Clear Sky Science · en
Dimeric PKM2 in chondrocytes impairs mitochondrial homeostasis in osteoarthritis
Why worn-out joints matter
Many people live with the pain and stiffness of osteoarthritis, a common joint disease that slowly wears away the smooth cartilage cushioning our bones. Once this cartilage is badly damaged, the main option is joint replacement surgery. This study explores a hidden energy switch inside cartilage cells that appears to speed up or slow down this damage. By understanding and controlling this switch, the work points toward new treatments that might protect joints and delay or even prevent the need for surgery.
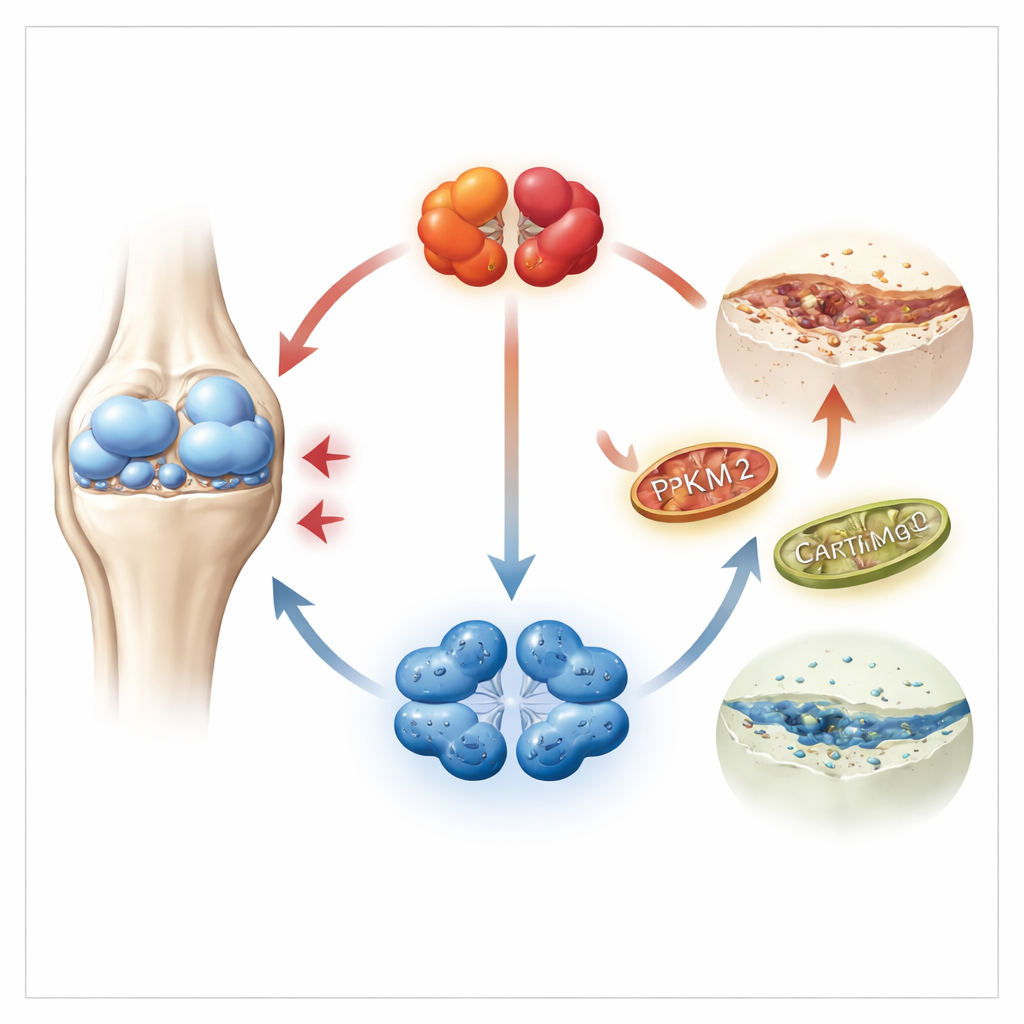
A closer look inside aching cartilage
Cartilage is built and maintained by chondrocytes, specialized cells that live in a low-oxygen environment and mostly rely on sugar-burning for energy. The researchers focused on a key enzyme in this process called PKM2, which can exist in two shapes: a tightly packed four-part form and a looser two-part form. Using large genetic datasets from human cartilage, as well as samples from patients and mice, they found that PKM2 levels were strongly increased in osteoarthritic cartilage, while its sister form PKM1 was barely detectable. Importantly, diseased cartilage showed a shift toward the two-part (dimer) version of PKM2, which is better known for controlling cell behavior than for making energy.
Turning a destructive cycle into a protective one
The team then asked what happens if PKM2 is reduced or kept in its four-part form. In cartilage cells grown in the lab, lowering PKM2 levels, or using a small molecule called TEPP-46 that locks PKM2 into its four-part state, tilted the balance toward building cartilage rather than breaking it down. Cells made more of the structural proteins that give cartilage its strength and fewer of the enzymes that chew up the tissue. In a mouse model where osteoarthritis is triggered by an unstable knee, injections that reduced PKM2 or stabilized its four-part form preserved cartilage structure, lowered damage scores, and improved walking patterns and pain-related responses.
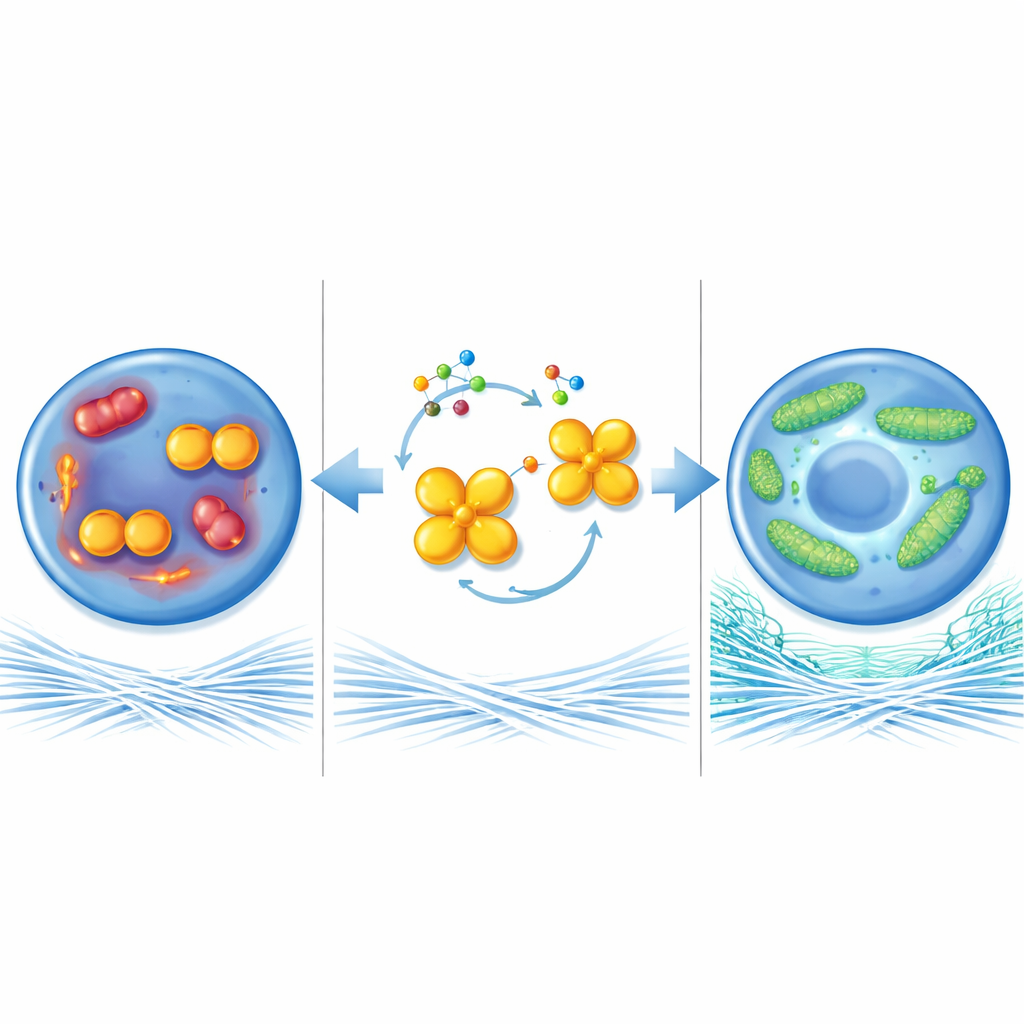
Protecting the cell’s power stations
To understand how this enzyme influences cartilage health, the researchers looked at mitochondria, the tiny power stations inside cells. In osteoarthritis-like conditions, mitochondria in chondrocytes became small and fragmented, produced more harmful reactive molecules, and generated less energy. When PKM2 was removed or forced into its four-part shape, mitochondria instead formed long, connected networks, burned fuel more efficiently, and made more energy-rich molecules. This mitochondrial “fusion” depended largely on another protein, MFN1, which helps link mitochondria together. MFN1 levels were low in diseased cartilage but rose when PKM2 dimer levels fell. Blocking MFN1 in the joints of PKM2-deficient mice erased the protective effects and allowed cartilage damage to progress, showing that MFN1 is a key player in this pathway.
Breaking the harmful signal chain
The study further uncovered a signaling chain linking PKM2 to mitochondrial changes. The loose, two-part PKM2 physically interacted with ERK, a well-known signaling protein that responds to stress and inflammation. Under osteoarthritis-like stimulation, this interaction became stronger and ERK became more active, which in turn suppressed MFN1 and pushed mitochondria toward a fragmented, unhealthy state. Reducing PKM2 dimerization or locking PKM2 into tetramers weakened this PKM2–ERK interaction, lessened ERK activity, and restored MFN1 levels. Interestingly, a related fusion protein, MFN2, was influenced differently, suggesting that MFN1 has a unique, non-redundant role in keeping cartilage cells and their mitochondria in balance.
What this means for future joint care
Put simply, the work shows that when PKM2 accumulates in its two-part form inside cartilage cells, it disturbs the cell’s power systems, weakens the cartilage-building machinery, and accelerates osteoarthritis. Shifting PKM2 toward its four-part form, or otherwise reducing the two-part version, helps mitochondria stay healthy and encourages cartilage repair. In mice, this strategy not only preserved joint tissue but also eased pain-related behavior. While more research is needed before such approaches reach the clinic, targeting PKM2 dimerization offers a promising new way to slow or prevent joint wear, potentially giving patients more years of comfortable movement before surgery becomes necessary.
Citation: Liu, B., Liang, Y., Wang, C. et al. Dimeric PKM2 in chondrocytes impairs mitochondrial homeostasis in osteoarthritis. Cell Death Dis 17, 370 (2026). https://doi.org/10.1038/s41419-026-08621-4
Keywords: osteoarthritis, cartilage, mitochondria, PKM2, mitofusin 1