Clear Sky Science · en
Deciphering the liquid-solid interactions in dealkalization of O3 layered oxides
Why this battery study matters
As the world looks for cleaner and cheaper ways to store renewable energy, sodium-based batteries are emerging as a promising partner to today’s lithium-ion cells. But a subtle surface chemistry problem makes some of the most attractive sodium battery materials unstable when they meet liquids during manufacturing. This study digs into that hidden interaction between solid battery particles and the liquids used to clean them, and shows how choosing the right solvent can make the difference between a robust battery and one that falls apart before it ever reaches the grid.
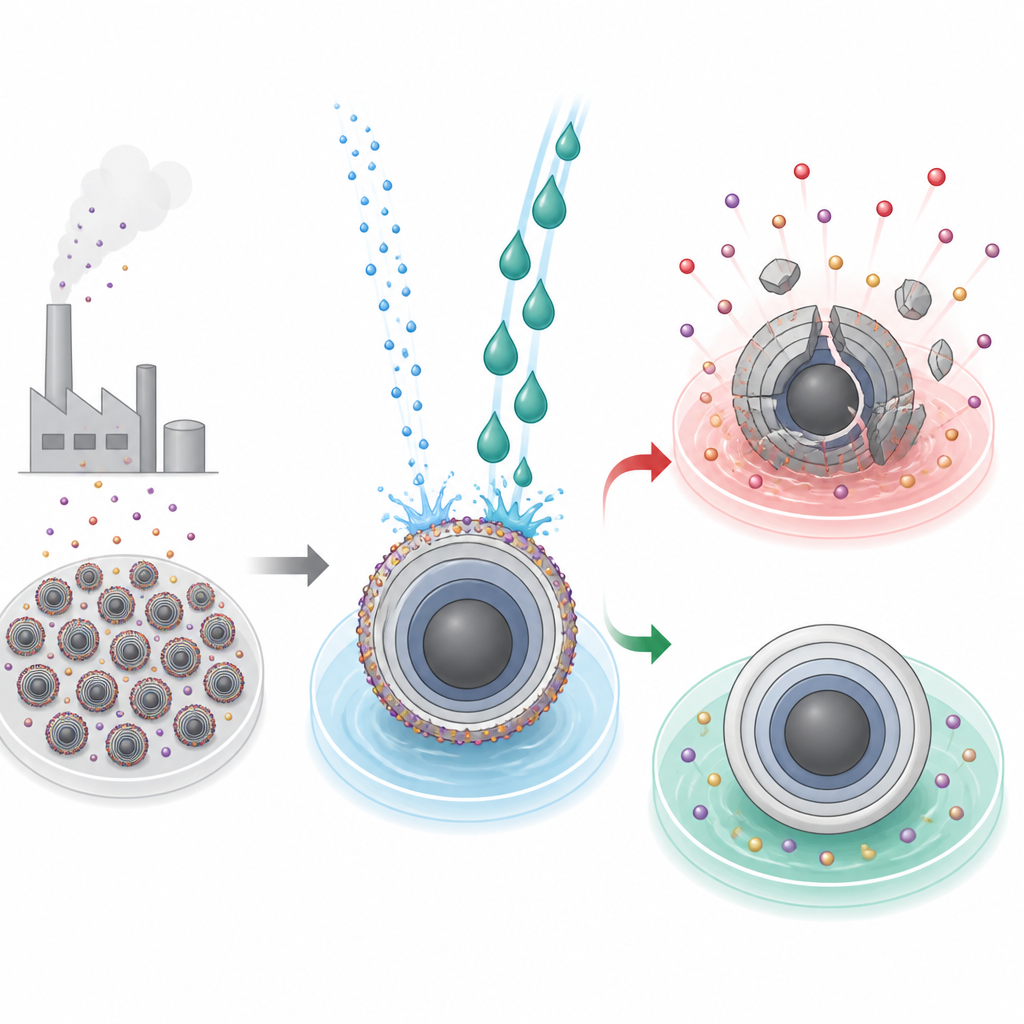
The hidden grime on battery particles
Modern high-energy batteries rely on layered oxide particles in the positive electrode, where ions move in and out during charging and discharging. To make these materials, manufacturers use strongly alkaline conditions that leave behind an unwanted crust of residual alkali compounds on the particle surfaces. In lithium-based materials, this crust can be washed away with water using established industrial processes. For sodium-based counterparts, however, the same wash can spell disaster: water not only removes the residue but also strips out active sodium ions and destabilizes the crystal framework, leading to cracks, structural collapse, and a sharp loss of battery capacity.
Two liquids, two very different outcomes
The researchers focused on a typical sodium layered oxide, NaNi1/3Fe1/3Mn1/3O2, already used in pilot-scale sodium batteries. They compared water with ethylene glycol, a common automotive coolant, as cleaning liquids. Both solvents efficiently dissolved the unwanted surface alkali, but their impact on the underlying particles diverged. Ethylene-glycol–treated material showed smoother particle surfaces, better electrode slurry flow during manufacturing, stronger adhesion to current collectors, higher recovered capacity, and improved cycling stability. In contrast, water-washed samples suffered from deep cracks, loss of sodium throughout the particle, slower ion transport, and much lower capacity, along with greater gas generation linked to leftover carbonate breakdown at high voltage.
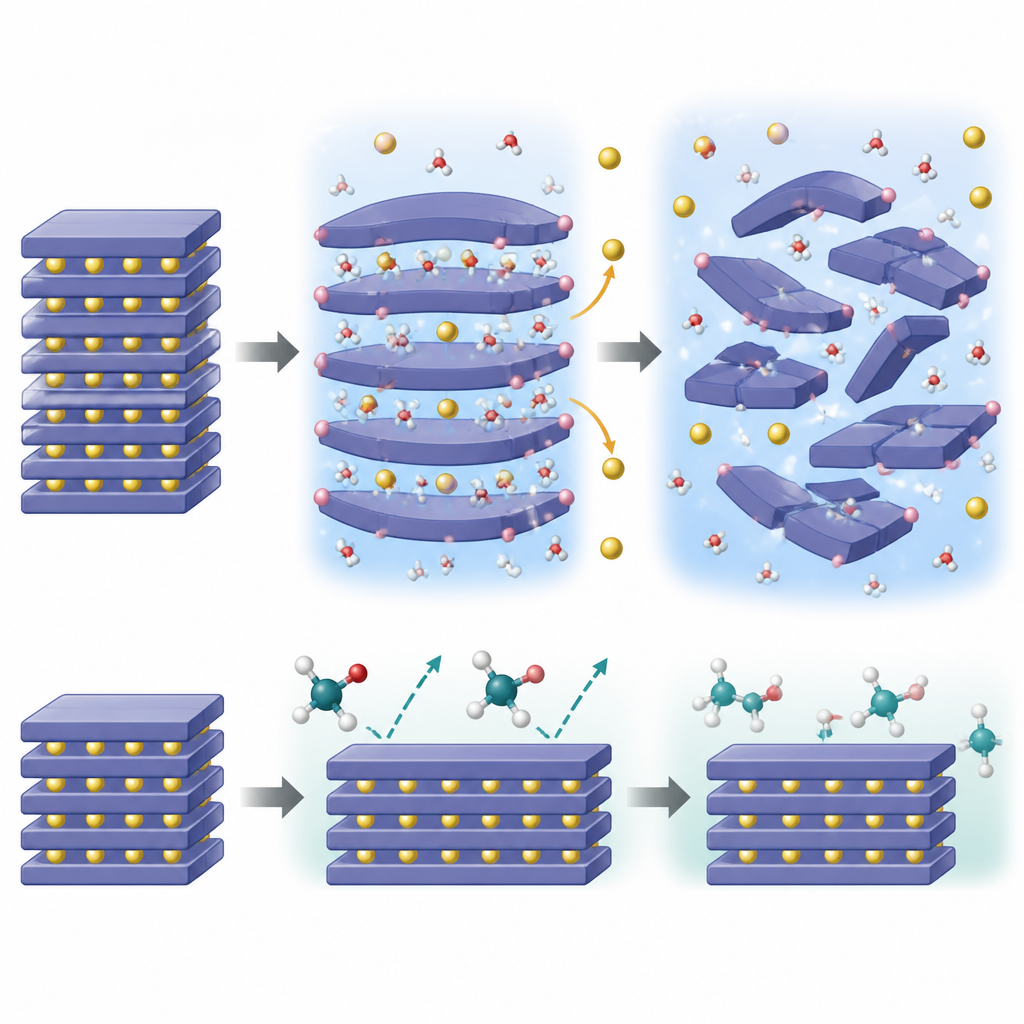
What happens inside the particles
To understand why water is so destructive, the team combined advanced imaging, surface chemistry probes, neutron diffraction, and temperature-dependent X-ray measurements with quantum-mechanical simulations. They found that small water molecules can slip between the sodium layers inside the crystal, expand the spacing, and then release protons that bond to the oxygen framework. This “self-propagating” invasion leads to sodium ions leaking out to keep charge balance, shrinking the sodium layers and triggering a cascade of phase changes and lattice distortions. Over time, the once-ordered layered structure transforms into more disordered forms with slipped or prismatic layers, eventually losing its ability to store and release sodium reversibly.
Why ethylene glycol behaves more gently
The calculations show that ethylene glycol molecules are simply too bulky to easily wedge themselves between the sodium layers. Their insertion into the structure is energetically unfavorable, so their action remains confined near the surface. Although ethylene glycol can still help adjust surface alkalinity and remove residual sodium compounds, it does not set off the same internal chain reaction of proton invasion and sodium loss. Measurements confirm that mass transfer between the solid and the glycol liquid is limited, and the layered framework stays largely intact even after treatment. This “size effect” means ethylene glycol can scrub the surface without prying open the structure from within.
From one material to a general design rule
Beyond this specific sodium oxide, the authors extended their analysis to other lithium- and sodium-based layered cathodes. They showed that whether water molecules can spontaneously intercalate depends on the spacing between alkali-ion layers and the strength of the bond between those ions and oxygen. Lithium oxides, with smaller ions and tighter layers, resist deep water penetration, so damage remains mostly at the surface. Sodium oxides, with wider layers and weaker bonds, are much more vulnerable unless specific transition metals are substituted to strengthen the bonding. The team proposes a simple stability yardstick based on the loss of active alkali ions, which can be used to compare how different materials and solvents fare during liquid processing.
What this means for future batteries
In plain terms, this work shows that not all cleaning liquids are equal when it comes to preparing sensitive battery materials. Water, while cheap and convenient, can quietly hollow out sodium-based layered oxides from the inside, robbing them of the very ions they need to store energy. Ethylene glycol, by contrast, can remove harmful surface residue while leaving the internal structure largely untouched. By linking these outcomes to molecular size, bonding strength, and layer spacing, the study offers practical guidelines for choosing and designing liquid treatments that protect performance rather than undermine it, helping sodium batteries move closer to reliable large-scale use.
Citation: Zhang, W., Zhu, J., Song, A. et al. Deciphering the liquid-solid interactions in dealkalization of O3 layered oxides. Nat Commun 17, 4166 (2026). https://doi.org/10.1038/s41467-026-70581-2
Keywords: sodium batteries, cathode materials, surface chemistry, solvent effects, energy storage