Clear Sky Science · en
DUSP12 promotes cell cycle progression and protects cells from ZNF622 mediated apoptosis
Why cell survival choices matter
Every second, our cells quietly decide whether to keep growing or to self-destruct. When this balance is disturbed, cancers can arise or life-saving cells can be lost during injury. This study explores two little-known cellular “switches,” called DUSP12 and ZNF622, that help decide whether a stressed cell repairs itself and divides, or instead activates a built-in death program. Understanding how these switches work could open new doors both for killing cancer cells and for protecting healthy tissues from damage.
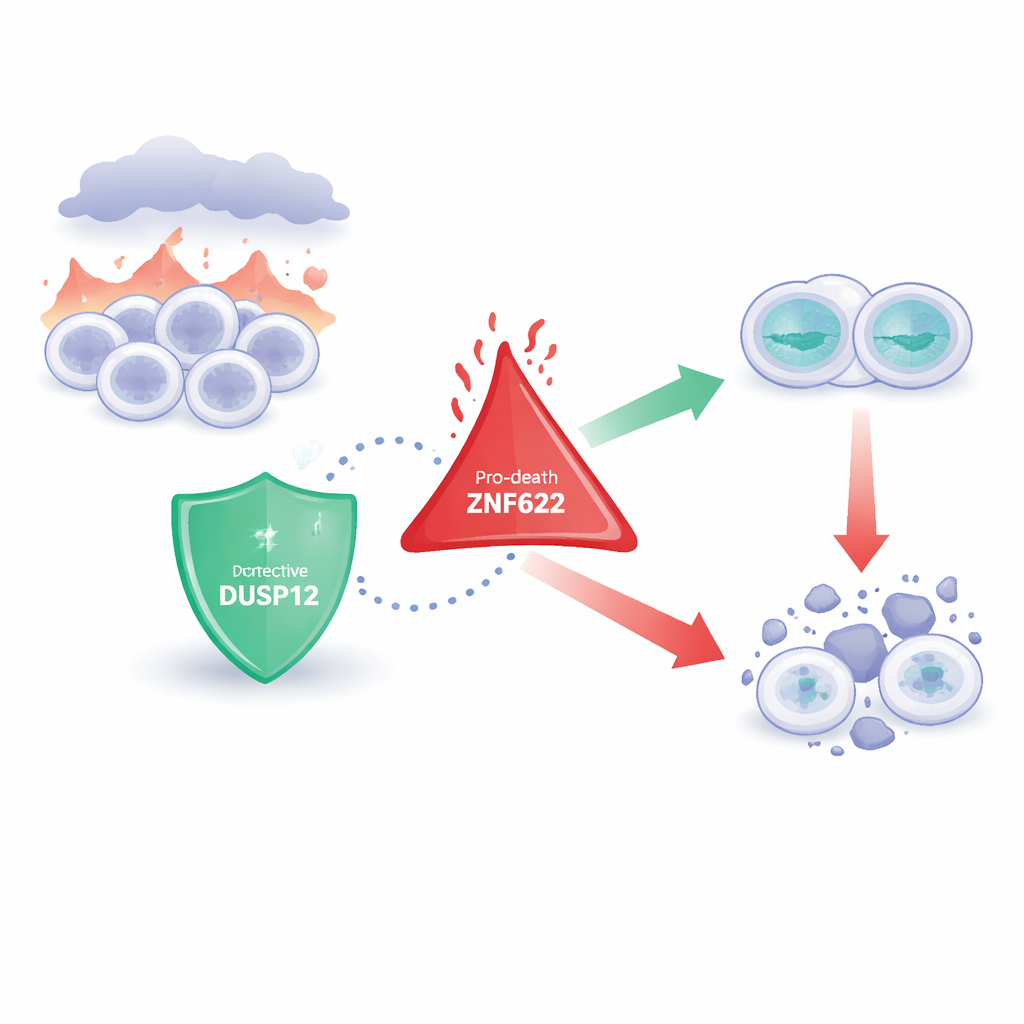
Two opposing cellular guardians
The researchers focus on a family of enzymes that remove tiny chemical tags from proteins, subtly changing how those proteins behave. DUSP12 is one such enzyme, long suspected to influence cell growth and survival but poorly understood. ZNF622, in contrast, is a protein with “finger-like” structures that help it interact with many partners and has been linked to cell growth, stress responses, and especially to triggering cell death. Earlier work hinted that both molecules were important in cancer, but how they might talk to each other—and what that meant for a cell’s fate—was unknown.
Finding a hidden partnership
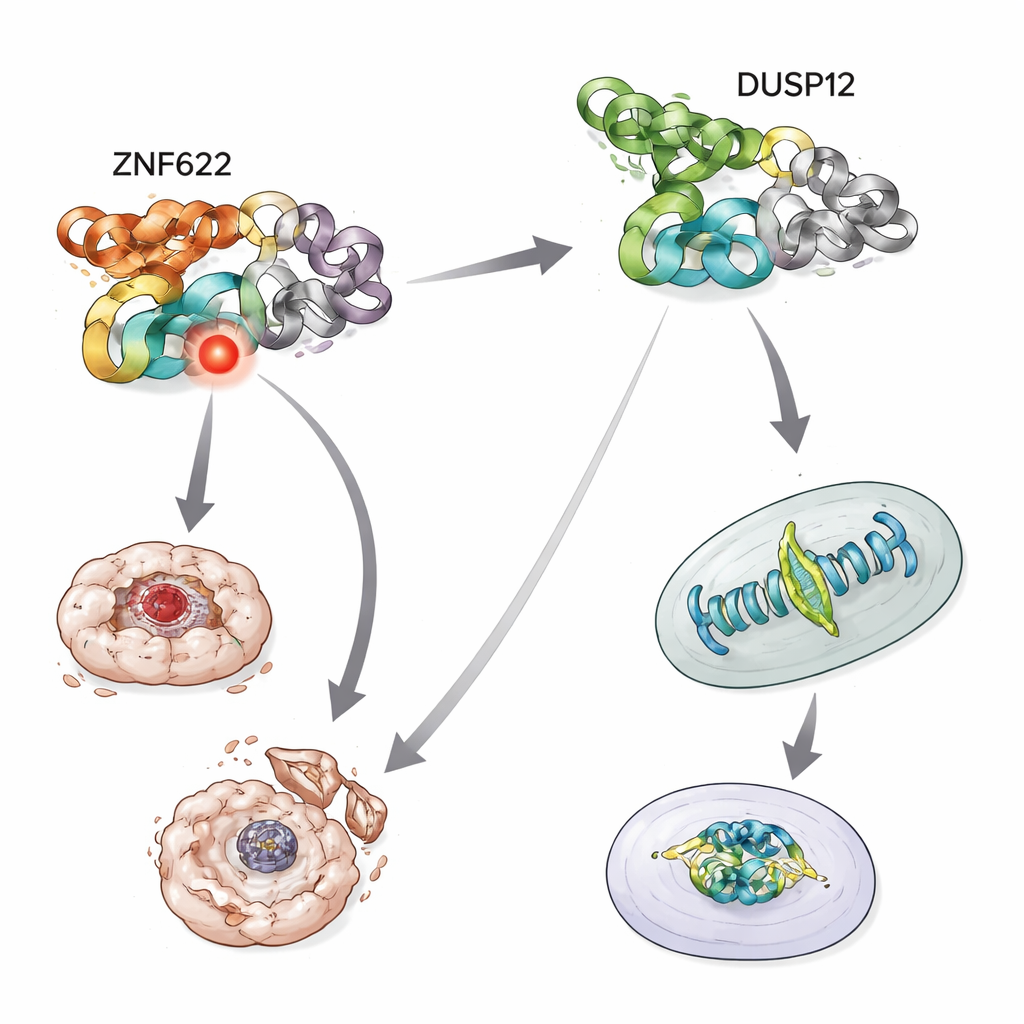
To uncover DUSP12’s partners, the team used biochemical fishing tricks in human cells, tagging DUSP12 so that any proteins sticking to it could be pulled out and identified by mass spectrometry, a technique that weighs and catalogs protein fragments. In cells paused during division, ZNF622 repeatedly appeared alongside DUSP12. Further tests confirmed that the two proteins form a complex both inside cells and in simplified test-tube systems. Mapping DUSP12’s structure showed that this partnership depends on a special metal-grabbing region at one end of DUSP12—the zinc-binding domain—which acts as a docking site for ZNF622.
Keeping cell division on track
Cell division is a delicate choreography in which chromosomes must line up and separate cleanly. When the scientists reduced DUSP12 levels in human cancer cells, more cells stalled during division and showed misaligned chromosomes, and the time needed to complete division increased. Cells also piled up in the late stages of the cell cycle, suggesting they were struggling to progress. When ZNF622 was artificially boosted, cells again developed severe division problems, including tangled or multipolar division machinery. Intriguingly, a specific site on ZNF622 could be either chemically tagged or untagged, and DUSP12 activity shifted ZNF622 toward the untagged form. Only the normal version of ZNF622, whose tag could change back and forth, produced strong division defects; fixed “always on” or “always off” mimics did not. This indicates that DUSP12’s fine-tuning of this single site helps keep cell division orderly.
Balancing life and death under stress
The team then asked what happens when cells are exposed to toxic stresses, such as chemotherapy drugs that usually push cancer cells toward self-destruction. When DUSP12 was depleted, cells became more prone to die in response to several anti-cancer agents, showing higher activity of enzymes that execute cell death. In contrast, reducing ZNF622 made cells more resistant to dying under the same treatments. Flipping the experiment, overproducing DUSP12 shielded cells from these stresses, while overproducing ZNF622 made death more likely. When both were increased together, DUSP12 blunted ZNF622’s pro-death effect, and raising DUSP12 stepwise progressively dialed down cell death even in the presence of constant ZNF622. This level-dependent tug-of-war shows that the two proteins act as opposing knobs on the same survival circuit.
What this means for cancer and beyond
These findings paint DUSP12 as a cellular bodyguard that binds ZNF622 and restrains its tendency to derail cell division and trigger death, especially under drug-induced stress. In cancers where DUSP12 is abnormally high, this protection may help tumor cells survive chemotherapy, making DUSP12 an attractive target for drugs designed to strip away that shield and let cancer cells die. Conversely, in situations where preserving cells is crucial—such as protecting the liver during surgery or slowing nerve cell loss in degenerative diseases—boosting DUSP12 or dampening ZNF622 might help tissues withstand damaging conditions. By clarifying how these two proteins converse at a key decision point between life and death, the study offers a new handle for tuning cell fate in both cancer therapy and tissue protection.
Citation: Abdusamad, M., Guo, X., Ramirez, I. et al. DUSP12 promotes cell cycle progression and protects cells from ZNF622 mediated apoptosis. Cell Death Dis 17, 315 (2026). https://doi.org/10.1038/s41419-026-08618-z
Keywords: cell cycle, apoptosis, cancer therapy resistance, protein phosphatase, stress response