Clear Sky Science · en
MFF budding from mitochondria regulates melanosome size and maturation
Why the Color-Making Machinery of Cells Matters
Our skin, hair, and eyes owe their color to tiny pigment factories inside cells called melanosomes. Keeping these structures the right size and in good working order is essential not only for appearance, but also for protecting our tissues from sunlight. This study uncovers a surprising player in how melanosomes grow, divide, and mature: a protein best known for shaping mitochondria, the cell’s power plants. By showing that this protein also sculpts melanosomes, the researchers connect energy-producing organelles with pigment-making ones, offering fresh clues to how color is controlled in health and disease.
A New Job for a Mitochondrial Helper
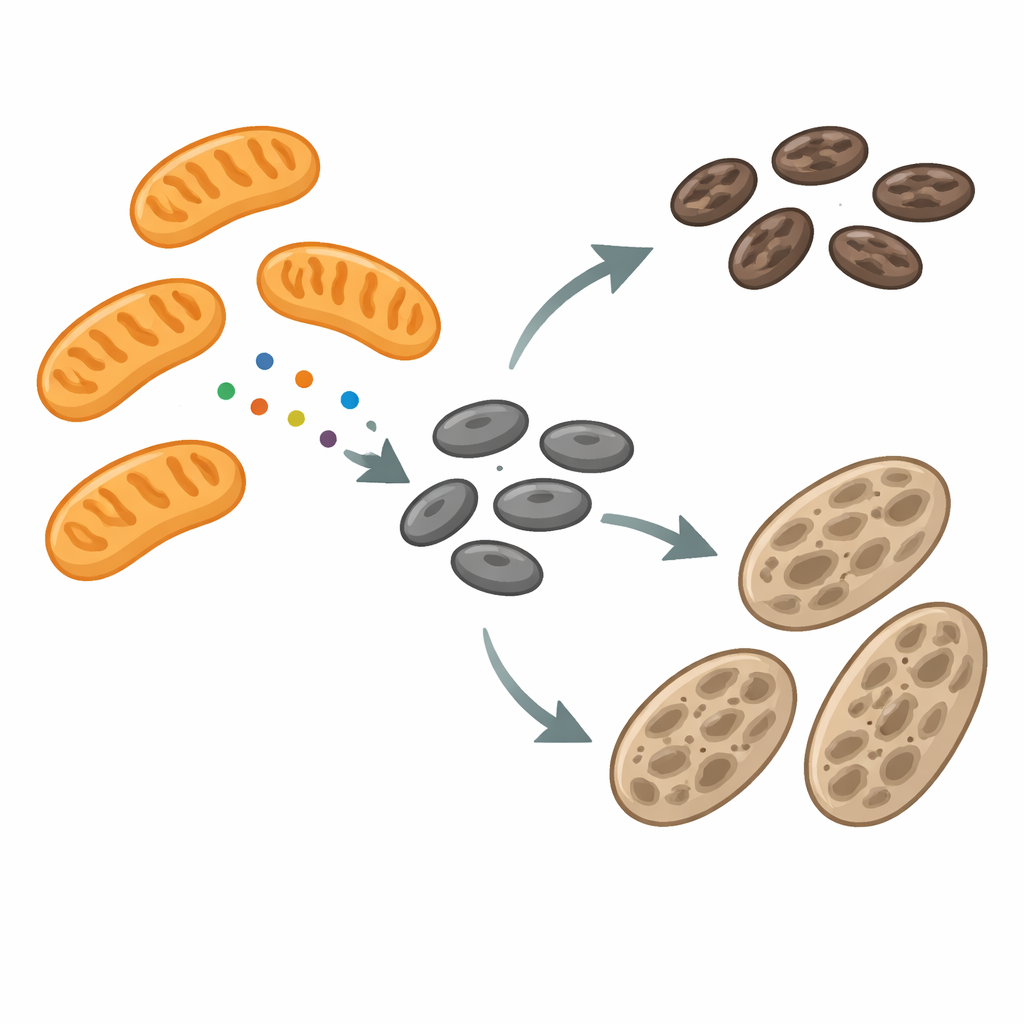
Melanosomes are specialized compartments that make and store melanin, the dark pigment that shields cells from ultraviolet damage. As they develop, melanosomes pass through several stages, gradually filling with pigment before being passed to neighboring skin cells. During this journey, their membranes constantly remodel—stretching out into tubules, pinching off small pieces, and sometimes fusing together. The machinery that drives this remodeling has been only partly understood. The authors focused on a protein called mitochondrial fission factor (MFF), known for helping mitochondria divide. To their surprise, they found MFF not just on mitochondria, but also on melanosomes, especially at sites where these pigment granules narrow and appear ready to split.
When Pigment Stalls, Contacts and Signals Change
To probe how mitochondria and melanosomes communicate, the team compared normal pigment-producing mouse cells with albino cells that cannot make melanin because of a faulty tyrosinase gene. The albino cells accumulated many immature melanosomes and showed more frequent physical contacts between melanosomes and mitochondria. RNA sequencing and protein analysis revealed that, in these albino cells, genes and proteins linked to mitochondria were turned up, including MFF located on the outer mitochondrial membrane. Yet mitochondrial shape looked largely normal, while melanosomes were both larger and more dynamic, undergoing frequent fission and fusion. This pattern hinted that MFF might be repurposed away from mitochondria toward melanosomes, where it could influence how these pigment organelles split and mature.
MFF Travels to Melanosomes and Controls Their Splitting
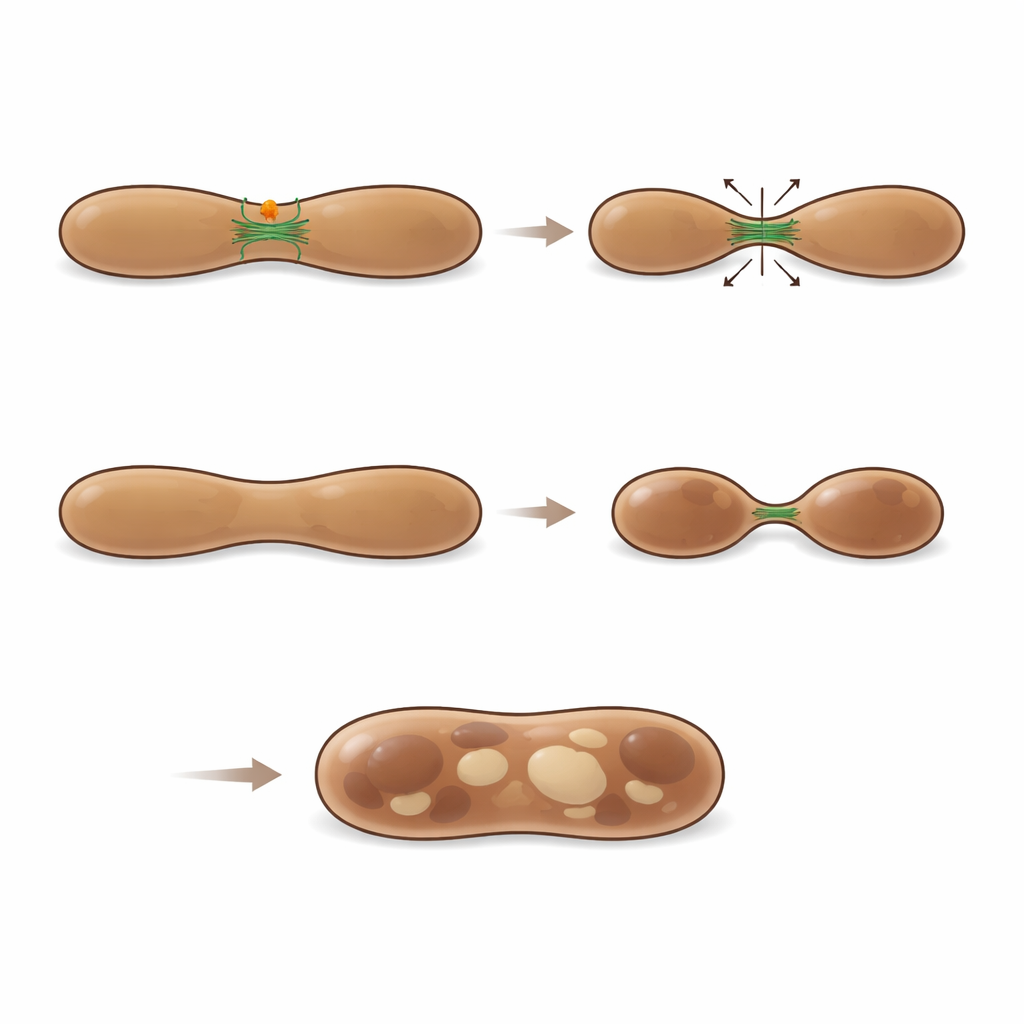
Using high-resolution microscopy and immunogold electron microscopy, the researchers traced MFF directly on the surface of melanosomes at all stages of their life cycle. Time-lapse imaging even captured MFF leaving mitochondria as small puncta and moving onto LAMP1- and LysoTracker-marked melanosomes. At these sites, melanosome membranes often narrowed and pinched, forming tubules and vesicles. Quantitative analysis showed that about a quarter of visible melanosome fission regions were decorated by MFF, suggesting it marks a major class of splitting events. When the scientists reduced MFF levels in pigment cells, early melanosomes became abnormally enlarged, often appearing as hybrid structures that had features of both immature and more advanced stages. These swollen organelles shared melanin more extensively with early markers, and live imaging showed fewer successful fission events, underscoring that MFF is needed for proper division and maturation of melanosomes.
Balancing Breakdown and Building with Actin Filaments
MFF’s usual partner in mitochondrial division is another protein, DRP1. However, in melanosomes, DRP1 and MFF rarely overlapped, and silencing each one had very different consequences. Loss of DRP1 mainly dampened the genetic program that drives pigment production. In contrast, loss of MFF enlarged early melanosomes and triggered a strong increase in genes and proteins linked to lysosomes—cellular recycling centers—and to catabolic enzymes such as cathepsin B. These enzymes concentrated inside melanosomes, making them more acidic and more prone to break down their own pigment machinery. When cathepsin B was also silenced, melanosomes in MFF-deficient cells swelled further and accumulated in large numbers, showing that enhanced internal digestion normally serves as a backup to prevent buildup of dysfunctional, oversized pigment granules.
How the Cell’s Skeleton Helps Slice Pigment Granules
To understand how MFF physically helps melanosomes divide, the authors mapped which proteins bind to it. They found numerous regulators of the actin cytoskeleton, including components of the WASH complex and the ARP2/3 actin-nucleating machinery. Imaging showed actin filaments clustering at melanosome necks where MFF puncta sat, bridging constricted regions between two budding compartments. When key ARP2/3 subunits were silenced, early melanosomes enlarged in much the same way as when MFF was lost, indicating that actin-driven filament formation is essential for proper splitting. Inhibiting ARP2/3 activity also erased the melanosome-enlarging effect of MFF loss, tying MFF function tightly to actin dynamics rather than to DRP1. Together, these findings reveal that MFF recruits or stabilizes an actin-based fission machine at melanosomes, enabling them to shed unneeded components and progress toward fully pigmented, properly sized organelles.
What This Means for Pigment Disorders
The study shows that MFF, long viewed as a specialist in mitochondrial division, also acts outside mitochondria to regulate the size, shape, and maturation of melanosomes through actin-driven fission. When MFF is missing, early pigment granules cannot split properly, grow too large, and increasingly resemble degradative compartments, prompting the cell to ramp up their breakdown. These insights help explain how giant melanosomes arise in certain inherited forms of albinism and related conditions, and they highlight MFF and its actin partners as potential targets for modulating pigmentation or understanding how pigment cells malfunction in disease.
Citation: Magalhães Rebelo, A.P., Maracani, A., Greco, S. et al. MFF budding from mitochondria regulates melanosome size and maturation. Nat Commun 17, 3932 (2026). https://doi.org/10.1038/s41467-026-70572-3
Keywords: melanosomes, mitochondria, MFF protein, pigmentation, actin cytoskeleton