Clear Sky Science · en
CSF1R T567M mutation induces microglial dysfunction and synaptic impairment in patient iPSC-derived cerebral organoids of CSF1R-related disorder
When the Brain’s Caretakers Go Awry
Some rare brain diseases strike adults in the prime of life, stealing memory, mood, and movement while brain scans reveal mysterious damage to the white matter—the wiring that connects brain regions. This study digs into one such disorder linked to a gene called CSF1R, which helps control the brain’s immune cells, known as microglia. By growing tiny lab-made brain models from patient cells, the researchers show how a single genetic change can throw these caretakers off balance, disrupt brain development, and weaken the connections between nerve cells that underlie thinking and behavior.
A Rare but Revealing Brain Disease
CSF1R-related disorder is an inherited, progressive condition that typically appears in adulthood and causes cognitive decline, movement problems, and psychiatric symptoms. It is closely related to a disease historically called ALSP, marked by damage to the brain’s white matter. Many different mistakes in the CSF1R gene have been found, most of them affecting a region that acts like the gene’s main on–off switch. The new work focuses on a recently discovered mutation, called T567M, that sits outside this classic control region. Because this spot had not been linked to disease before, scientists did not know whether or how it could cause such serious brain damage.
Building Mini-Brains from Patient Cells
To explore this question, the team took blood cells from a person carrying the T567M mutation and reprogrammed them into induced pluripotent stem cells—a versatile cell type that can be coaxed to become many tissues. From these, they created two matched sets of cells: one kept the patient’s mutation, and the other was precisely “repaired” using gene-editing tools, serving as a healthy comparison. They then grew three key brain components: microglia, nerve cells, and three-dimensional cerebral organoids, which are small, simplified models of the developing human brain. This setup allowed them to watch, side by side, how the mutation reshaped brain cells and their interactions.
Overactive Microglia and Stressed Brain Wiring
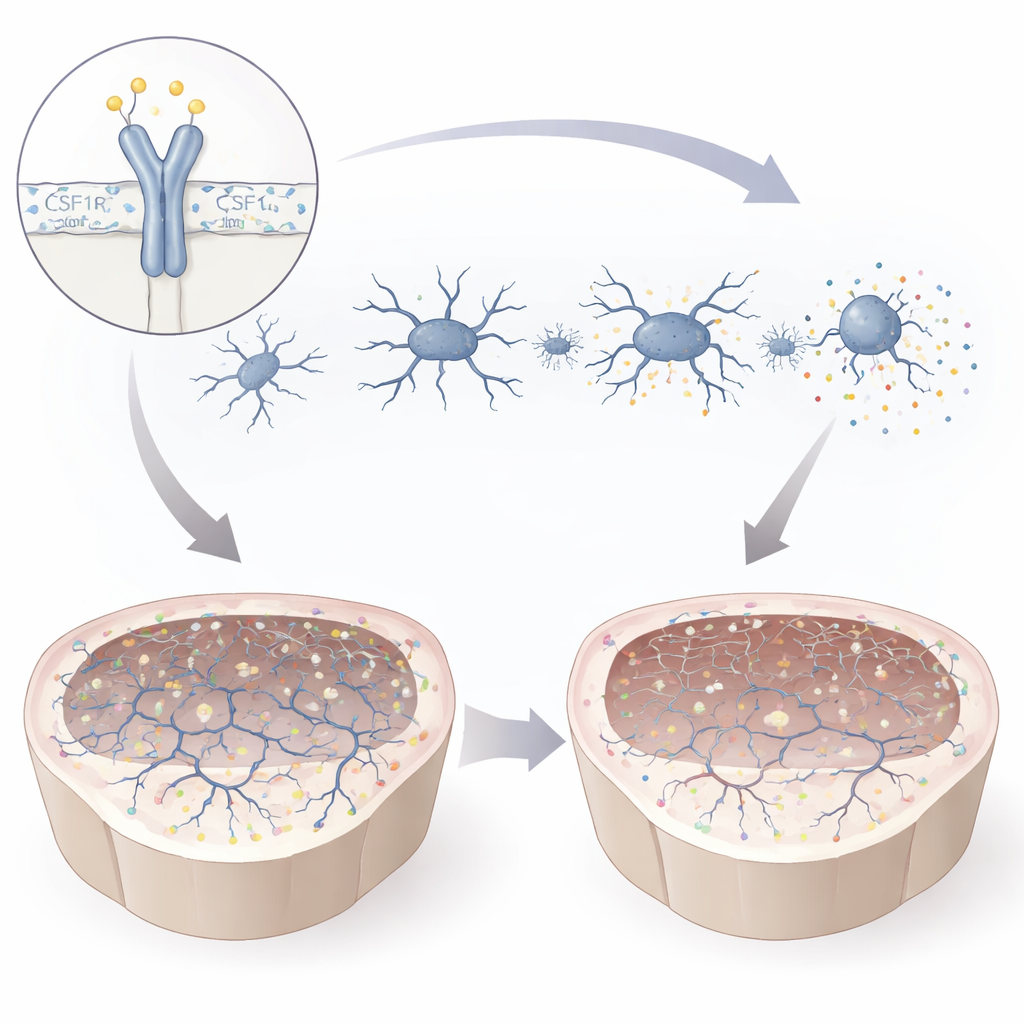
The mutation weakened the CSF1R protein and altered its chemical activation at a crucial site, leaving microglia partially short of its signal. In culture, mutant microglia became smaller and less branched, a shape associated with an activated, stressed state. They pumped out inflammatory molecules and migrated poorly toward signals that would normally draw them to injured areas. At the same time, they were more eager to engulf surrounding material, including myelin and bead-like particles that mimic cellular debris. Gene activity profiling revealed a shift away from programs that support nerve cell growth and synapses, and toward pathways linked to immune activation. Together, these changes paint a picture of microglia that are both overreactive and functionally compromised—poorly equipped to support healthy brain circuits.
Stalled Neurons and Weakened Signals in Mini-Brains
The mutation also disturbed the development of nerve cells themselves. Early precursor cells divided more than usual, but fewer matured into fully developed neurons with long, complex branches. In cerebral organoids grown from mutant cells, markers of mature neurons were reduced, and electrical recordings showed that neurons were smaller, fired fewer electrical impulses, and exchanged weaker signals with their neighbors. Both excitatory and inhibitory communication between cells dropped sharply, indicating a broad decline in synaptic function. When mutant microglia were added to mutant organoids, levels of key synaptic proteins at the junctions between neurons fell even further, whereas healthy microglia boosted these same proteins in normal organoids. This suggests that flawed microglia and vulnerable neurons act together to undermine brain connectivity.
What This Means for Patients and Future Treatments
By tracing the effects of the T567M mutation from gene to cell to mini-brain, the study shows that even a subtle change in CSF1R outside its main control region can destabilize microglia, derail brain development, and weaken synapses, offering a plausible explanation for the memory and movement problems seen in patients. The work highlights CSF1R signaling as a central guardian of “neural homeostasis”—the balance that keeps brain cells communicating smoothly. Just as important, the patient-derived two- and three-dimensional models provide a powerful testing ground for future therapies, from drugs that modulate microglia to cell-replacement strategies, raising hope that personalized treatments for CSF1R-related disorders may eventually be within reach.
Citation: Chi, L., Tu, H., Li, Z. et al. CSF1R T567M mutation induces microglial dysfunction and synaptic impairment in patient iPSC-derived cerebral organoids of CSF1R-related disorder. Cell Death Discov. 12, 148 (2026). https://doi.org/10.1038/s41420-026-02995-2
Keywords: CSF1R-related disorder, microglia, cerebral organoids, synaptic dysfunction, neurodegeneration