Clear Sky Science · en
The role of NINJ1 in diseases
When Cells Choose to Burst
Our cells are wrapped in delicate outer skins that keep the inside of life separate from the outside world. For decades, scientists assumed that when those skins finally tear during cell death, it was just an unlucky pop—like a balloon bursting. This review article shows that, instead, a small membrane protein called NINJ1 acts like a built-in demolition switch. By deciding when and how cells burst open, NINJ1 helps shape inflammation, nerve repair, blood vessel growth, infection control, and even cancer—making it an attractive but tricky target for future medicines.
A Tiny Gatekeeper at the Cell’s Edge
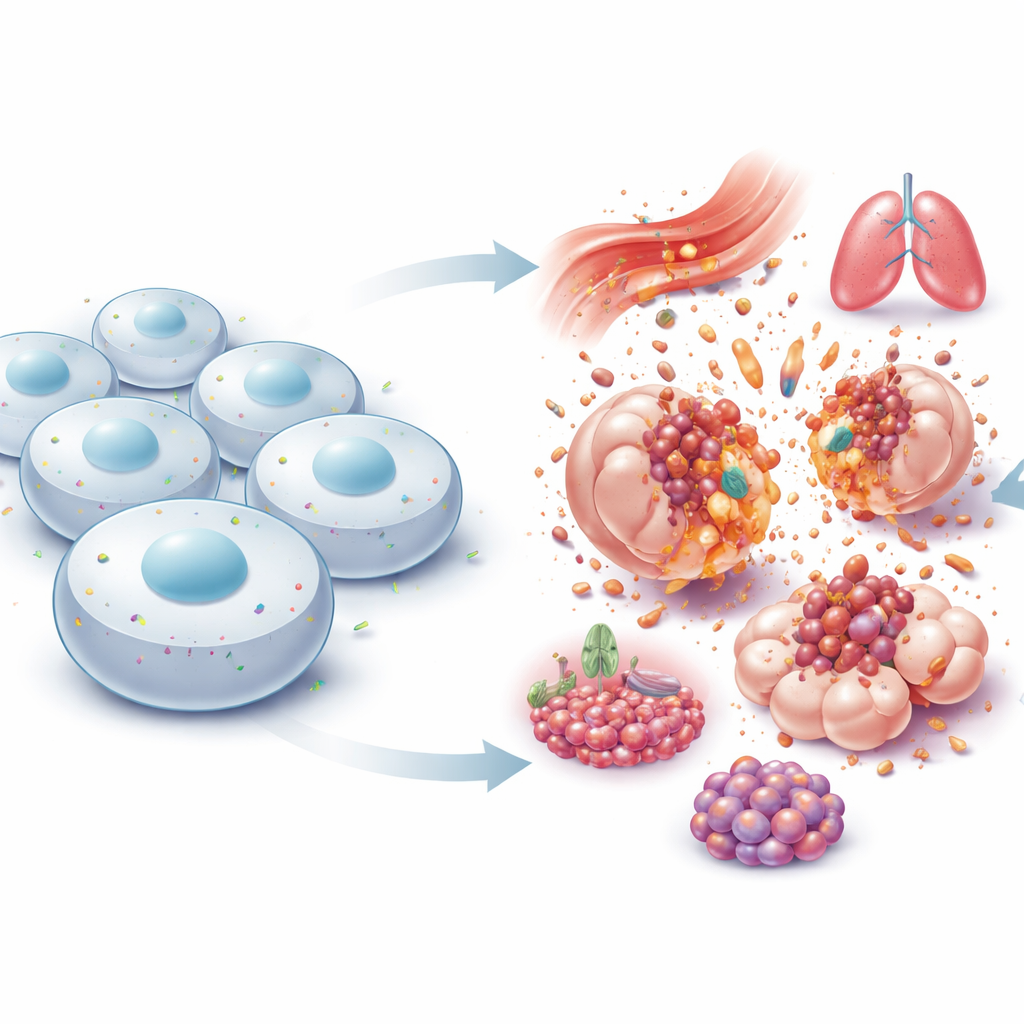
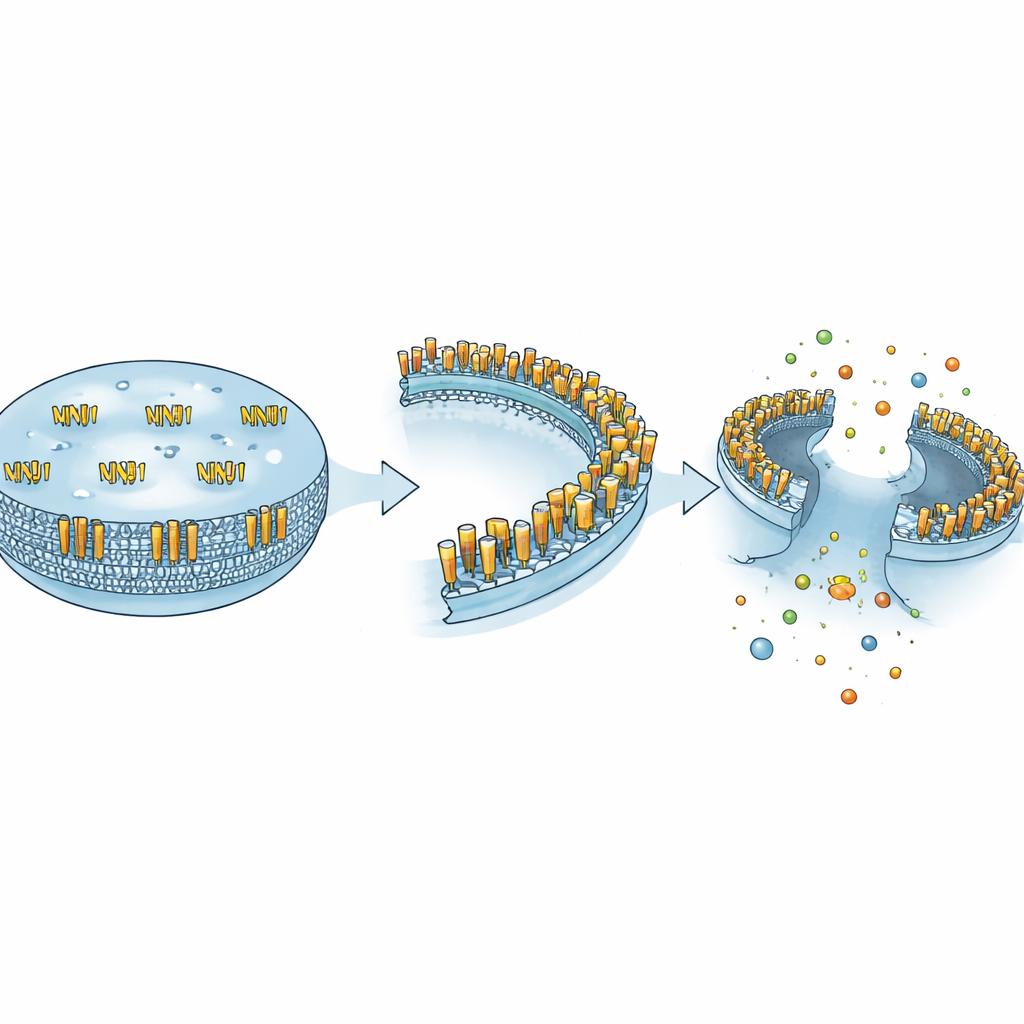
NINJ1 sits in the outer membrane of cells and was originally discovered in injured nerves, where it seemed to help damaged fibers regrow. For years, it was treated as a simple “sticky” protein that lets cells hold on to each other. That view changed in 2021, when researchers showed that NINJ1 is the active executor of the final tear in many forms of explosive cell death. Instead of the membrane failing passively, NINJ1 molecules change shape, gather into large ring-like structures, and slice out pieces of the membrane, allowing the cell’s contents to spill into surrounding tissue. This overturns the old idea of a purely accidental rupture and reveals that the last step of lytic cell death is itself carefully regulated.
One Protein, Three Faces
The authors argue that NINJ1’s seemingly contradictory behaviors can be understood by its ability to adopt different physical forms. In its quiet state, NINJ1 exists mainly as single units paired in dimers that help cells stick to one another. When a cutting enzyme such as MMP-9 clips off its outer segment, that fragment is released into the surrounding fluid and behaves like a chemokine—a small signal that attracts and guides immune cells. Under strong stress signals, NINJ1 units instead reorganize into big oligomeric rings anchored in the membrane, which carve out membrane patches and cause full rupture. These three forms—adhesive, soluble messenger, and membrane “cutter”—give one protein the ability to control cell contact, cell movement, and cell demolition, depending on context.
How Cell Bursting Shapes Health and Disease
Because NINJ1 governs whether cells gently fade away or explosively burst, it sits at the crossroads of many diseases. In inflammatory conditions like severe pancreatitis or gout, NINJ1-driven rupture amplifies pain and swelling by releasing danger signals from dying cells; blocking NINJ1 in animal models can ease tissue damage. In blood vessels, NINJ1 has a double life: certain forms worsen diabetic vessel injury, while its soluble fragment can calm inflammation in artery plaques. During infections, NINJ1-mediated bursting helps the body expose microbes and rally immune defenses, yet in sepsis and severe viral disease, the same process can fuel overwhelming, body-wide inflammation and clotting. In the brain and nerves, NINJ1 supports nerve regrowth and vessel repair in some settings, but also participates in damaging inflammation after stroke or trauma.
NINJ1 in Cancer and Future Treatments
In cancer, NINJ1 again shows two faces. Depending on the status of the tumor suppressor p53 and the surrounding tissue signals, it can either aid tumor growth and spread or help trigger inflammatory, therapy-induced cell death. This complexity makes NINJ1 a tempting but hazardous drug target. The review maps current experimental strategies: natural compounds and vitamins that lower NINJ1 levels, amino acids and small molecules that prevent its ring-like assemblies, and designer peptides or antibodies that block its adhesive or chemotactic roles. The authors argue that truly safe therapies must be “form-specific”—aimed at the pore-forming rings in acute tissue damage, at the soluble fragment in chronic inflammation, or at the adhesive form in diseases driven by harmful immune cell trafficking.
Turning a Cellular Breaker Switch into a Drug Target
Overall, the article concludes that NINJ1 is not a simple on–off switch for cell death, but a versatile control hub whose structure determines its job. By deciding whether a cell holds on, sends out help signals, or bursts apart, NINJ1 links together nerve repair, immunity, blood vessel health, infection outcomes, and cancer behavior. Harnessing this breaker switch for therapy will require pinpoint control—hitting the harmful form in the right cells at the right time while preserving its protective roles in host defense and tissue repair. If that precision can be achieved, NINJ1-based treatments could offer a new way to tame damaging inflammation and cell death across a wide range of diseases.
Citation: Bao, S., Chen, F., Guo, Z. et al. The role of NINJ1 in diseases. Cell Death Discov. 12, 193 (2026). https://doi.org/10.1038/s41420-026-03064-4
Keywords: NINJ1, cell death, inflammation, membrane rupture, therapeutic targets