Clear Sky Science · en
KAT5-mediated acetylation enhances the deubiquitination of HASPIN by OTUB2 and promotes breast cancer progression
Why this cancer story matters
Breast cancer remains a leading cause of death for women worldwide, largely because aggressive tumors spread to distant organs. This study digs into the tiny molecular "switches" that help breast cancer cells grow and travel through the body. By uncovering a new way cancer cells keep a growth-driving protein alive and active, the researchers point to fresh opportunities for more precise treatments that could one day slow or stop deadly disease spread.
A protein that pushes cancer cells forward
At the center of this work is a protein called HASPIN, a type of enzyme that helps cells divide by guiding how chromosomes are handled during cell division. The team found that HASPIN levels are much higher in breast cancer tissue than in normal breast tissue, and that patients whose tumors contain more HASPIN tend to have worse overall survival. When the researchers dialed down HASPIN in breast cancer cell lines, the cells grew more slowly, formed fewer colonies, and were less able to move and invade through laboratory membranes. When they boosted HASPIN, the opposite happened: cells became more aggressive. These experiments show that HASPIN behaves like an “oncogene” in breast cancer—one of the internal engines that fuels tumor growth and spread.
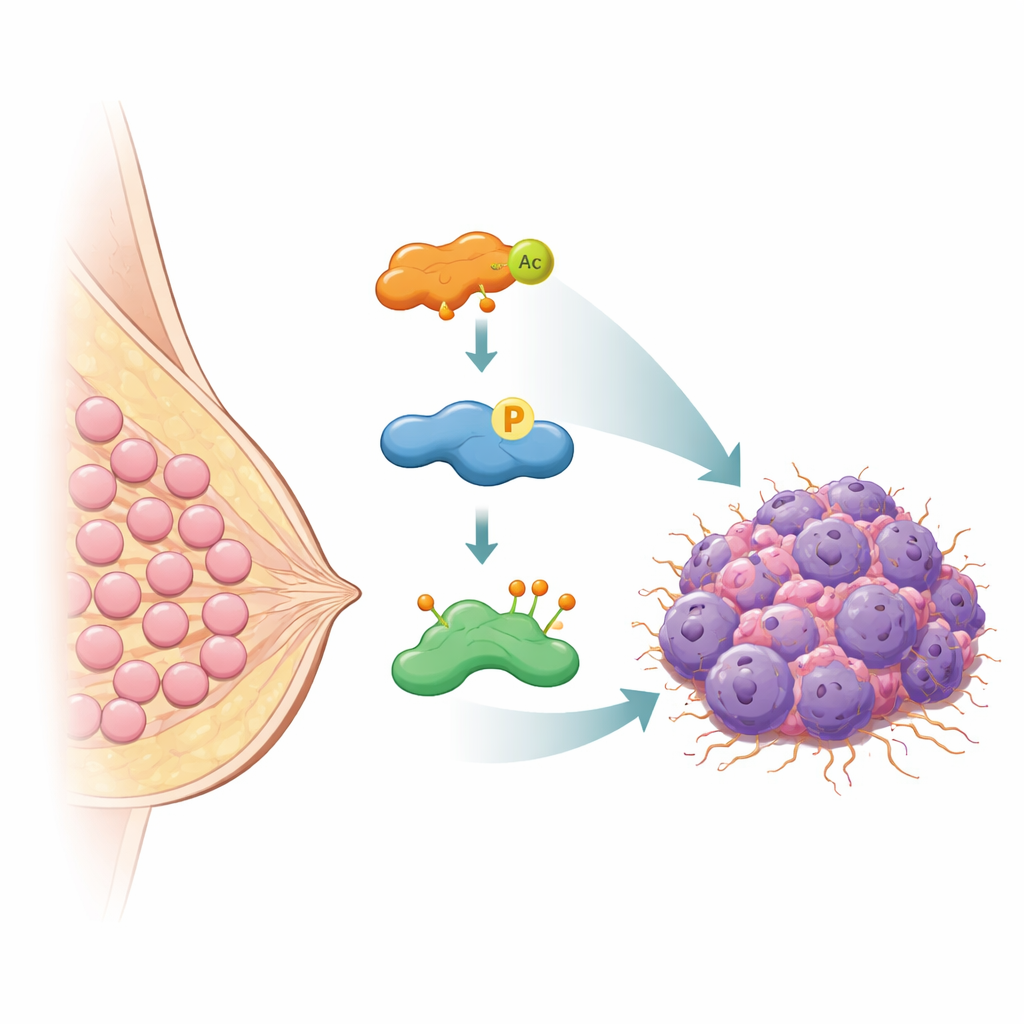
The molecular bodyguard that saves HASPIN from destruction
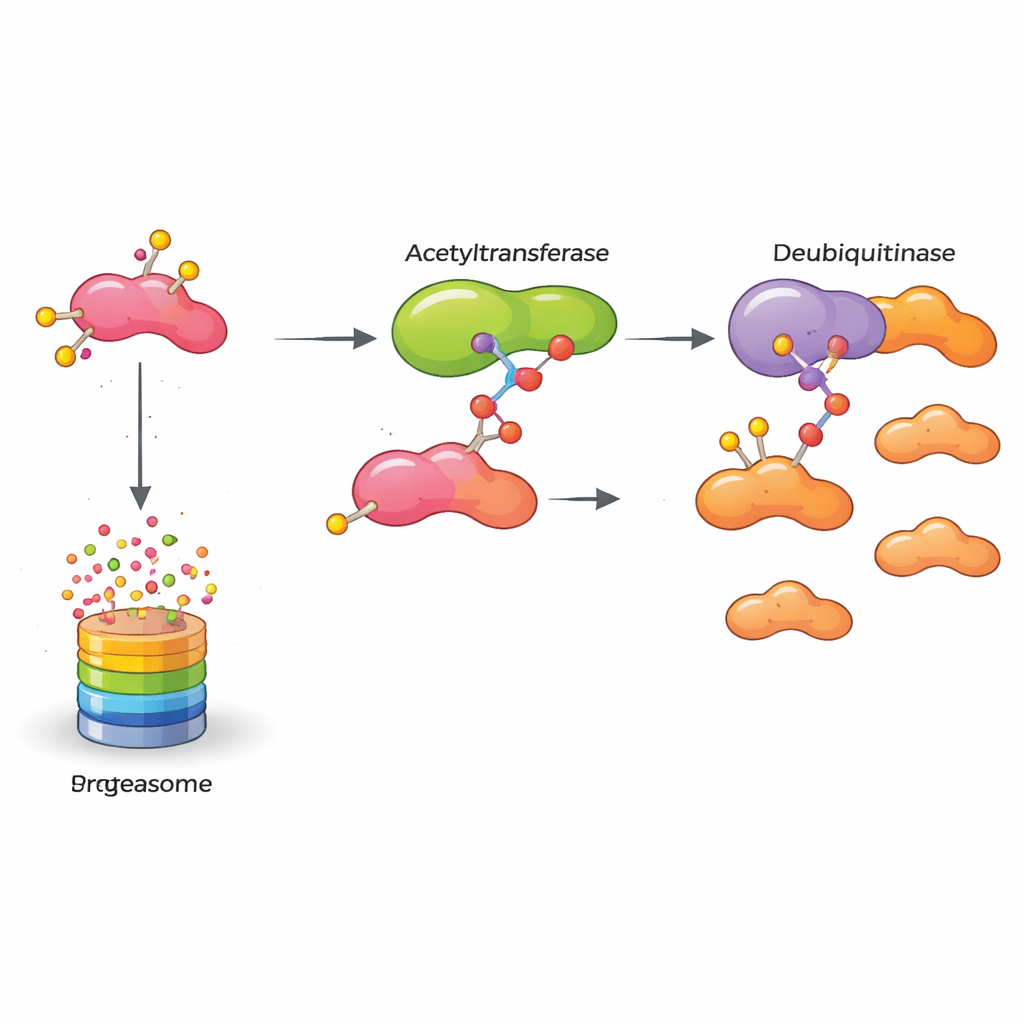
Cells normally prevent dangerous protein build-up by marking unneeded proteins for destruction with small tags called ubiquitin, which send them to the cell’s waste-disposal machinery. The authors discovered that a protein called OTUB2 acts as a molecular bodyguard for HASPIN. OTUB2 belongs to a family of enzymes that remove these ubiquitin tags. Using multiple biochemical methods, the team showed that OTUB2 directly binds to HASPIN inside breast cancer cells. When OTUB2 was removed, HASPIN became heavily tagged with a particular form of ubiquitin that signals for protein breakdown and its levels dropped. When OTUB2 was increased, these tags were stripped away, HASPIN was spared from destruction, and its levels rose. In mouse models, tumors with more OTUB2 grew faster and spread more readily to the lungs, but these effects depended on HASPIN, underscoring how closely their fates are tied.
A chemical tweak that tightens the partnership
The story does not end with OTUB2. The researchers found that HASPIN also carries another common chemical mark called an acetyl group at a specific position. This acetyl mark is added by an enzyme named KAT5, known for modifying proteins that control DNA and gene activity. When KAT5 added an acetyl group to HASPIN at one key spot, HASPIN’s grip on OTUB2 became stronger. As a result, OTUB2 could more effectively remove the degradation tags from HASPIN, boosting HASPIN’s stability even further. Mutating this single site to prevent acetylation weakened the interaction with OTUB2 and allowed more ubiquitin tags to accumulate, causing HASPIN to break down faster. In essence, KAT5 turns a dial that controls how tightly HASPIN and OTUB2 cooperate.
From cell culture dishes to patient tumors
To connect these findings to real-world disease, the team examined breast cancer samples from patients. Both HASPIN and OTUB2 proteins were consistently higher in tumor tissue than in nearby normal tissue. Across dozens of cases, tumors that had more OTUB2 also tended to have more HASPIN, mirroring the molecular partnership seen in the lab. Importantly, patients whose tumors showed high levels of either protein had poorer survival over time. This clinical link supports the idea that the KAT5–HASPIN–OTUB2 chain is not a laboratory curiosity but a working engine inside human cancers that helps them grow and spread.
What this means for future treatments
Put together, the study reveals a molecular relay that keeps a cancer-promoting protein switched on: KAT5 chemically modifies HASPIN, which then latches more tightly onto OTUB2, which in turn shields HASPIN from being destroyed. This stabilized HASPIN drives breast cancer cells to multiply and invade. For non-specialists, the key takeaway is that blocking any link in this chain—KAT5’s acetyl mark, OTUB2’s protective action, or HASPIN itself—could weaken the tumor’s internal machinery. While turning these insights into drugs will take time, the work opens new paths to therapies designed to selectively disarm aggressive breast cancers by letting their own quality-control systems finally take out the molecular trash.
Citation: Guo, J., Kang, K., Wang, S. et al. KAT5-mediated acetylation enhances the deubiquitination of HASPIN by OTUB2 and promotes breast cancer progression. Cell Death Dis 17, 411 (2026). https://doi.org/10.1038/s41419-026-08658-5
Keywords: breast cancer, HASPIN, OTUB2, protein degradation, targeted therapy