Clear Sky Science · en
A bacterial YopJ-family acetyltransferase suppresses host immune response by Nε-acetylation of JAK1
How Bacteria Quiet the Body’s Alarm System
When harmful microbes invade, our cells normally sound the alarm by releasing chemical messengers that switch on powerful antiviral and antibacterial defenses. This study reveals how the pneumonia-causing bacterium Legionella pneumophila deploys a tiny protein tool, called Lem17, to muffle that alarm at its source. By tampering with a central cellular switch, the JAK‑STAT pathway, the bacterium can slip past immune defenses and multiply inside our cells.
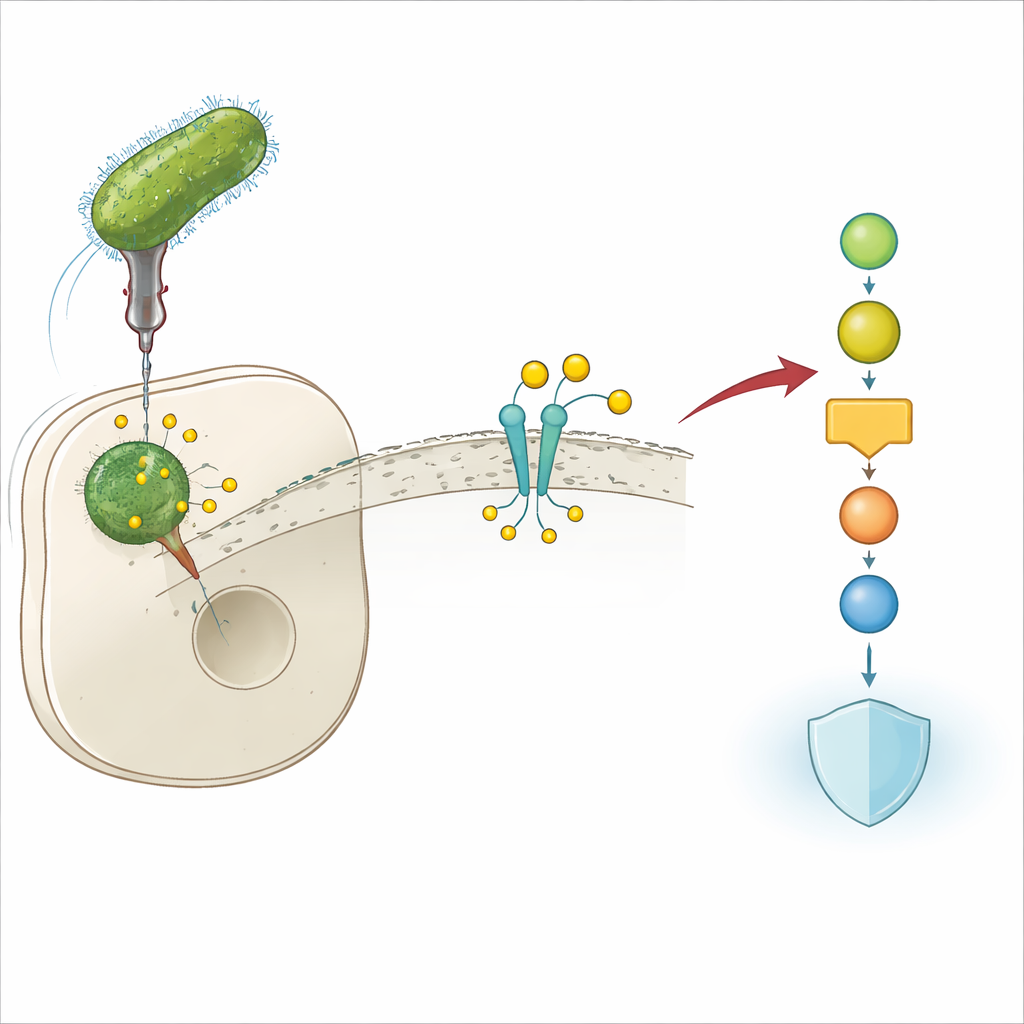
A Bacterial Trick to Evade Defenses
Legionella pneumophila lives inside human immune cells and can cause a severe form of pneumonia known as Legionnaires’ disease. To survive in this hostile environment, the bacterium injects many "effector" proteins into its host cell. These effectors rewire key cellular circuits that would otherwise block infection. The authors discovered that one of these effectors, Lem17, is crucial for Legionella’s ability to grow inside cells: when the gene for Lem17 is deleted, the bacteria replicate poorly in mouse and human cell models. This immediately suggested that Lem17 plays a central role in disarming host defenses.
The Body’s Signaling Switch Under Attack
Our cells rely on the JAK‑STAT pathway to translate distress signals, such as interferons and interleukins, into a broad antiviral and antibacterial response. These signaling molecules bind to receptors on the cell surface, which then recruit an enzyme called JAK1. JAK1 activates downstream STAT proteins that move into the nucleus and turn on hundreds of protective genes. The researchers showed that Lem17 specifically targets JAK1. It binds directly to JAK1 at a region that normally recognizes cytokine receptors, effectively competing with those receptors and blocking JAK1 from being properly recruited when danger signals arrive.
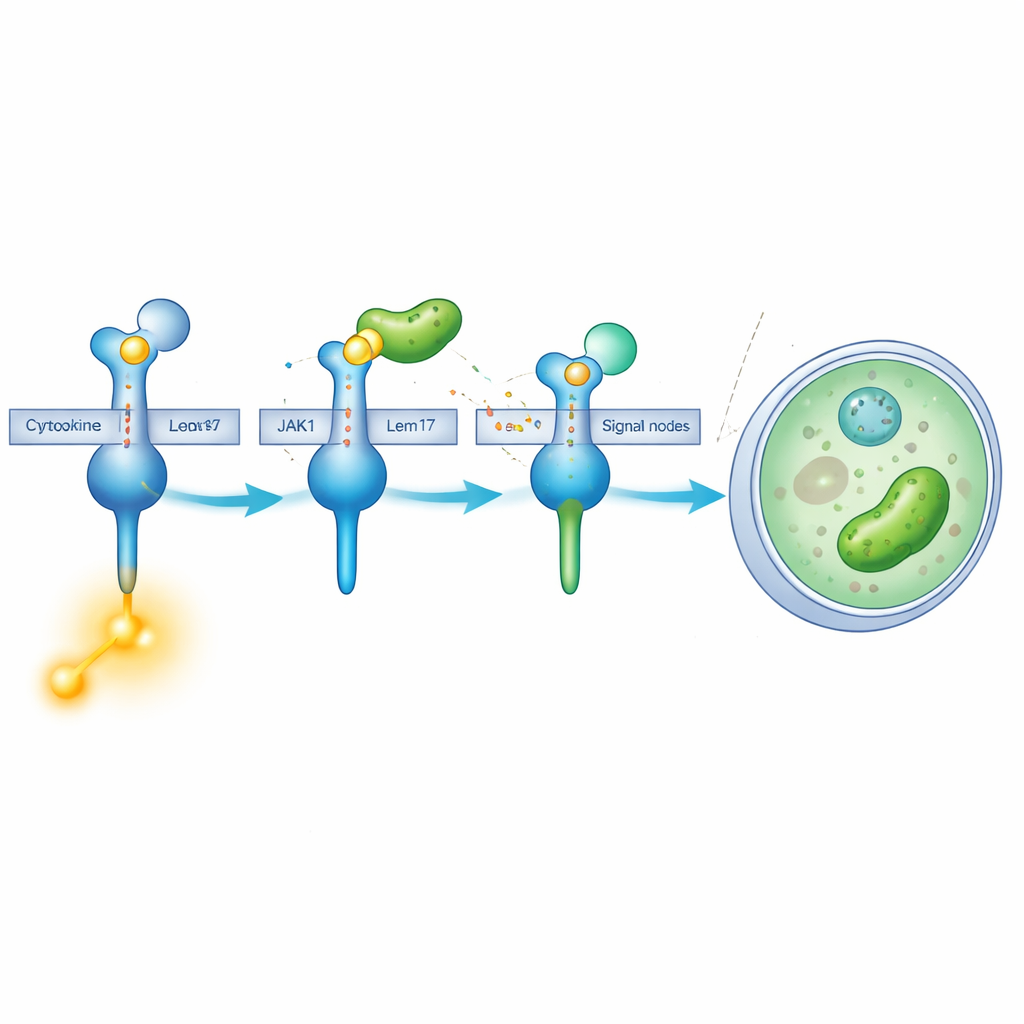
Chemical “Masking” of a Key Enzyme
Lem17 does more than just get in the way. It belongs to a family of enzymes that attach small acetyl groups onto other proteins. Using cell-based experiments and mass spectrometry, the team found that Lem17 decorates several lysine sites on JAK1 with these acetyl marks. Many of the modified sites cluster in two important regions: one that helps JAK1 dock onto receptors and another that performs its kinase function—the chemical step that turns signals into action. When JAK1 is acetylated by Lem17, its ability to use energy molecules and phosphorylate substrates drops sharply, meaning the signaling cascade is effectively shut down.
Host Molecule Turns the Bacterial Weapon On
The study also explains how Lem17 itself is activated only inside host cells. Structural work revealed that Lem17 binds tightly to inositol hexaphosphate (IP6), a small molecule naturally abundant in eukaryotic cells but not in bacteria. IP6 fits into a positively charged pocket in Lem17 and locks into place under a flexible "lid" domain. Without IP6, Lem17 cannot bind its acetyl donor molecule and remains inactive. This requirement ensures that Lem17’s acetyltransferase activity switches on only after Legionella has entered a host cell, preventing premature activation inside the bacterium.
Immune Silencing and Bacterial Growth
By measuring gene activity and protein modifications in human cells treated with interferon, the authors showed that Lem17 strongly dampens JAK‑STAT signaling: phosphorylation of STAT1 and STAT2 is reduced, and many interferon-stimulated genes fail to turn on. Cells in which JAK1 is removed or silenced become more permissive to Legionella replication, mimicking the effect of Lem17. Together, these results support a clear model: Legionella uses Lem17 to bind and chemically mask JAK1, blocking the relay of cytokine signals and allowing the bacterium to thrive inside a protected cellular niche.
Why This Matters for Infection and Therapy
This work uncovers a precise way in which a bacterial pathogen disables one of the immune system’s central alarm switches. By showing that Lem17 both competes with receptors for access to JAK1 and chemically inactivates JAK1’s function, the study highlights the JAK‑STAT pathway as a key battleground in bacterial infections, not just in viral disease. Understanding this mechanism could guide future strategies to boost host defenses, for example by designing drugs that block Lem17’s interaction with JAK1 or prevent its acetyltransferase activity, thereby restoring the cell’s ability to mount a robust immune response.
Citation: Chen, TT., Zheng, SR., Yang, B. et al. A bacterial YopJ-family acetyltransferase suppresses host immune response by Nε-acetylation of JAK1. Nat Commun 17, 2910 (2026). https://doi.org/10.1038/s41467-026-69623-6
Keywords: Legionella pneumophila, JAK-STAT signaling, bacterial effectors, protein acetylation, innate immunity