Clear Sky Science · en
SSTNIV, a syndecan-1-targeting peptide chimera, reverses immune suppression and inhibits myeloma progression
Why this research matters to patients
Multiple myeloma is a cancer of bone marrow cells that often comes back after treatment, leaving patients with fewer and fewer options. This study introduces a new lab-designed peptide, called SSTNIV, that attacks the cancer on two fronts at once: it harms myeloma cells directly and also loosens the grip of an unhealthy immune environment that protects the tumor. The work, done in advanced mouse models and human samples, suggests a different way to treat drug resistant disease by targeting both the tumor and its protective neighborhood.
A sticky molecule that helps myeloma thrive
Myeloma cells sit in bone marrow surrounded by support cells and immune cells that together form a tumor friendly niche. A surface molecule called syndecan-1 is found at very high levels on myeloma cells and is also shed into the surrounding space. Both forms act like a scaffold that brings together growth receptors and gripping proteins on cell surfaces, helping cancer cells survive, spread, and dodge immune attack. High levels of shed syndecan-1 in patient blood are linked with worse outcomes, making it an attractive target for therapy.
Building a smarter peptide drug
The researchers had previously created two small peptides that each blocked one syndecan-1 driven pathway: one restored cell death signals inside myeloma cells, and the other interfered with cancer cell movement while freeing up immune cells to move. In this work they trimmed each peptide down to its shortest active core and fused them into a single chimera called SSTNIV with a long lifetime in the bloodstream. In cell culture tests, SSTNIV triggered strong apoptosis in myeloma cells and in tumor associated macrophages, while leaving beneficial T cells mostly unharmed. At the same time, it slowed the migration of myeloma cells, suppressive macrophages, and regulatory T cells, but boosted the movement of cancer killing T cells.
Testing the peptide in an aggressive myeloma model
To see if these effects could matter in a whole organism, the team used a mouse model that closely mimics advanced, treatment resistant myeloma in people. Mice received either standard bortezomib chemotherapy, one of the original single function peptides, the new SSTNIV peptide, or combinations of peptide plus bortezomib. Blood protein tests and bone marrow analysis showed that SSTNIV on its own cut tumor burden as effectively as the best combination of the older peptide with chemotherapy. Adding bortezomib to SSTNIV reduced detectable myeloma cells in bone marrow to nearly normal levels and greatly limited spread to organs like the spleen and liver. Mice treated with SSTNIV lived longer and showed better preserved bone marrow structure and blood counts, suggesting that normal blood formation was recovering.
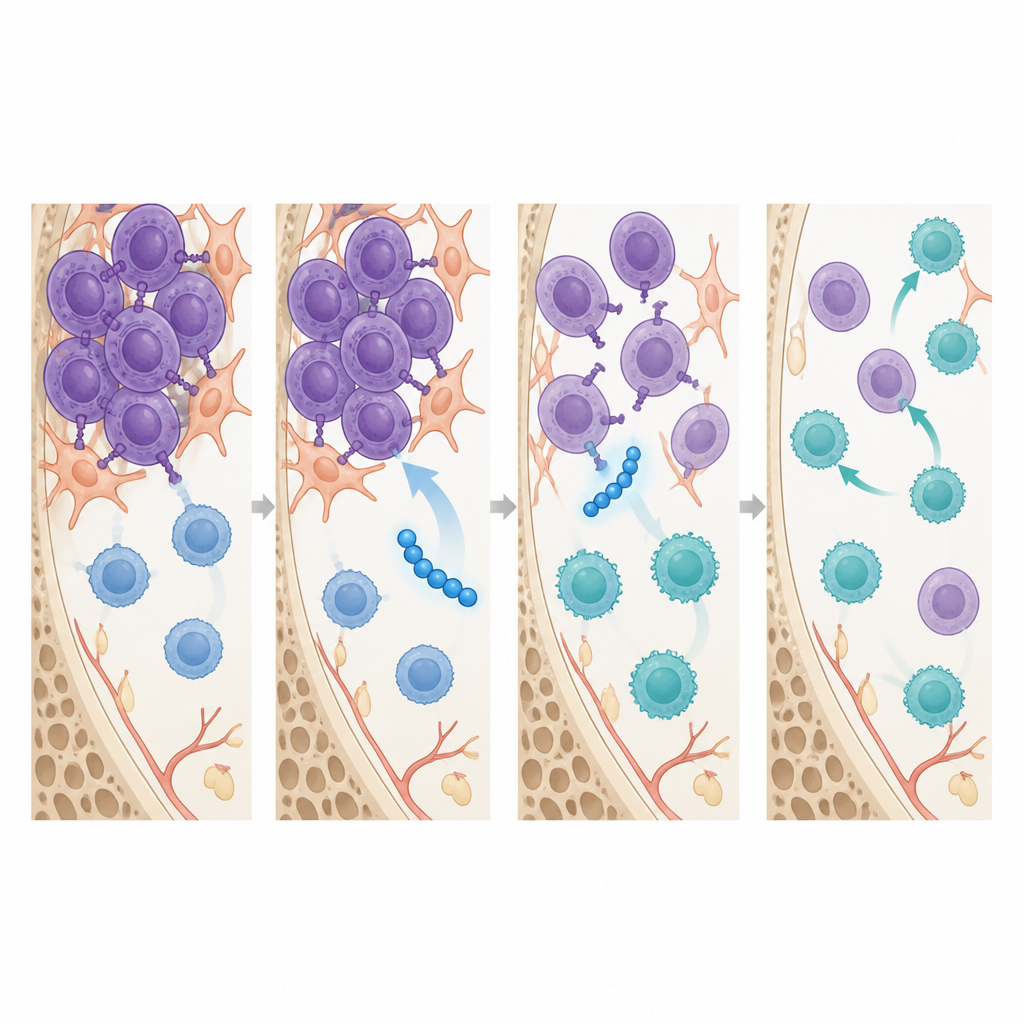
Reawakening the immune response
Beyond shrinking tumors, SSTNIV reshaped the immune landscape inside bone marrow. Within a week of treatment, before large changes in tumor size appeared, mice given SSTNIV had more cytotoxic CD8 T cells in their marrow and far fewer regulatory T cells and tumor associated macrophages, the two cell types that usually dampen immune attack and help tumors resist drugs. Imaging studies showed that SSTNIV physically disrupted the receptor clusters assembled by syndecan-1 on myeloma cells, reactivating a stress sensor that triggers cell death and shutting down signals that drive cell movement. Similar syndecan-1 based receptor complexes were detected in human myeloma bone marrow samples but not in healthy donors, supporting the idea that these pathways are disease specific targets.
What this could mean for future myeloma treatment
For a lay reader, the takeaway is that this research explores a new type of precision peptide that attacks myeloma cells and their protective immune cocoon at the same time. In advanced mouse disease, SSTNIV reduced cancer cells in bone marrow, limited spread to other organs, restored healthier blood production, and shifted immune cells toward a more attack ready state, especially when combined with existing chemotherapy. While these findings are still preclinical, they suggest that drugs modeled on SSTNIV could one day help treat patients whose myeloma has stopped responding to current therapies by disabling a key support system the cancer relies on.
Citation: Jung, O., Beauvais, D.M., Ibaan, G.L. et al. SSTNIV, a syndecan-1-targeting peptide chimera, reverses immune suppression and inhibits myeloma progression. Sig Transduct Target Ther 11, 175 (2026). https://doi.org/10.1038/s41392-026-02709-1
Keywords: multiple myeloma, syndecan-1, tumor microenvironment, peptide therapy, immune cells