Clear Sky Science · en
Rapid CAR screening and circRNA-driven CAR-NK cells for persistent shed-resistant immunotherapy
Why this matters for future cancer treatments
Many people have heard of CAR-T cell therapies that reprogram the immune system to hunt down cancers. But these treatments have struggled against tough solid tumors like pancreatic cancer. This study shows a new way to engineer a different type of immune cell—natural killer (NK) cells—so they can keep working even when tumors use one of their favorite tricks to evade attack: shedding the very target the therapy is supposed to recognize. The work also introduces a new RNA technology that may make such cell therapies faster to design and easier to manufacture.
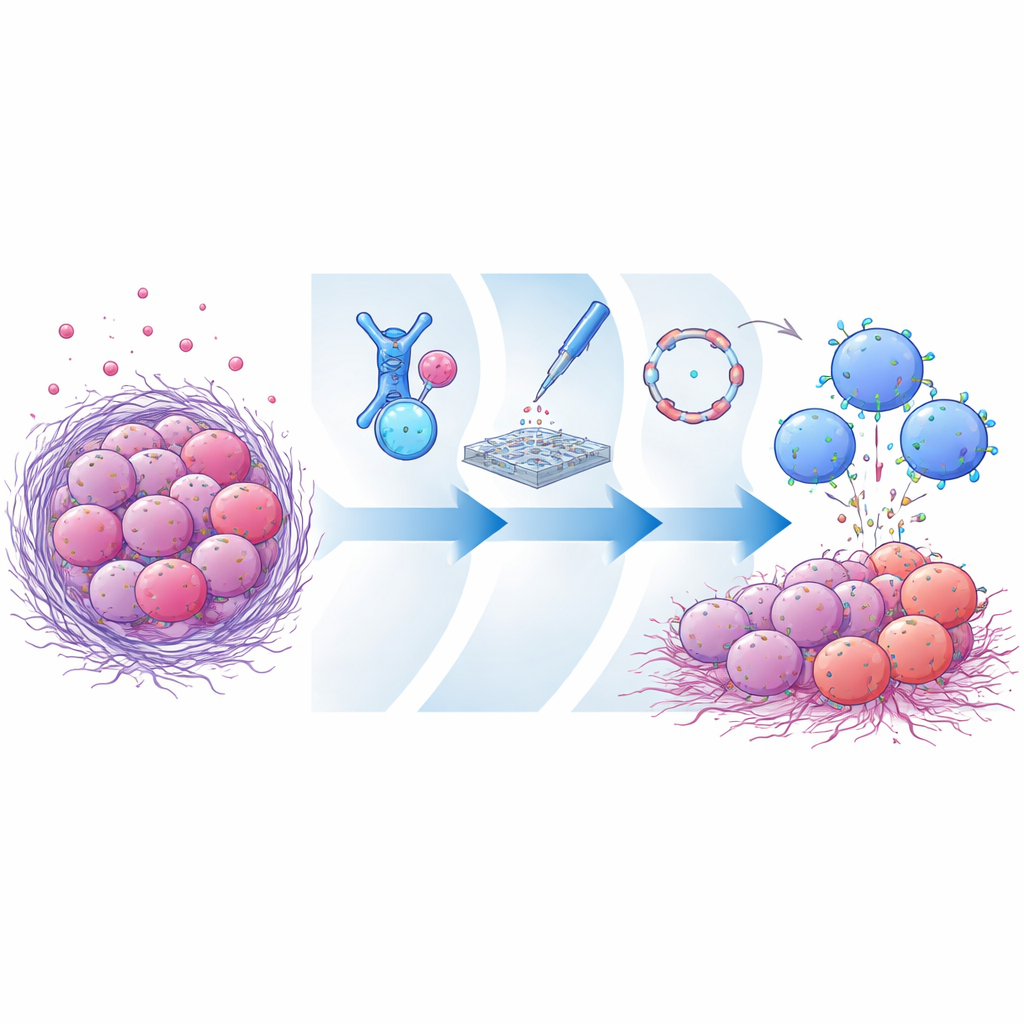
The tumor’s disappearing target trick
Many aggressive cancers carry high levels of a surface molecule called mesothelin. Because healthy tissues have much less of it, mesothelin has been a popular bullseye for experimental drugs and cell therapies. However, tumors can clip off the outer portion of mesothelin and release it into the bloodstream. These free-floating fragments act like decoys, soaking up therapeutic antibodies or CAR receptors before they ever reach the cancer cell surface. Earlier therapies mostly bound to distant parts of mesothelin that are easily shed, so they were particularly vulnerable to this decoy effect. The challenge is to find a binding site that stays on the cell surface after shedding while still allowing immune cells to form a tight, effective contact with the tumor.
Finding a better handle on the cancer cell
The researchers first created a huge library of antibody fragments that recognize mesothelin and displayed them on yeast cells. Through several rounds of selection, they enriched for candidates that bound strongly to the intact, membrane-tethered version of mesothelin but barely to its soluble shed form. One standout antibody fragment, named CLMS10, attached near the portion of mesothelin that remains anchored after cleavage—very close to the cell membrane. Computer modeling suggested that this position overlaps the cutting site itself, physically blocking the enzymes that snip mesothelin free or at least favoring the membrane-bound form. When CLMS10 was built into CAR designs and tested in primary human NK cells, these cells killed mesothelin-positive cancer cells efficiently and were largely unaffected by high levels of soluble mesothelin, unlike several benchmark CARs.
Rapid testing directly in human NK cells
Instead of using slow viral methods or surrogate cell lines, the team adopted an engineering-style, rapid testing platform directly in human donor NK cells. They encoded different CAR designs as linear messenger RNAs and delivered them by electroporation, allowing the NK cells to transiently express each candidate receptor. This made it possible to quickly compare how well various antibody fragments, signaling domains, and add-on immune boosters worked in realistic NK cells rather than in immortalized stand-ins. Through these side-by-side functional tests, they found that combining CLMS10 with an internal signaling module containing the OX40 and CD3ζ domains markedly increased tumor killing, NK cell degranulation, and metabolic fitness. Co-delivering the cytokine interleukin-21 further sharpened performance, helping NK cells sustain their activity in nutrient-poor, suppressive tumor-like conditions.
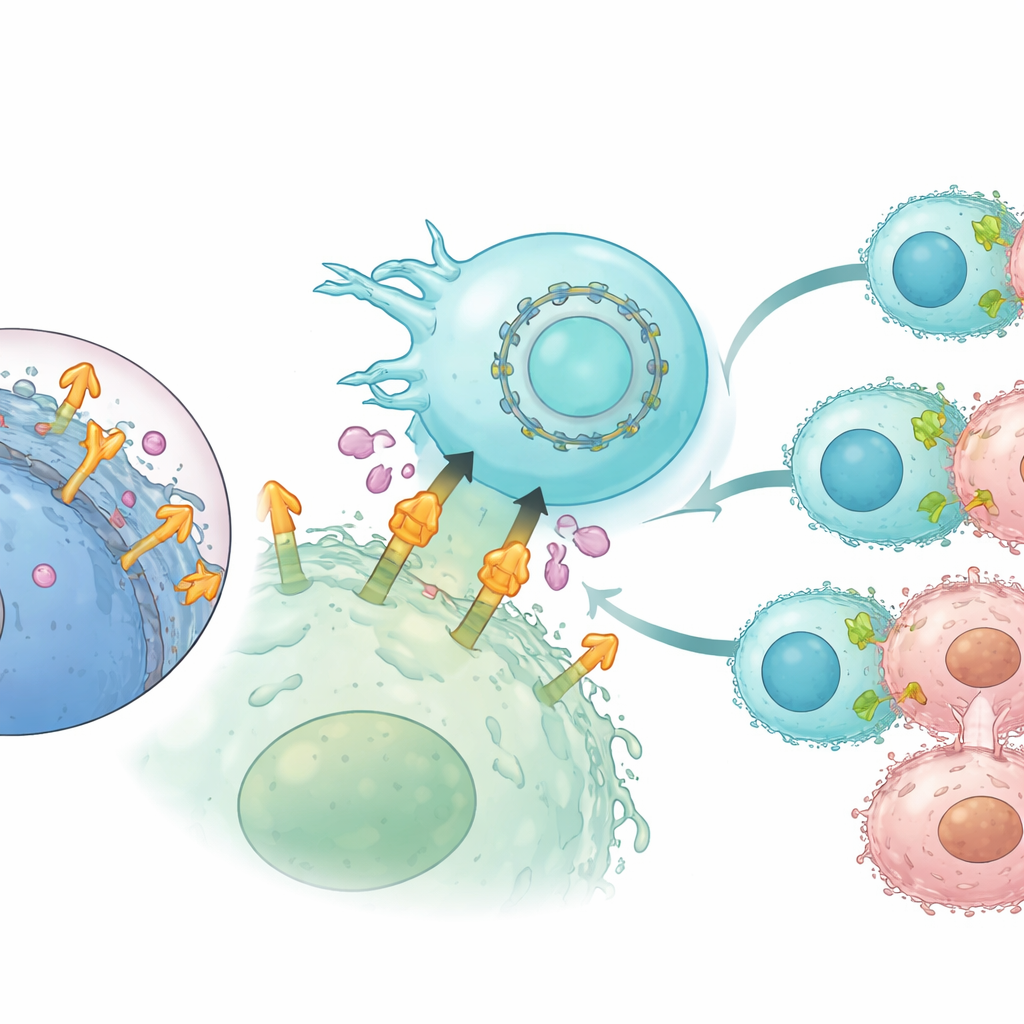
Making CAR expression last longer with circular RNA
Because regular mRNA is short-lived, its use in cell therapies can limit how long engineered receptors remain on the cell surface. To address this, the authors turned to circular RNA, a closed-loop RNA format that is naturally more resistant to degradation. They encoded the CLMS10-based CAR in circular RNA and introduced it into NK cells, again together with interleukin-21 RNA. Compared with linear mRNA, circular RNA drove more durable CAR expression over several days without harming cell viability. In repeated tumor-challenge experiments in the lab, NK cells programmed with circular RNA maintained stronger and longer-lasting cancer cell killing, continued to secrete key immune messengers, and remained effective even when exposed to high levels of shed mesothelin.
Proving shed resistance in a tough tumor setting
To mimic the real tumor environment, the team used cancer-associated fibroblasts—support cells that surround many solid tumors and are known to ramp up mesothelin shedding and dampen immune responses. In mixed cultures of pancreatic cancer cells and these fibroblasts, standard mesothelin-targeted CAR-NK cells lost much of their potency. In contrast, CLMS10-based circular RNA CAR-NK cells kept killing cancer cells despite the surge in shed antigen. When tested in mice bearing a fibroblast-rich, mesothelin-expressing pancreatic tumor model, these optimized CAR-NK cells shrank tumors as effectively as traditional lentivirus-engineered CAR-NK cells, while retaining advantages for rapid, nonviral manufacturing. The animals showed no obvious signs of toxicity from the temporary interleukin-21 boost.
What this means for patients
This research outlines a blueprint for building smarter cell therapies that can withstand one of solid tumors’ key escape strategies. By pairing an antibody that grabs a stable, membrane-close portion of mesothelin with a circular RNA platform that keeps the receptor on NK cells longer, the authors created a shed-resistant, persistent CAR-NK product that performs well in challenging pancreatic cancer models. While further work is needed before human trials, the approach could accelerate the design of off-the-shelf CAR-NK therapies for hard-to-treat cancers, offering a path toward more reliable and durable immune-based treatments.
Citation: Chung, JY., Hong, J., Yee, SM. et al. Rapid CAR screening and circRNA-driven CAR-NK cells for persistent shed-resistant immunotherapy. Sig Transduct Target Ther 11, 129 (2026). https://doi.org/10.1038/s41392-026-02623-6
Keywords: CAR-NK cell therapy, mesothelin shedding, circular RNA, pancreatic cancer, antibody engineering