Clear Sky Science · en
Active pixel power control for crosstalk-free all-optical neural interrogation
Shining Light on the Brain Without Disturbing It
Modern neuroscience often relies on light to both watch and control the activity of brain cells. This powerful idea—using lasers to read and write neural signals—promises deep insight into how brains generate behavior and disease. But there is a catch: the very light used to take pictures of neural activity can accidentally trigger the same light-sensitive switches that scientists use to control neurons, muddying the results. This paper introduces a way to carefully tailor the laser light at each tiny point in the image so that researchers can monitor and manipulate the brain at the same time, without the two tasks interfering with each other.
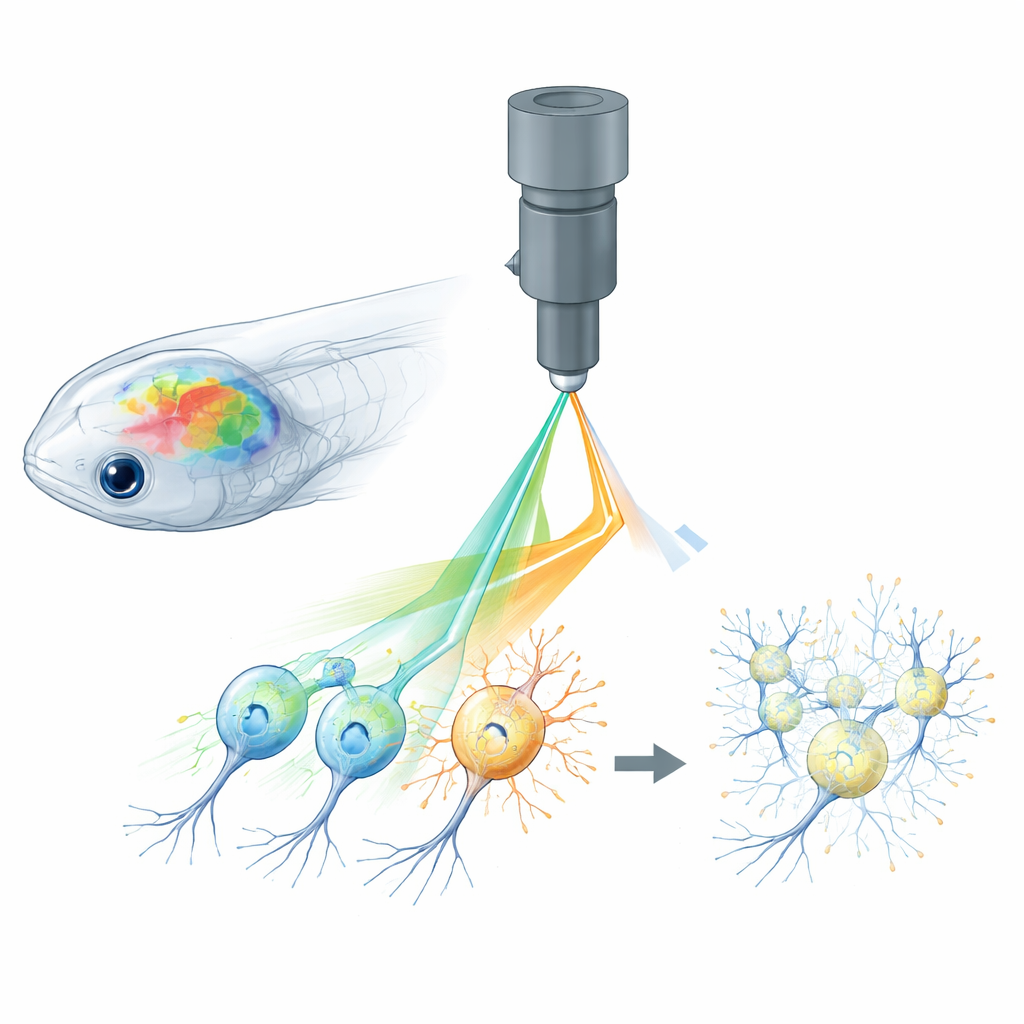
Why Watching and Controlling Neurons Is Hard
All-optical neuroscience combines two tools: glowing sensors that report when neurons are active, and light-activated proteins that let scientists turn neurons on or off. In small animals like zebrafish, fruit flies, and mice, two-photon microscopes can focus laser light deep into the brain to read out whole networks of cells in 3D, while holographic patterns of light stimulate chosen neurons with pinpoint accuracy. However, the same laser used to read calcium signals from fluorescent sensors can unintentionally activate the light-gated channels used for control. This “cross-talk” means that the imaging process itself alters the brain’s activity, blurring the line between genuine responses and artifacts created by the experiment.
Pinpoint Power Control at the Pixel Level
The authors tackle this problem with what they call active pixel power control, or APPC. Instead of shining the same laser power across every point in the image, they use a very fast light modulator to adjust the laser intensity for each tiny pixel as the beam scans. Before experiments, they record where the light-activated channels are located by imaging a standard fluorescent tag fused to those proteins. From this map, they construct a custom power pattern: pixels that overlap with light-sensitive regions get much less power, while other pixels keep the higher power needed for clear calcium signals. The modulator updates in real time, synchronized with the fast scanning mirrors of the microscope, so that laser power is sculpted across the brain with single-pixel precision.
Testing the Method in a Tiny Transparent Brain
To see if APPC truly prevents unwanted activation, the team worked in larval zebrafish, whose small, see-through brains are ideal for whole-brain imaging. They used popular optogenetic channels (such as ChR2 and a red-shifted variant called ChrimsonR) together with a green calcium sensor, all driven by a single femtosecond laser. By systematically lowering imaging power only on neurons that expressed light-sensitive channels, while keeping normal power elsewhere, they found a “sweet spot” around 5 milliwatts where imaging still produced reliable calcium signals but no longer drove extra, artificial activity in those neurons. Importantly, they showed that this local power reduction also prevented cross-talk effects from spreading into downstream neurons, preserving the true wiring and signal flow of the circuit.
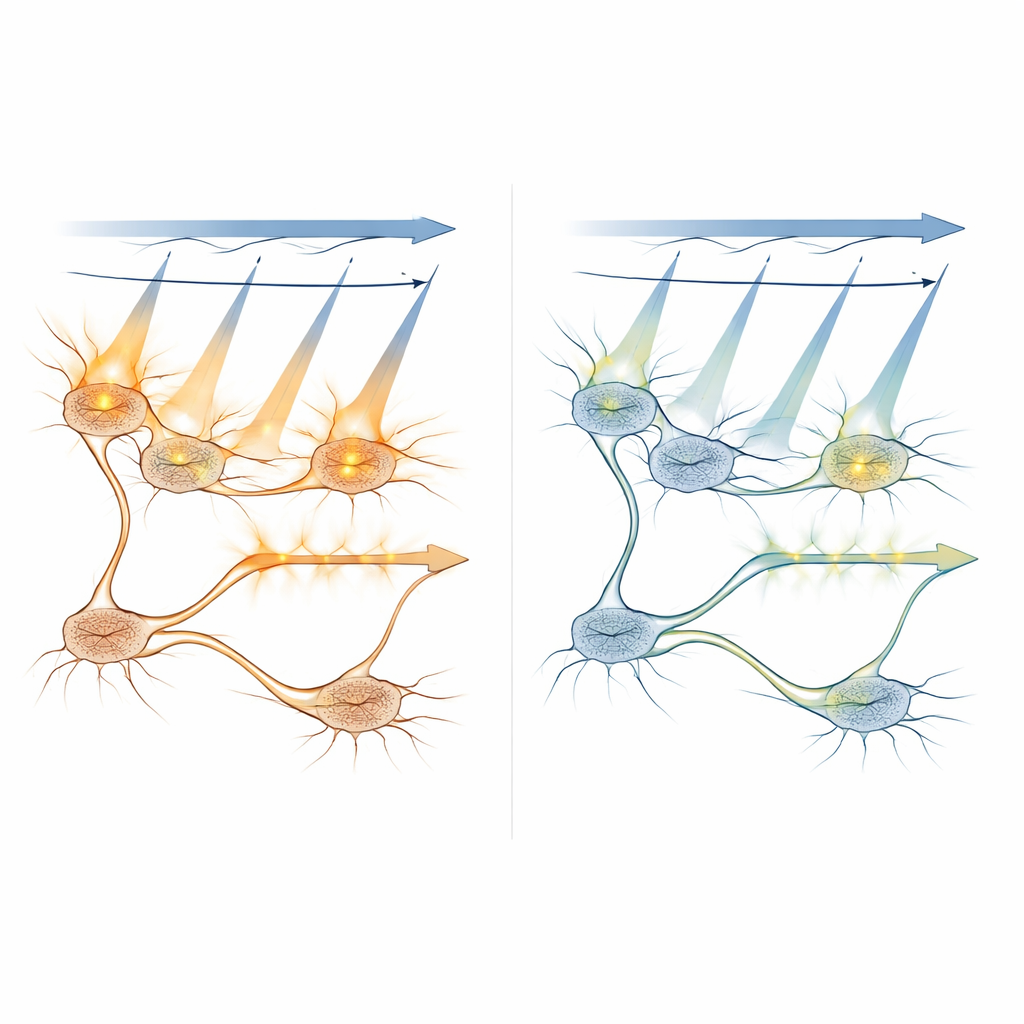
Exploring How Light Spreads and Activates Cells
The researchers combined computer models with in vivo measurements to understand how scanning light activates light-sensitive proteins under different conditions. They simulated how often individual molecules would be switched on as the laser swept across a neuron, and how this probability changed with laser power, exposure time, and how far the focal point was from the center of the cell. Experiments confirmed that out-of-focus planes more than about 8–10 micrometers away contribute little to unwanted activation, helping to define safe spacing between imaging layers in 3D scans. They also tested a strategy that restricts the bright imaging light to the inner part of the cell body—away from the membrane where the light-gated channels sit—showing that this can further cut cross-talk, even though it reduces signal strength.
Making Precision Brain Experiments More Accessible
Beyond offering a clever technical fix, APPC has practical advantages. It works with standard two-photon microscopes that are already common in many labs, needs only one laser, and does not require specialized engineered proteins or perfect color separation between tools. The authors argue that APPC can be extended to more complex systems, including setups with two lasers, where it would complement other strategies that separate stimulation and imaging by wavelength. By empirically tuning the “near cross-talk-free” power level for each neuron type and experiment, APPC provides a general recipe for studying how specific cells influence brain-wide activity without accidentally stirring up extra signals. In everyday terms, it lets scientists dim the light exactly where it would otherwise blind their own instruments, clearing the view of the living brain in action.
Citation: Yan, G., Tian, G., Fu, Y. et al. Active pixel power control for crosstalk-free all-optical neural interrogation. Nat Commun 17, 3195 (2026). https://doi.org/10.1038/s41467-026-69419-8
Keywords: optogenetics, two-photon imaging, neural circuits, zebrafish brain, active pixel power control