Clear Sky Science · en
Enhancing the performance of Magnets photosensors
Shaping Cells with Beams of Light
Imagine being able to turn genes on and off with a beam of light as easily as flipping a switch. This study shows how scientists have upgraded a popular family of light-sensing protein "switches," called Magnets photosensors, so they respond more precisely and powerfully to blue light. These improved switches could help researchers control cell behavior with less light, less damage, and more flexibility in bacteria and even mammalian cells.
Why Light Is a Powerful Control Knob
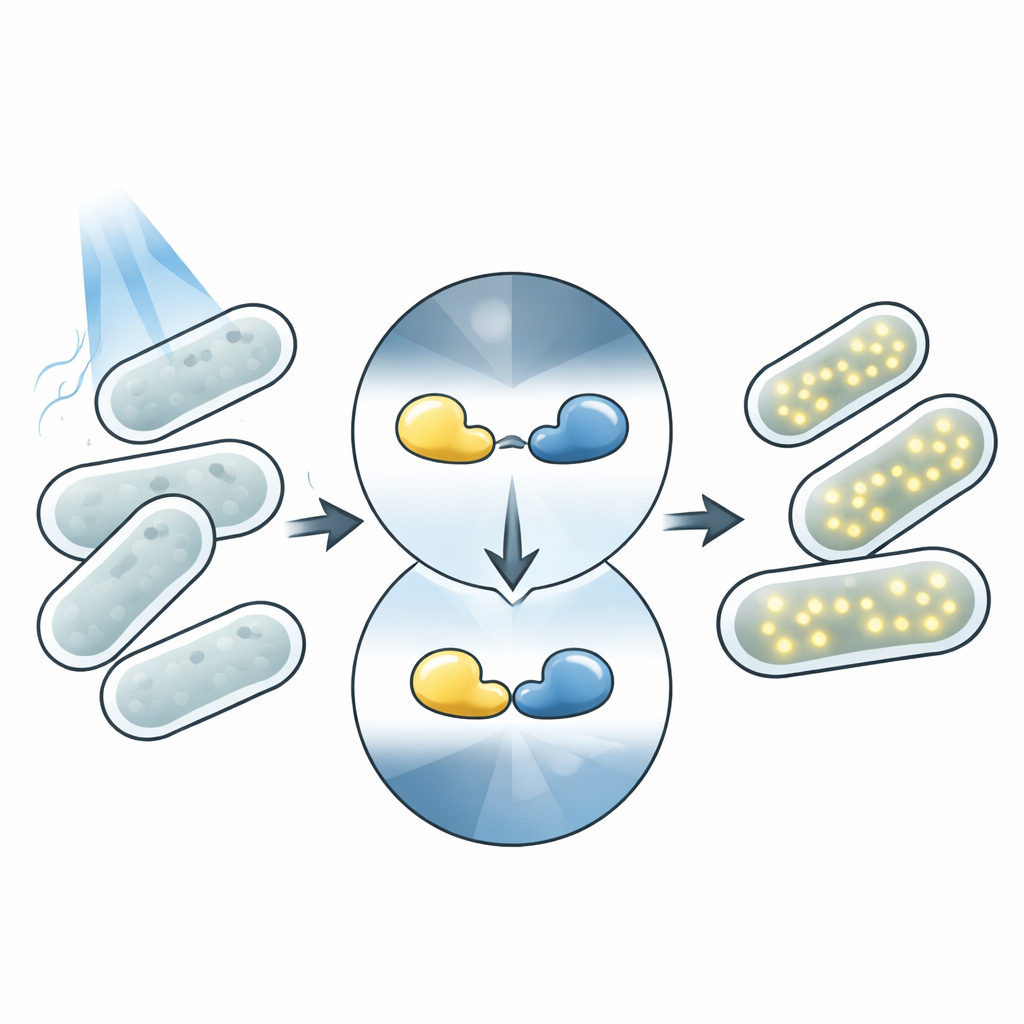
Light is an ideal remote control for biology. Unlike chemical additives, light can be applied instantly, removed just as fast, and focused on tiny regions without touching the cells. Many organisms naturally contain proteins that change shape when they absorb light, triggering downstream effects. Modern optogenetics repurposes these proteins as controllable parts: a light-sensing "head" is fused to an "action" module such as an enzyme or gene switch. When light shines, the sensor changes shape and activates the attached function. Magnets photosensors are a pair of such proteins that stick together under blue light and fall apart in the dark, making them a workhorse tool for building light-controlled systems.
How the Magnet Switch Works in Cells
In this work, the team focuses on a blue-light responsive system called Opto-T7RNAP. Here, the Magnets pair is fused to two halves of a split enzyme, T7 RNA polymerase, which drives expression of a target gene. In the dark, the halves stay apart and the gene is mostly silent. Under blue light, the Magnets domains bind each other, pulling the enzyme halves together so the gene turns on and produces a red fluorescent protein that can be easily measured. This setup tightly links the DNA sequence of each light-sensing protein to a visible output, allowing the researchers to read out performance of thousands of variants simply by measuring how brightly the cells glow.
Random Change and Careful Selection
Rather than guessing which single amino acids to tweak, the authors used random mutagenesis to introduce changes throughout each Magnet protein and then let the data reveal which versions worked best. They created large libraries of mutated nMag and pMag variants and introduced them into bacteria carrying the Opto-T7RNAP and fluorescent reporter gene. Using fluorescence-activated cell sorting, they performed a multi-step selection: first enriching cells that glowed strongly under dim blue light, then discarding cells that glowed too much in the dark, and finally isolating individual clones for detailed testing. The glow level of each variant was followed either cell-by-cell by flow cytometry or in bulk cultures over time with automated spectrophotometry.
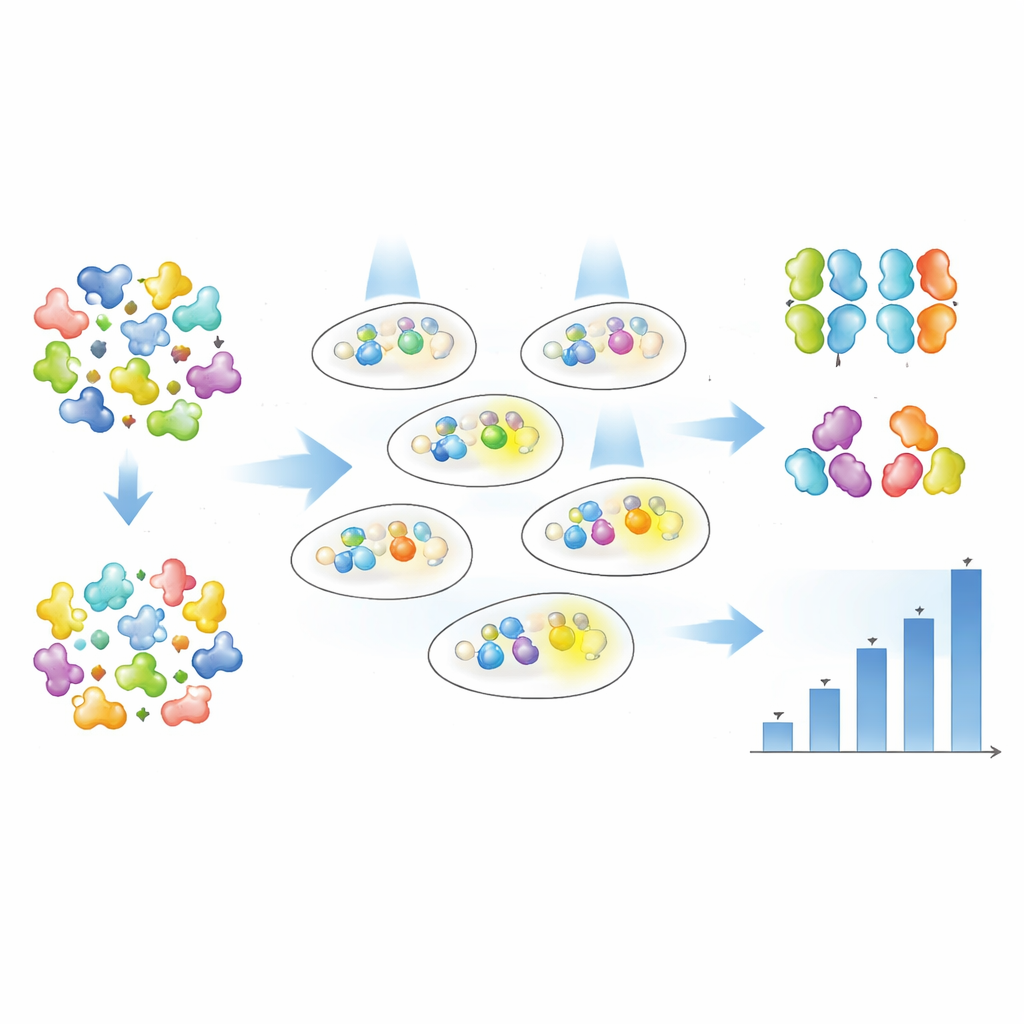
Fine-Tuning Sensitivity and Strength
From just one round of screening, the team found 19 distinct Magnet variants with a rich mix of behaviors. Some mutants made the system much more light sensitive, meaning the same gene output could be reached with far lower light intensity. Others mainly increased how strongly the gene was turned on at full light, and some did both. Importantly, the authors showed that “how much light is needed” (sensitivity) and “how much gene output you get” (activation) can be tuned largely independently. In some variants, half-maximal activation was reached with roughly half the light, yet the maximum output stayed similar; in others, maximum output increased several-fold while sensitivity stayed roughly the same. By comparing many variants across temperatures, they also identified versions that maintain stronger activity at body-like conditions and have robust behavior throughout the growth of the cell population.
New Building Blocks for Light-Controlled Biology
To a non-specialist, the main message is that these scientists have turned a single light switch into an entire dimmer panel. Instead of one fixed blue-light sensor, they now offer a menu of Magnet variants that require more or less light, produce stronger or milder gene responses, or perform well across different temperatures. Because Magnets are already widely used in synthetic biology and have been adapted to mammalian cells, these improved versions can be plugged into many existing designs. In practical terms, that means experiments that need gentle, long-term illumination can use highly sensitive variants, while applications that must avoid accidental activation by room light can use less sensitive ones. The work thus broadens the toolbox for precisely sculpting cellular behavior with light in research and biotechnology.
Citation: Baumschlager, A., Weber, Y., Cánovas, D. et al. Enhancing the performance of Magnets photosensors. Nat Commun 17, 4138 (2026). https://doi.org/10.1038/s41467-026-70695-7
Keywords: optogenetics, light-controlled gene expression, protein engineering, photosensory proteins, synthetic biology