Clear Sky Science · en
Ferroptosis of smooth muscle cells in vascular diseases: from basic principles to clinical translation
Why Dying Vessel Cells Matter for Heart and Brain Health
Heart attacks, strokes, aneurysms, and high blood pressure all share a common stage: our blood vessels. This review article explores a newly recognized way that the muscle cells in vessel walls can die, called ferroptosis, which is tightly linked to iron and fat damage. By piecing together dozens of recent studies, the authors show how this type of cell death can both weaken blood vessels and, in some cases, help rein in harmful cell overgrowth. Understanding this double-edged process could open new paths to prevent or treat a wide range of vascular diseases.
How Iron-Driven Cell Death Works
Ferroptosis is a form of controlled cell death that depends on iron and the breakdown of fats in cell membranes. When too much iron builds up inside a cell, it helps generate highly reactive molecules that attack the fatty components of the cell’s outer shell. If the cell’s own defense systems, such as antioxidant enzymes and protective small molecules, are overwhelmed, the membrane becomes damaged and the cell collapses. Under the microscope, these cells show shrunken, distorted energy factories and ruptured outer membranes. Because blood vessel muscle cells are rich in certain fats and constantly handle mechanical and chemical stress, they are especially vulnerable when iron and oxidation get out of balance.
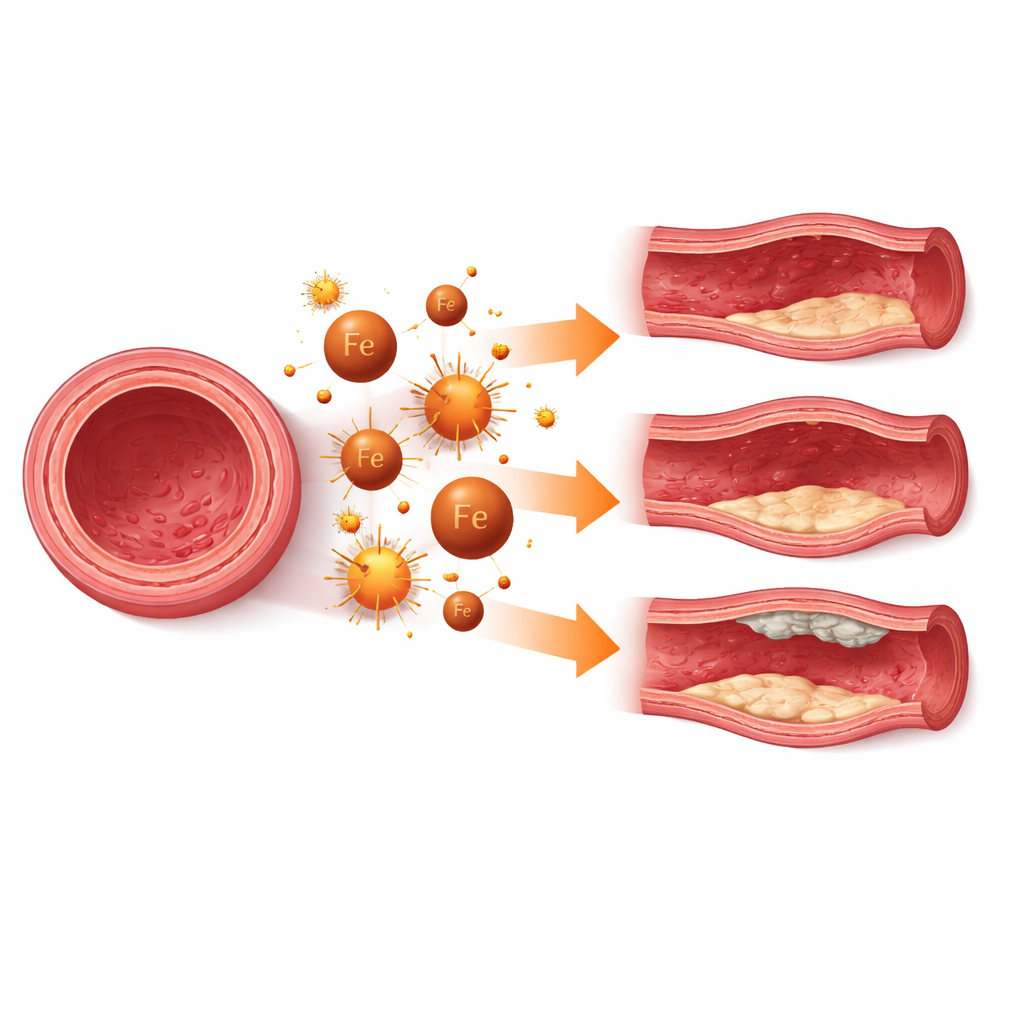
When Cell Death Weakens the Vessel Wall
In many diseases, ferroptosis in vascular smooth muscle cells acts as a slow wrecking ball on the vessel wall. In balloon-like outpouchings of major arteries, known as aneurysms, loss of these muscle cells makes the wall thinner and easier to tear. Studies in cells, animals, and human tissue all show signs of iron build-up, fat damage, and ferroptosis markers in aneurysm regions. Blocking ferroptosis with experimental drugs or by boosting natural defenses can slow aneurysm growth and preserve the normal, contractile state of these cells. Similar patterns appear in intracranial aneurysms, where protecting brain vessel muscle cells from ferroptosis may help maintain the strength of delicate arteries supplying the brain.
Iron, Aging Plaques, and Hardening Arteries
Ferroptosis also shapes how arteries age and clog. In atherosclerosis, the disease behind most heart attacks and strokes, unstable plaques often contain dying or dead smooth muscle cells, damaged fats, and heavy inflammatory activity. The review highlights work showing that ferroptosis in these cells can thin the fibrous cap that stabilizes a plaque, making it more likely to rupture. In vascular calcification, common in chronic kidney disease and diabetes, iron-fueled fat damage pushes muscle cells toward a bone-like state and encourages calcium deposits in the vessel wall. In both settings, experimental treatments that reduce iron overload, restore antioxidant capacity, or directly inhibit ferroptosis can lessen calcification and strengthen plaques in animal models.
When Triggering Cell Death Is Helpful
Strikingly, the same process can be beneficial when smooth muscle cells grow too much. In conditions like neointimal hyperplasia after stent placement, or pulmonary arterial hypertension in the lungs, these cells shift into a highly proliferative, synthetic state that thickens vessel walls and narrows the passage for blood. Here, the problem is not cell loss but cell excess. The review describes studies where gently promoting ferroptosis in overactive muscle cells, or restoring their sensitivity to it, can curb abnormal growth and limit the buildup of scar-like tissue. Conversely, when ferroptosis is overly suppressed in these diseases, vessels remodel in ways that worsen blood flow and drive up pressure.
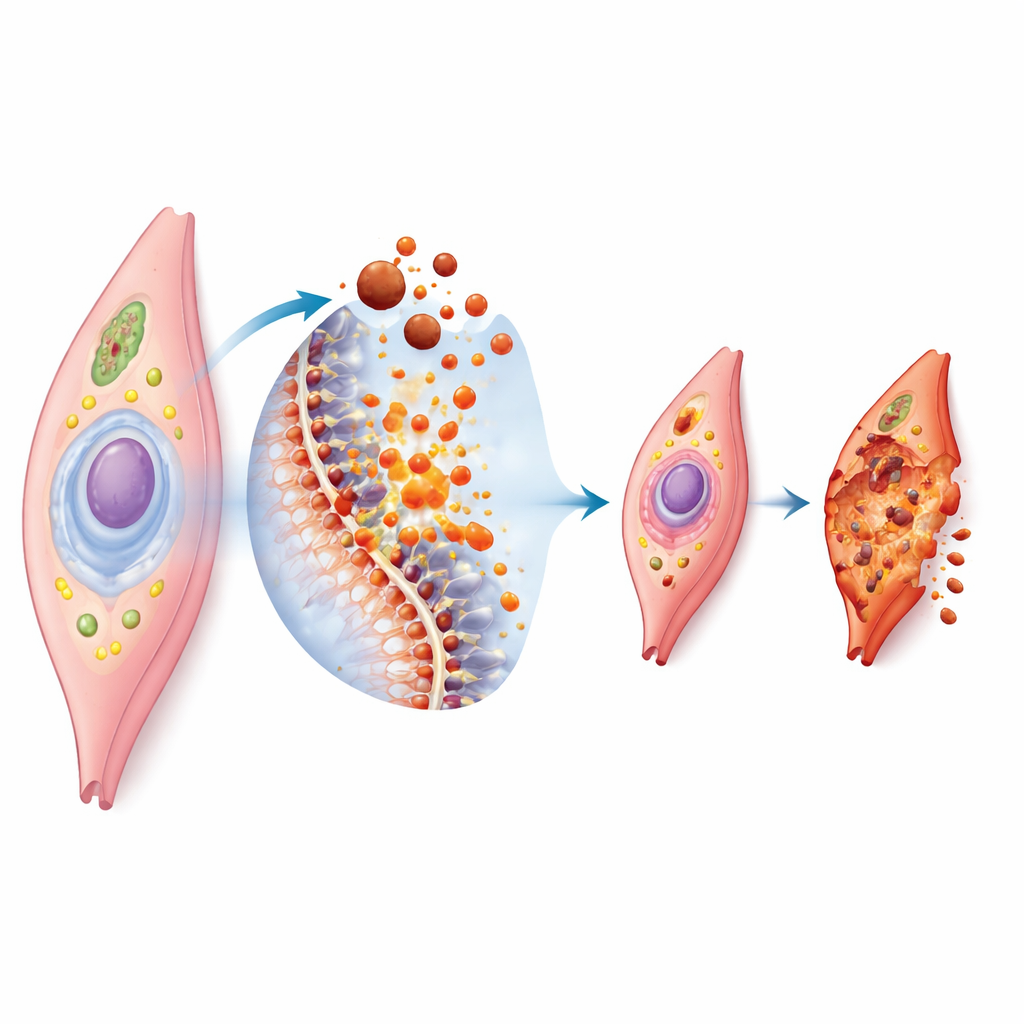
Layers of Control and Emerging Treatments
The article emphasizes that ferroptosis in vessel muscle cells is governed by a dense control network. Chemical tags on RNA, switches in gene activity, fine-tuning of proteins after they are made, and signals from neighboring immune and stem cells all push the balance toward or away from ferroptosis. These layers converge on three core functions: how much iron enters and is stored, how easily membrane fats are oxidized, and how strong the antioxidant defenses remain. Many experimental therapies now target these levers, from small molecules and natural compounds to RNA-based tools, engineered nanoparticles, and exosomes that carry protective cargo directly to the vessel wall.
What This Means for Patients
Overall, the review concludes that ferroptosis is a central, but context-dependent, driver of vascular disease. When it runs unchecked, it destroys the smooth muscle cells that give vessels their strength and flexibility, contributing to aneurysms, plaque instability, and calcification. When it is too strongly blocked, it enables runaway cell growth and thickening of vessel walls, as in certain forms of high blood pressure and post-surgical scarring. The challenge for future medicine is to learn when to dampen ferroptosis and when to harness it, and to do so in a way that targets the right cell types at the right disease stage. If that balance can be achieved, therapies aimed at this iron-linked cell death pathway could become powerful tools to preserve blood vessel health and reduce the burden of heart, brain, and lung vascular disorders.
Citation: Yang, Y., Nawabi, A.Q., Yao, Y. et al. Ferroptosis of smooth muscle cells in vascular diseases: from basic principles to clinical translation. Cell Death Discov. 12, 140 (2026). https://doi.org/10.1038/s41420-026-02950-1
Keywords: ferroptosis, vascular smooth muscle cells, aneurysm, atherosclerosis, vascular calcification