Clear Sky Science · en
MYCN inhibits TrkC-mediated differentiation in neuroblastoma cells via disruption of the PKA signalling pathway
Why this childhood cancer study matters
Neuroblastoma is a childhood cancer that can behave in dramatically different ways: some tumors quietly mature and shrink, while others spread aggressively and resist treatment. This study asks a deceptively simple question with big clinical consequences: why do certain signals push some neuroblastoma cells to grow up into harmless nerve-like cells, but drive others to multiply as cancer? By uncovering how a powerful cancer gene called MYCN blocks a natural maturing signal, the authors reveal a potential new way to coax high‑risk tumors into a safer, more treatable state.
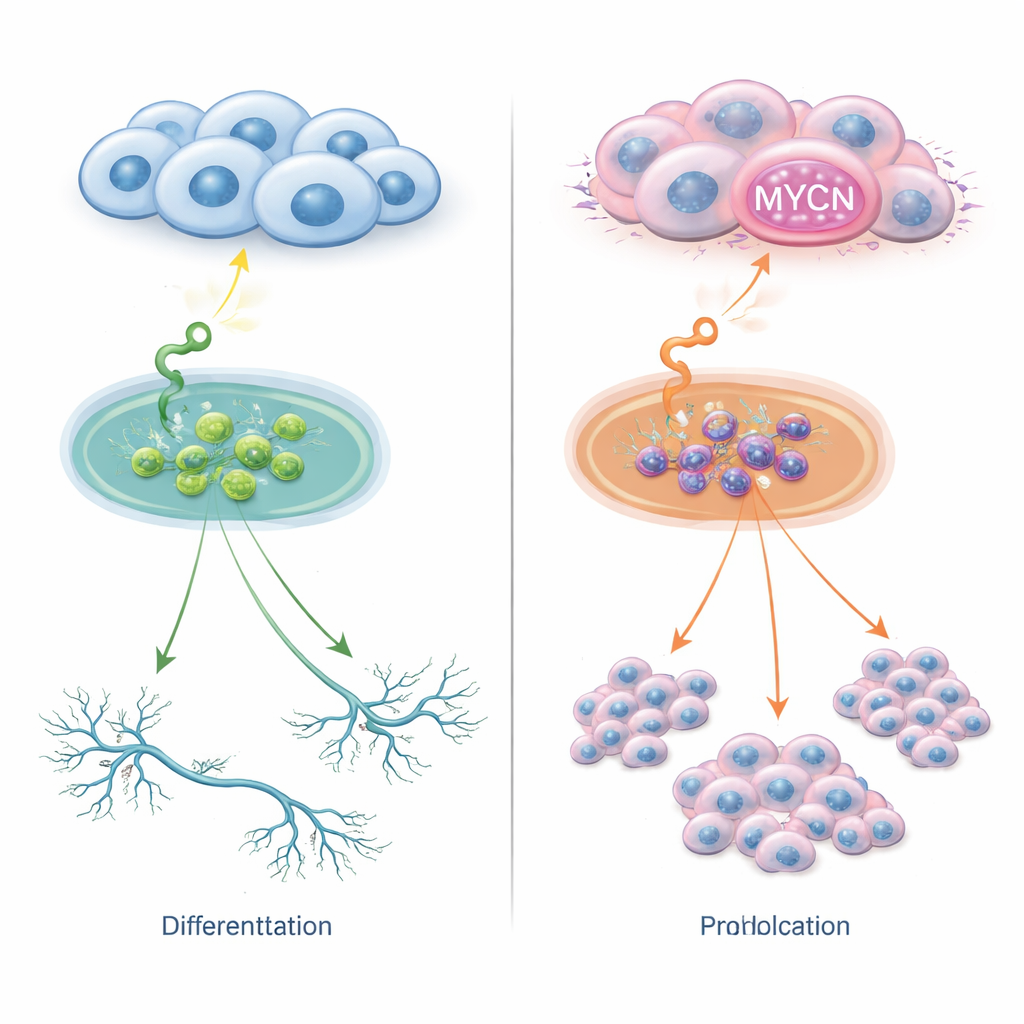
Two paths for the same signal
Neuroblastoma cells carry receptors on their surface that detect growth factors in their surroundings. One such receptor, called TrkC, senses a molecule named NT‑3. In ordinary settings, this partnership can encourage young nerve cells to develop into more mature forms. The researchers engineered several neuroblastoma cell lines to carry TrkC and then stimulated them with NT‑3. In cells without extra copies of the MYCN gene, NT‑3 pushed the cells to extend long, nerve‑like branches and adopt a more mature appearance, with little change in cell number. In stark contrast, in cells with too much MYCN—either through overexpression or full gene amplification—the very same NT‑3 signal no longer triggered maturation but instead fueled rapid cell division. Thus, a single outside cue can lead to either differentiation or aggressive growth, depending on how much MYCN the cells contain.
Following the signaling footprints inside the cell
To understand how this split in behavior arises, the team mapped chemical “on/off” switches inside the cells over time using a technique called phosphoproteomics. They compared thousands of modified proteins after NT‑3 stimulation in the different cell lines. Cells without MYCN amplification showed a strong and unique activation pattern, whereas MYCN‑rich cells had a more muted response. When the authors focused on which signaling enzymes were most active, a recurring player stood out in the differentiating cells: the protein kinase A (PKA) pathway and its partner CREB, a regulator in the cell nucleus that turns on genes needed for maturation. Pathway analysis confirmed that processes tied to nerve development and structural remodeling—such as axon guidance and cytoskeletal changes—were enriched specifically when PKA/CREB activity rose.
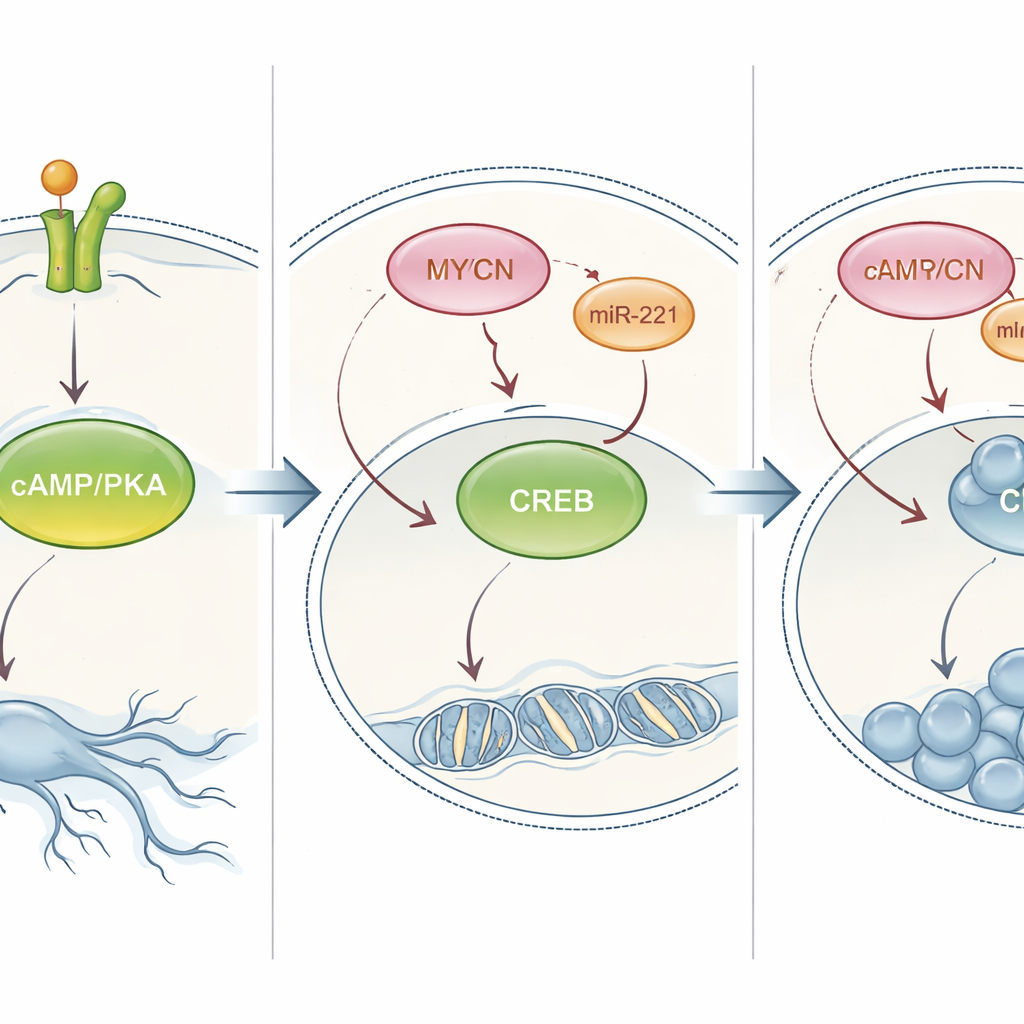
Flipping the PKA switch to change tumor behavior
The researchers next tested whether this PKA pathway truly controls the cell’s choice between maturing and multiplying. In cells that normally differentiated in response to NT‑3, chemically blocking PKA reduced their nerve‑like outgrowths and increased cell numbers. In MYCN‑amplified cells that usually only proliferate, artificially boosting PKA—either with a drug that mimics a natural messenger or by forcing the cells to make always‑active versions of PKA or CREB—reversed their behavior: the cells slowed division and grew long processes resembling neurons. This effect held not only in cell dishes but also in a zebrafish model, where activating PKA caused implanted human neuroblastoma tumors to shrink. Together, these experiments show that PKA/CREB signaling is a central lever that can redirect high‑risk neuroblastoma cells toward a more mature, less dangerous state.
How MYCN and a microRNA jam the differentiation circuit
The study then examined how MYCN shuts down this beneficial pathway. When MYCN levels were reduced in amplified cells, PKA and CREB proteins increased, and NT‑3 once again promoted differentiation instead of growth. Analyses of patient tumor data revealed that genes encoding PKA components are expressed at lower levels in MYCN‑amplified tumors than in other cases, suggesting that MYCN broadly dampens this pathway in children with high‑risk disease. The authors also uncovered a role for a small regulatory RNA, miR‑221, which is strongly associated with MYCN and poor outcomes. High miR‑221 levels correlated with MYCN expression in both tumors and cell lines. When miR‑221 was blocked in MYCN‑amplified cells, CREB levels rose and NT‑3 could once more drive neuronal maturation. This indicates that MYCN suppresses the PKA/CREB circuit both by turning down key genes and by raising miR‑221, which further lowers CREB.
What this means for future treatments
Overall, the work paints a clear picture: in low‑risk neuroblastoma, NT‑3 acting through TrkC and an active PKA/CREB pathway helps immature tumor cells grow up into benign, nerve‑like cells. In high‑risk tumors with extra MYCN, this maturation route is blocked, and the same signal is rerouted to fuel uncontrolled growth, aided in part by the microRNA miR‑221. By restoring PKA/CREB activity—using drugs that boost this pathway or by dampening MYCN‑driven blockers—clinicians may one day be able to convert aggressive neuroblastomas into more differentiated, treatable tumors. This strategy fits with a broader therapeutic goal in childhood cancer: not just to kill cancer cells, but to nudge them back onto a normal developmental path.
Citation: Maher, S., Roe, A., Wynne, K. et al. MYCN inhibits TrkC-mediated differentiation in neuroblastoma cells via disruption of the PKA signalling pathway. Cell Death Discov. 12, 176 (2026). https://doi.org/10.1038/s41420-026-03024-y
Keywords: neuroblastoma, MYCN, TrkC, PKA CREB pathway, tumor differentiation