Clear Sky Science · en
Proteomic landscape of Ewing sarcoma primary tumors and metastases
Why this childhood cancer study matters
Ewing sarcoma is a rare but aggressive bone cancer that mainly strikes children and teenagers. Families and doctors face a troubling puzzle: some tumors respond well to chemotherapy and patients do well, while others come back or spread despite the same treatments. This study takes an unusually deep look at the actual proteins inside 170 tumors from 74 young patients, asking what makes one tumor easier to treat than another and how the body’s own immune cells shape the disease.
Looking inside tumors at the protein level
Most previous work on Ewing sarcoma has focused on DNA and RNA, which tell us what the cancer could do. Proteins reveal what the cancer is actually doing. The researchers used advanced mass spectrometry to measure more than 10,000 proteins in each tumor sample, taken from patients at different stages: newly diagnosed, after chemotherapy, during relapse, and in metastases that had spread to the lungs or bones. By carefully dissecting the tumor regions and filtering for high cancer-cell content, they created one of the most detailed protein maps of this disease to date. This map let them compare how tumors change over time and how those changes relate to treatment response and survival.
How treatment reshapes the cancer
When the team compared primary tumors with samples taken after chemotherapy, they saw sweeping shifts in the protein landscape. Proteins involved in cell division, DNA repair, and building new cellular machinery dropped sharply after treatment, consistent with chemotherapy knocking back fast-growing cells. But in tumors that later relapsed or progressed, many of these same proteins rose again, suggesting that surviving cells reactivated growth and repair programs. At the same time, post-treatment tumors showed higher levels of immune-related proteins and markers of low-oxygen stress, hinting that chemotherapy leaves behind a stressed, inflamed, yet adaptable tumor remnant rather than a clean slate.
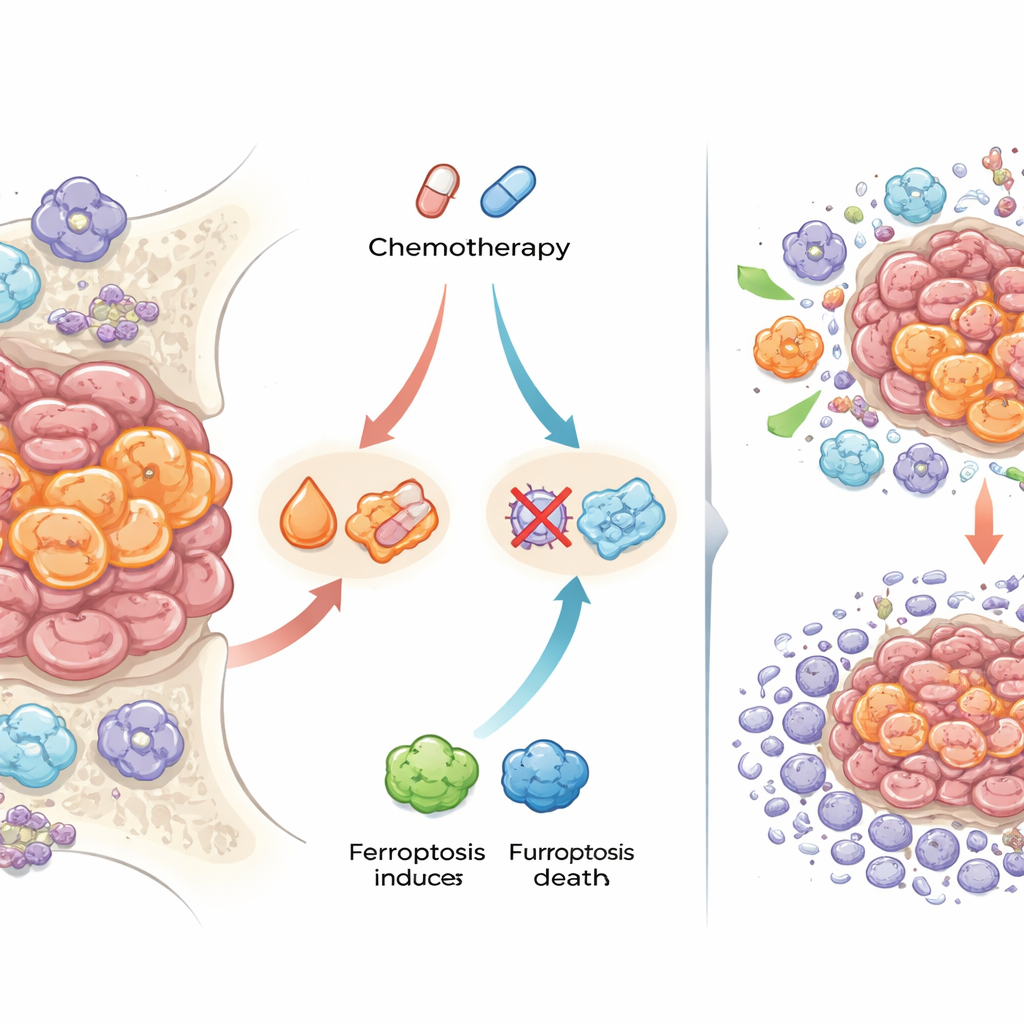
Iron, cell death, and drug resistance
One of the most striking findings involved iron-handling proteins and a special form of cell death called ferroptosis, which depends on iron-driven damage to cell membranes. After chemotherapy, cancer cells in many tumors carried higher levels of ferritin (which stores iron) and transferrin receptor (which imports iron), and stained positive for free iron deposits. They also expressed proteins known to block ferroptosis and reduce oxidative damage. To test whether this mattered for treatment, the researchers used lab-grown 3D “spheroids” of Ewing sarcoma cells that had been made resistant to standard drugs. When they combined chemotherapy with compounds that trigger ferroptosis, the resistant spheroids became more sensitive, needing lower doses of drug to be killed. This suggests that some Ewing sarcoma cells may survive chemotherapy precisely because they buffer iron and avoid ferroptotic death.
Immune cells that help or harm
Because tumors do not grow in isolation, the team also examined how immune cells within the tumor environment relate to outcome. By grouping primary tumors based only on their protein patterns, they discovered three major subclasses with different survival rates. One high-risk group showed many cell-cycle and ubiquitin-related proteins and low activity of the core proteasome, together with protein signatures of neutrophils and “checkpoint” molecules that can dampen immune attack. Another, better-outcome group had higher levels of proteins involved in presenting tumor fragments on the cell surface, a key step in activating T cells. Multiplexed immunofluorescence imaging confirmed that tumors in poor-outcome groups were richer in neutrophils, whereas those in better-outcome groups contained more macrophages, T cells, and HLA-positive antigen-presenting cells. In simple terms, “neutrophil-heavy” tumors tended to behave worse, while tumors with more engaged adaptive immune cells tended to behave better.
What this means for future treatment
Put together, this work shows that Ewing sarcoma is more than a single gene error; it is a complex ecosystem of cancer cells, iron metabolism, and immune players that shifts under the pressure of therapy. For patients and clinicians, the findings point to two promising ideas. First, drugs that push cancer cells toward ferroptosis might boost the effects of existing chemotherapy, especially in resistant disease. Second, protein-based markers of neutrophils, macrophages, T cells, and antigen presentation could help identify which patients might benefit from immune-based therapies or combination strategies. While more validation is needed, this proteomic atlas offers a roadmap for turning the hidden biology of Ewing sarcoma into more precise, and potentially more successful, treatments for young patients.
Citation: Gordon, S., Mohan, V., Shukrun, R. et al. Proteomic landscape of Ewing sarcoma primary tumors and metastases. Nat Commun 17, 3802 (2026). https://doi.org/10.1038/s41467-026-70449-5
Keywords: Ewing sarcoma, proteomics, ferroptosis, tumor microenvironment, pediatric cancer