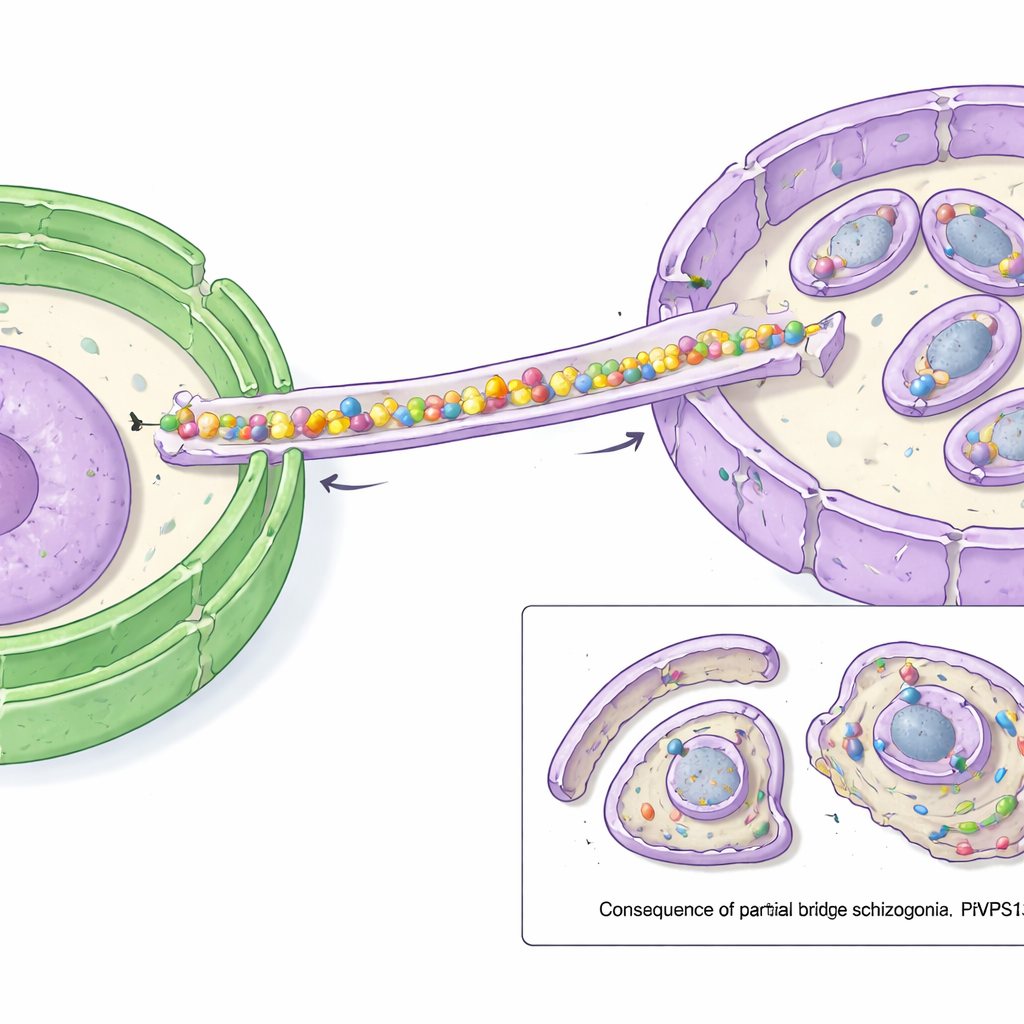
Why This Matters for Fighting Malaria
The study shows that PfVPS13L1 is a crucial lipid bridge that fuels the rapid expansion of the IMC, allowing malaria parasites to generate fully formed, invasive progeny. Without this bulk lipid transfer pathway from the ER, the parasites assemble defective offspring that cannot continue the infection cycle. Because similar bridge-like proteins operate in many types of cells, PfVPS13L1 represents a parasite-specific adaptation of a conserved mechanism. Targeting this bridge, or its partners that secure it to the IMC, could offer a new strategy for antimalarial drugs aimed at quietly starving the parasite’s invasion machinery of the membranes it needs.
Citation: Guillén-Samander, A., Perepelkina, N., Horáčková, V. et al. A bridge-like lipid transfer protein is critical for generation of invasive stages in malaria parasites. Nat Commun 17, 3030 (2026). https://doi.org/10.1038/s41467-026-70887-1
Keywords: malaria, lipid transfer, membrane biogenesis, Plasmodium falciparum, inner membrane complex