Clear Sky Science · en
Structural basis of glucosinolate recognition and transport by plant GTR1
Plant Shields with a Hidden Cost
Many of the vegetables on our plates, from mustard greens to canola oil’s parent plant rapeseed, defend themselves with a family of spicy, sulfur‑rich chemicals called glucosinolates. These compounds can deter hungry insects and may even have health benefits for humans, but in animal feed and some crops they are considered anti‑nutrients. This study uncovers, in atomic detail, how a tiny gatekeeper protein in plant cells, called GTR1, recognizes and moves glucosinolates, opening the way to crops that are both better defended and more nutritious.
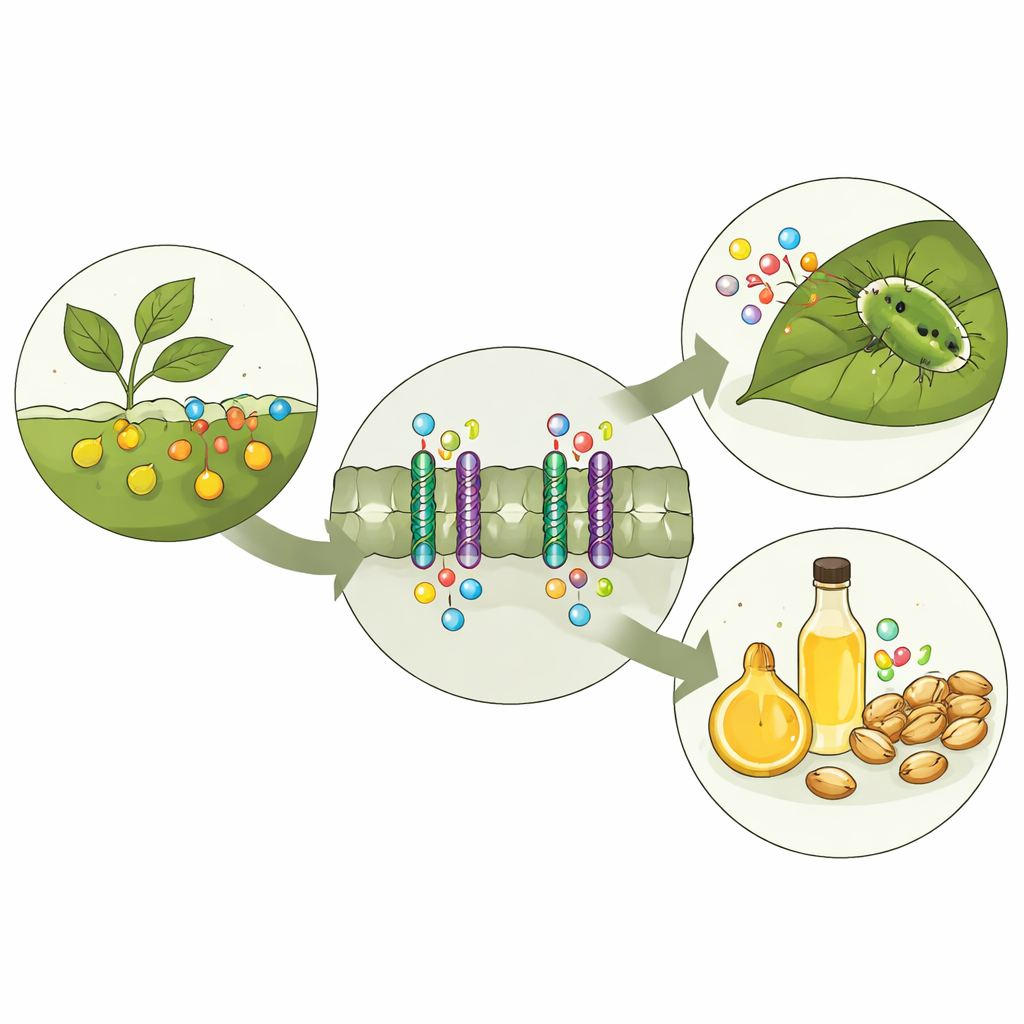
A Chemical Security System in Plants
Glucosinolates are stored separately from the enzymes that break them down. When an insect chews a leaf, the compartments rupture, and glucosinolates are quickly converted into sharp‑tasting and sometimes toxic products that discourage further feeding. To make this defense work, plants must ship glucosinolates from where they are made to where they are needed, such as seeds and specialized storage cells. In the model plant Arabidopsis, three related transporter proteins, GTR1, GTR2, and GTR3, handle this job. Earlier genetic work showed that when these transporters are disrupted, glucosinolate levels and patterns across the plant change, altering both defense and seed quality.
The Gate in the Cell Wall
GTR1 sits in the cell’s outer membrane and acts as a revolving door that couples glucosinolate uptake to the flow of protons (hydrogen ions) across the membrane. Using cryo‑electron microscopy, the authors captured four three‑dimensional snapshots of Arabidopsis GTR1: two without cargo (one open to the outside, one open to the inside) and two with bound glucosinolates. These images reveal that GTR1 has a core of twelve membrane‑spanning segments that form a central cavity, surrounded by a large “intracellular domain” that nestles against one of the helices. By trimming this domain or changing key contact points, the researchers showed that it is essential for proper transport, likely acting as a stabilizing scaffold that keeps the moving parts aligned.
How GTR1 Recognizes Its Cargo
The team examined how GTR1 binds two representative glucosinolates with different side chains. Both were found lodged in the same central pocket. The sugar and charged sulfate portions—features shared by virtually all glucosinolates—are gripped by a cluster of positively charged and polar amino acids on one side of the cavity. By systematically changing these residues, the authors showed that two of them, including a lysine that is part of a hallmark sequence shared across this transporter family, are crucial for transport. In contrast, amino acids that touch the variable side chains proved less important for activity, explaining how GTR1 can handle many glucosinolates. Subtle differences in these side‑chain‑contacting positions between GTR1, GTR2, and GTR3 likely tune which specific glucosinolates each protein prefers.
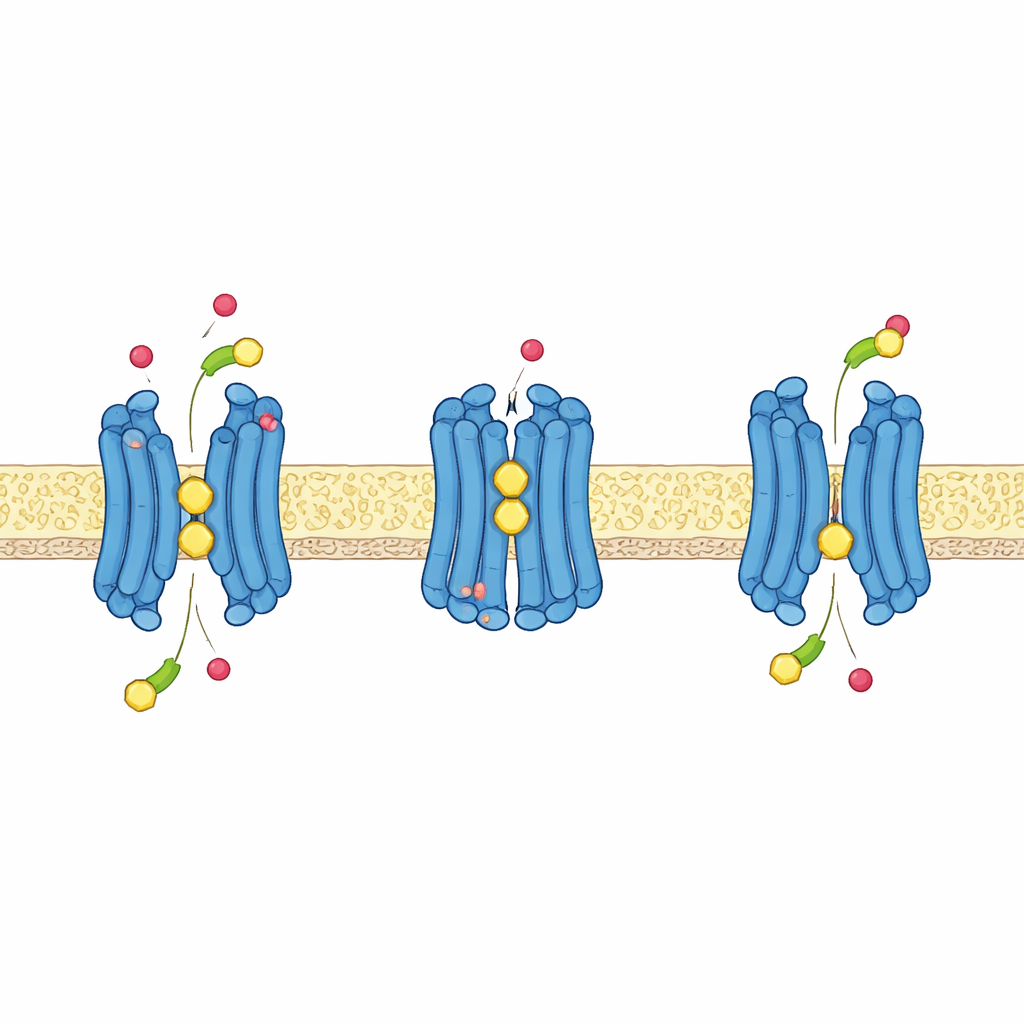
Using Protons to Power Transport
Like many nutrient transporters in plants, GTR1 taps the energy stored in a proton gradient—more protons outside the cell than inside—to actively pull glucosinolates inward. The structures, combined with computer simulations and transport tests at different acidity levels, reveal how this works at the atomic level. A short stretch of amino acids near the top of the central cavity, containing two glutamates and a lysine, rearranges as the protein switches between outward‑ and inward‑facing states. When certain glutamates take up protons, they release their grip on the lysine, freeing it to help bind the negatively charged sulfate group of glucosinolates and encouraging the protein to close around its cargo. Another glutamate, deeper in the protein, forms a key partnership with a nearby tyrosine; when protonated, this site helps drive the transporter from the outward‑facing to the inward‑facing state and makes the process sensitive to the cell’s electrical voltage.
From Atomic Pictures to Better Crops
Together, these results support an alternating‑access model in which GTR1 cycles through outward‑open, occluded, and inward‑open shapes while co‑transporting protons and glucosinolates. By clarifying exactly which protein features recognize the common glucosinolate “backbone,” which tune side‑chain preferences, and which couple transport to proton flow, the study provides a detailed blueprint for engineering glucosinolate movement in plants. In practical terms, this knowledge could be used to breed or design crops whose seeds accumulate fewer bitter, anti‑nutritional glucosinolates while leaves and stems retain strong chemical defenses against insects—offering farmers plants that are both hardier in the field and more valuable as food and feed.
Citation: Yan, R., Fan, J., Chi, C. et al. Structural basis of glucosinolate recognition and transport by plant GTR1. Cell Discov 12, 26 (2026). https://doi.org/10.1038/s41421-026-00884-7
Keywords: glucosinolates, plant transporters, GTR1, crop improvement, proton-coupled transport