Clear Sky Science · en
DNA cross-over motifs-based, programmable supramolecular hydrogels for the mechanoregulatory effects of cellular behaviour and cytoskeleton reorganization
Building Better Cell-Friendly Gels
Our bodies are made of more than just cells; they are held together by soft, water-rich scaffolds that surround every cell. These natural scaffolds not only support tissues but also send mechanical cues that tell cells how to grow, move, and stay healthy. This study shows how scientists can use DNA – the same molecule that stores genetic information – to build highly tunable, jelly-like materials that imitate these scaffolds and gently steer how cells behave.
From Genetic Code to Smart Jello
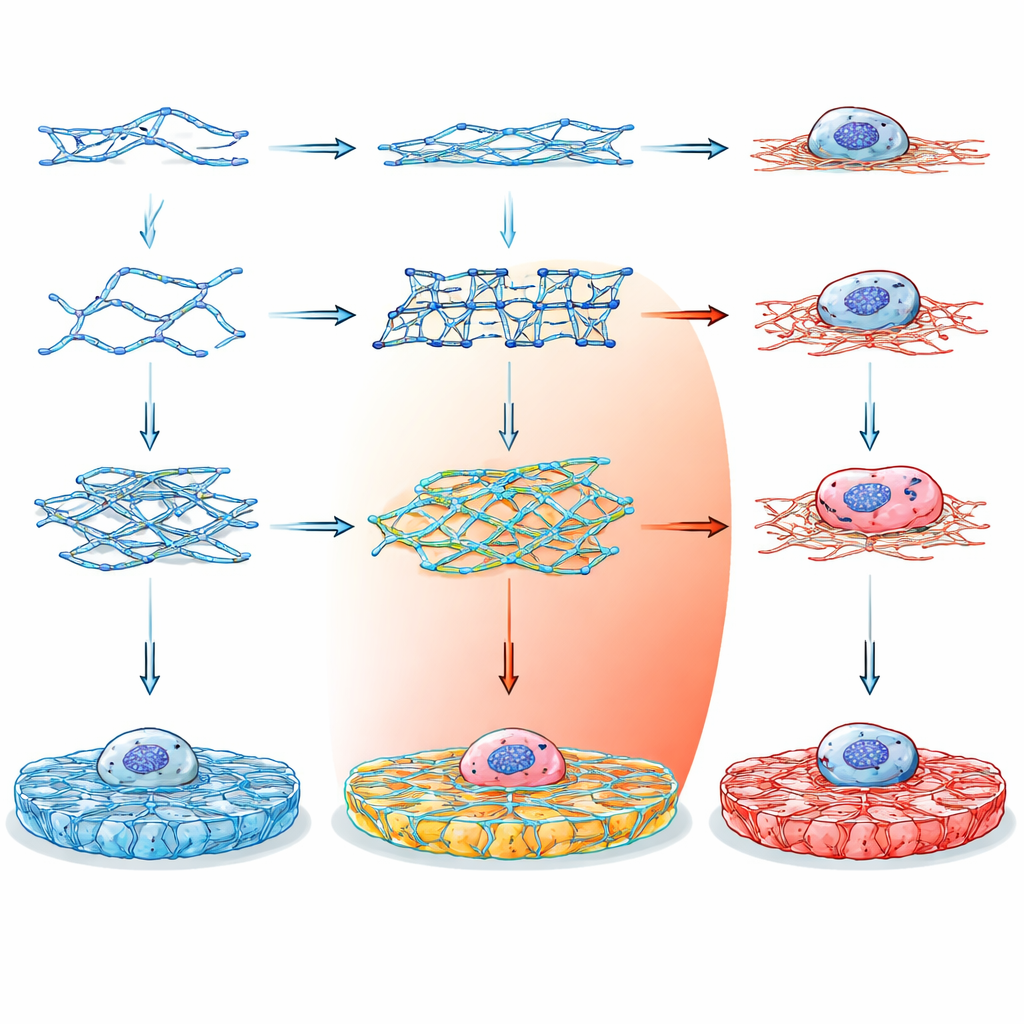
Instead of relying on traditional materials like collagen or alginate, which are hard to fine-tune without adding extra chemicals, the researchers turned to DNA as a construction material. DNA is attractive because its building rules are simple and predictable: specific bases pair up in well-known ways, allowing precise control over shape and connectivity at the nanometer scale. By designing short DNA strands that assemble into different multi-armed junctions, the team could program how the strands link into a three-dimensional hydrogel – a soft, water-filled solid that looks and feels like biological tissue.
Designing a Library of Custom Gels
The team created fourteen different DNA building blocks, grouped into three main families. Double crossover (DX) motifs are relatively simple, ladder-like units; paranemic crossover (PX) motifs have more frequent crosslinks, making them intrinsically stiffer; and tensegrity motifs form star-like units with three to six arms held under balanced tension. Some versions had blunt ends and could not form networks, serving as controls. Others carried sticky ends that allowed them to hook together into extended gels, either with flexible palindromic connections or more directional, non-palindromic links. By carefully choosing strand sequences and checking their stability with computational tools, the researchers ensured that each motif folded as planned and remained biologically safe.
Seeing and Feeling the DNA Networks
To confirm that these tiny building blocks really formed larger structures, the team used several visualization and mechanical probes. Fluorescent dyes that bind DNA revealed how different motifs organized into networks under the microscope: some formed loose, clumped webs, while others created more regular, evenly spaced meshes. Atomic force microscopy gave a dried-state view at the nanometer scale, showing that tensegrity-based gels produced thick, bundled fibers that in some cases resembled natural collagen. By gently poking the hydrated gels with a microscopic cantilever and performing bulk rheology measurements, they found that the gels spanned a wide range of stiffness, from about 50 to 185 kilopascals, and stayed solid-like even under strains much larger than those tissues typically experience in the body.
Cells Respond to the DNA Landscape
The real test was whether living cells would notice – and benefit from – these engineered mechanical environments. Human retinal pigment epithelial cells were grown on coverslips coated with the various DNA gels and compared with standard coatings such as poly-L-lysine, collagen, and commercial basement-membrane extracts. Across multiple DNA architectures and concentrations, cell viability increased, in some cases up to fourfold relative to the soft poly-L-lysine control. Cells on appropriately tuned DNA gels spread out more, with larger areas and better-developed internal scaffolds made of actin filaments. Their nuclei also enlarged, a hallmark of cells engaging strongly with their surroundings.
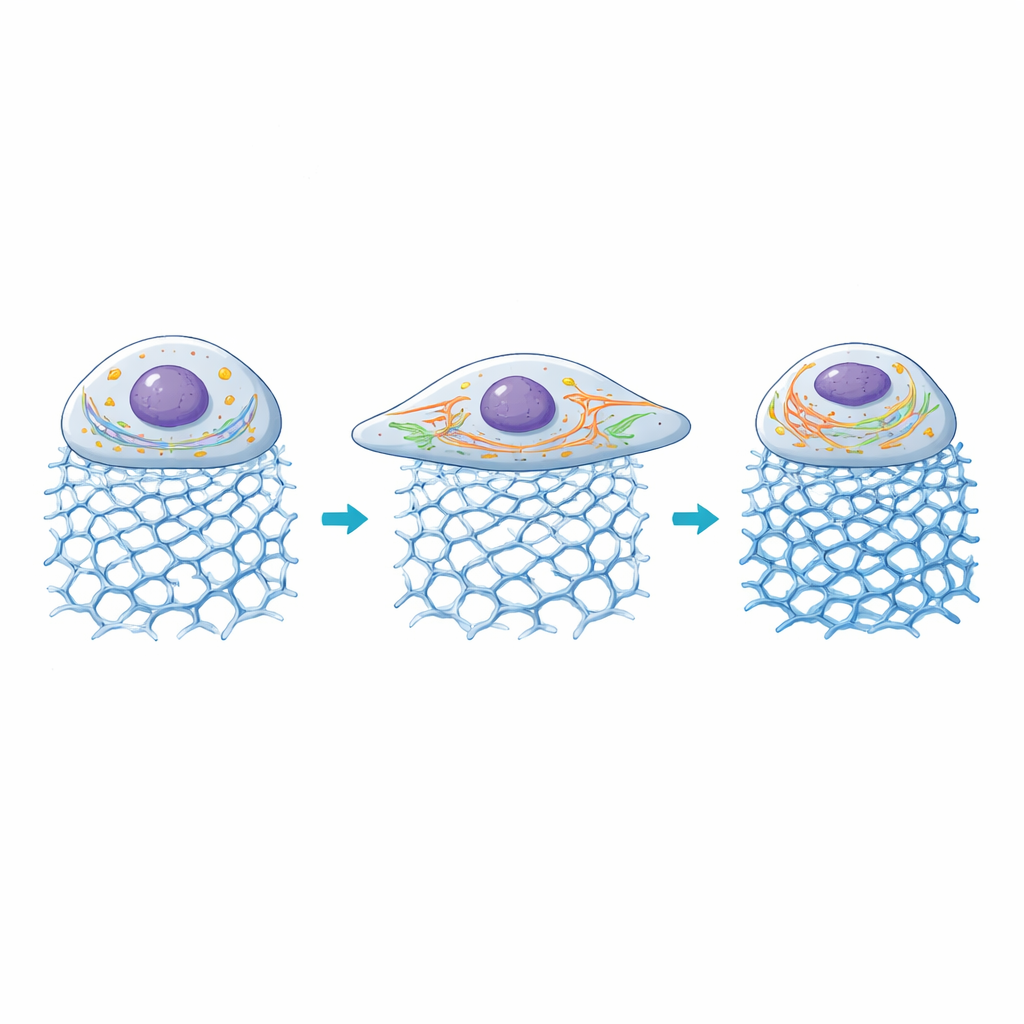
Inside the Cell: Powerhouses and Highways Adapt
The researchers then looked at how two key internal structures reacted: mitochondria, which supply energy, and the endoplasmic reticulum (ER), a network that helps process proteins and calcium signals. On gels with moderate stiffness, mitochondria became more fragmented, a state associated with higher energy turnover during active growth, while ER tubules elongated and spread through the enlarged cell body. As stiffness rose beyond roughly 100 kilopascals, cell area began to shrink again, mitochondrial networks became more fused, and ER signals declined, suggesting that very rigid environments push these cells out of their comfort zone. Overall, the gels allowed the scientists to link specific DNA architectures and stiffness levels to distinct patterns of organelle organization and mechanical signaling.
Toward Personalized Tissue Scaffolds
This work demonstrates that DNA can serve not just as a carrier of genetic information, but as a programmable construction material for building cell-sized landscapes with finely tuned mechanical properties. By mixing and matching different DNA motifs and sequences, it should be possible to dial in the stiffness and structure needed for particular cell types or tissues, and even to add responsive elements that react to changes in the environment. Such DNA-based hydrogels point toward future personalized scaffolds that not only support cells in three dimensions, but also actively guide their growth, health, and repair.
Citation: Singh, A., Yadav, A., Singh, N. et al. DNA cross-over motifs-based, programmable supramolecular hydrogels for the mechanoregulatory effects of cellular behaviour and cytoskeleton reorganization. npj Biomed. Innov. 3, 30 (2026). https://doi.org/10.1038/s44385-026-00083-9
Keywords: DNA hydrogels, mechanobiology, tissue engineering, cellular mechanotransduction, extracellular matrix