Clear Sky Science · en
A novel homozygous splicing variant in FRA10AC1: further delineation of the phenotype
When Gene Editing Goes Awry
Most of us think of genes as static blueprints, but in reality our cells are constantly cutting and pasting genetic messages before turning them into working proteins. This article explores what happens when a tiny fault in this editing process affects a gene called FRA10AC1, leading to serious developmental problems in children. By following one young patient and comparing him with the few known cases worldwide, the researchers show how a subtle glitch in processing genetic information can ripple out to the brain, kidneys, eyes, and other organs.
The Cell’s Tiny Editors
Inside every cell, long stretches of raw genetic code must be trimmed and joined together before they can be used. This task is handled by a molecular machine known as the spliceosome, which snips out unused segments and stitches together the useful ones. The FRA10AC1 gene makes a helper protein that sits on the edge of this machine and fine-tunes how cutting and joining occur. Earlier studies found that children who inherit faulty copies of FRA10AC1 from both parents can develop a distinctive pattern of delayed development, unusual facial features, poor growth, and changes in the thick bundle of nerve fibers that connects the two halves of the brain, called the corpus callosum. However, only ten such patients had been described worldwide, leaving many questions about the full range of symptoms and how different types of faults in the gene shape the outcome.
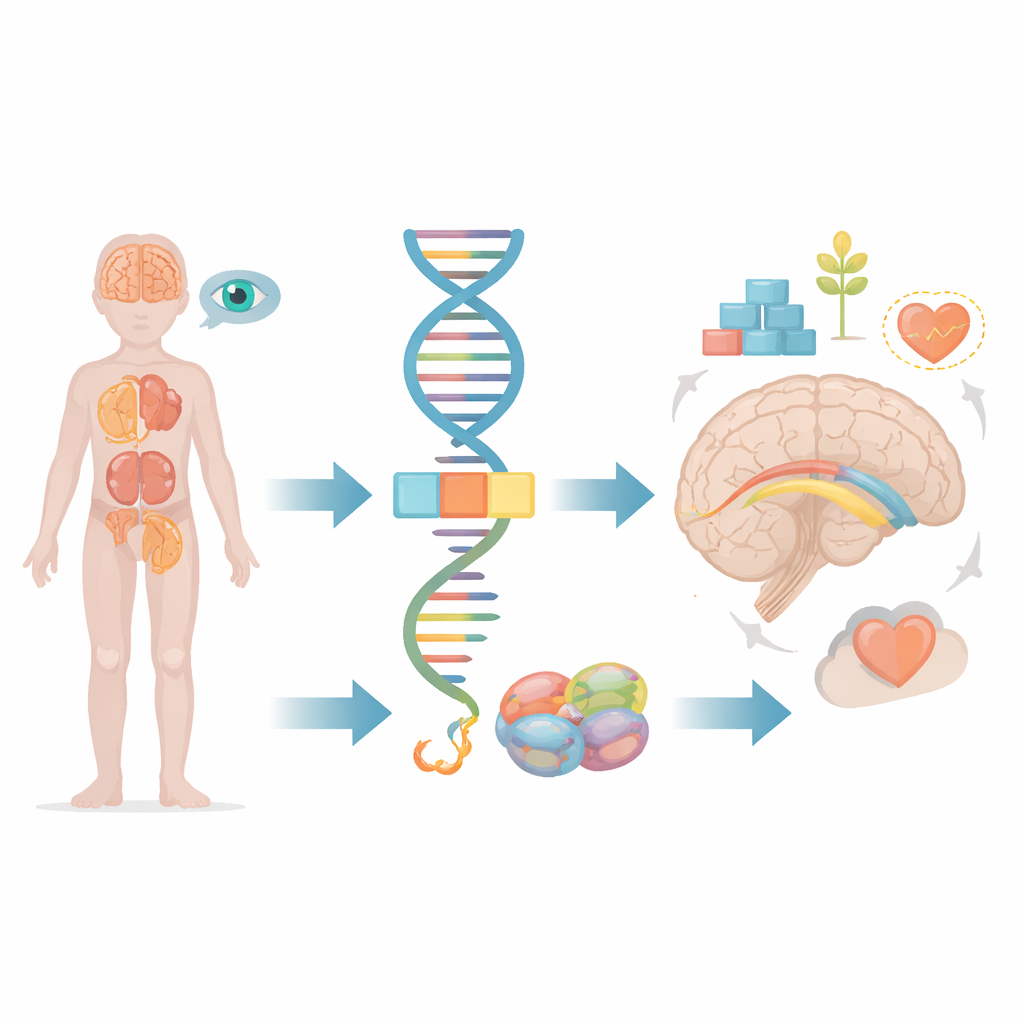
A Child’s Story Adds New Pieces
The authors describe an Egyptian boy born to related parents after an uncomplicated pregnancy. From early infancy he showed marked delay in all areas of development: he did not smile, follow with his eyes, respond to sounds, or develop head control at eight months of age. Doctors noted very low muscle tone, unstable trunk movements, and rapid, uncontrolled eye movements called nystagmus. His facial appearance included a long face, high forehead, folds of skin at the inner corners of the eyes, long eye openings, a flattened bridge of the nose, and a rounded nose tip with a long upper lip. As he grew older, he was eventually able to stand and say a few simple syllables, but he remained severely delayed and very hyperactive.
Hidden Changes in Brain, Eyes, and Kidneys
Scans of the child’s brain revealed several structural differences: the connecting bridge between the brain’s two halves was underdeveloped, the insulating material around nerve fibers was delayed in forming, and there was a small fluid-filled cyst deep in a region called the caudate nucleus. Eye examinations showed damage to cone cells in the retina, which are needed for sharp central and color vision, explaining his poor visual responses. Imaging of the abdomen uncovered another unusual finding: both kidneys were fused together and located low in the pelvis instead of their usual positions. These kidney and eye problems had been seen only rarely in earlier FRA10AC1 cases, so the team carefully searched the child’s entire set of protein-coding genes to check for any other plausible explanations and found none.
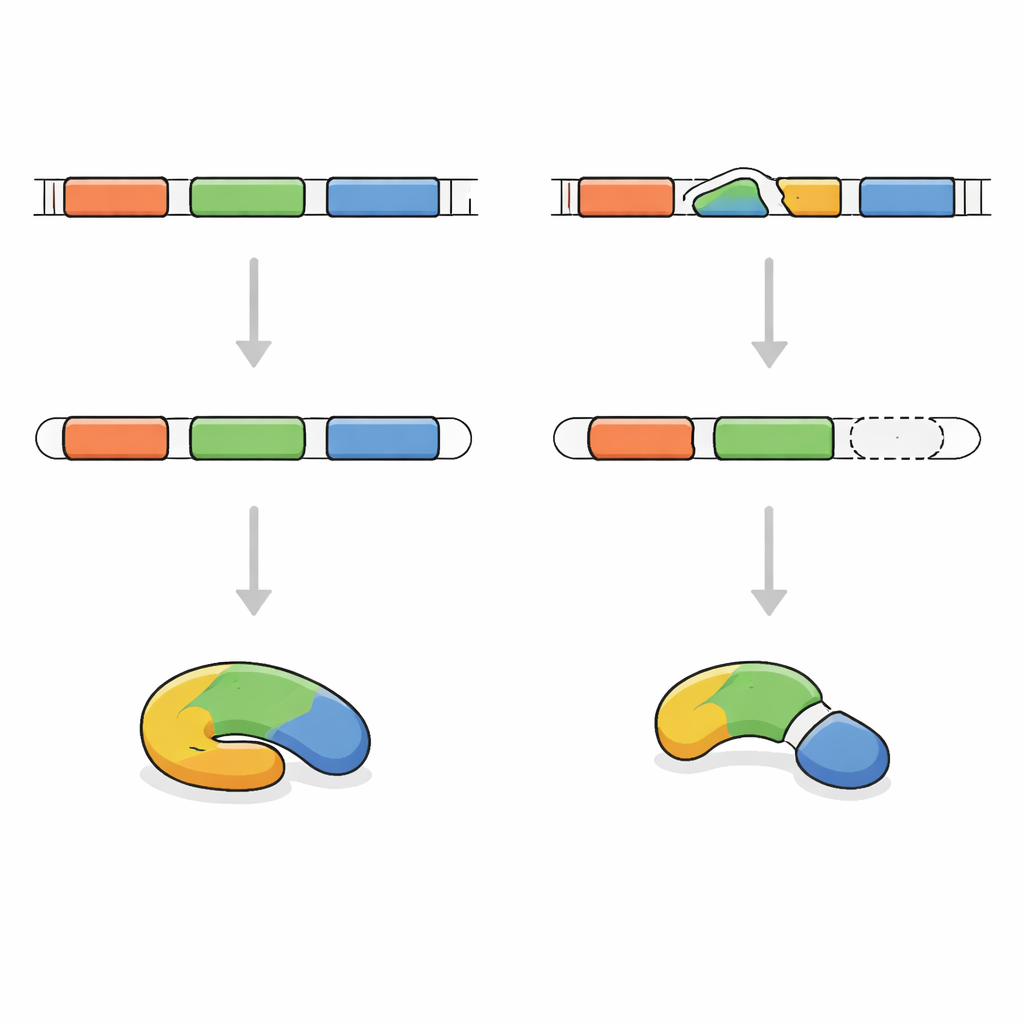
Tracking the Faulty Genetic Message
To pinpoint the cause, the researchers sequenced the protein-coding portions of the boy’s DNA. They discovered that both copies of his FRA10AC1 gene carried the same tiny change at a critical “join point” used during message editing. His parents, who were healthy, each carried one changed and one normal copy. The team then examined the FRA10AC1 message made from his blood cells. Instead of including all the expected segments, the message had skipped one piece entirely. This caused the instructions to shift out of step, introducing an early stop signal and leading to a shortened, probably useless protein. Based on international guidelines for judging genetic variants, this change was classified as clearly disease-causing.
What This Means for Families
By adding this boy’s case to the small number already known, the study helps clarify the picture of the FRA10AC1-related disorder. Children with complete loss of the protein, like this patient, tend to have more severe intellectual and motor difficulties and are more likely to have additional birth defects affecting organs such as the heart, kidneys, eyes, and skin. In contrast, previously reported children with a milder, partly working version of FRA10AC1 had less severe learning problems and fewer organ anomalies. For families and clinicians, this work underscores that a fault in this single gene can underlie a recognizable combination of facial features, developmental delays, and brain changes, and that careful checks of the kidneys and eyes are important once the diagnosis is suspected.
Citation: Abdel-Hamid, M.S., Abdel-Salam, G.M.H. A novel homozygous splicing variant in FRA10AC1: further delineation of the phenotype. J Hum Genet 71, 363–367 (2026). https://doi.org/10.1038/s10038-025-01447-6
Keywords: neurodevelopmental disorder, spliceosome, FRA10AC1 gene, genetic variants, corpus callosum