Clear Sky Science · en
Structural insights into the photochemistry of the LH1–RC complex from the marine purple phototrophic bacterium Rhodovulum sulfidophilum
How a Tiny Marine Bacterium Turns Light and Sulfur into Energy
In the shallow, sulfur‑rich muds of coastal seas lives a purple bacterium that quietly recycles chemicals and captures sunlight. This study zooms in on its main solar machine, revealing atom‑level details of how light is harvested and converted into usable energy. Understanding this natural "micro–solar panel" not only deepens our picture of life in the oceans, but also offers inspiration for future light‑driven technologies.
A Close Look at Nature’s Micro Solar Panel
The researchers focused on a marine microbe called Rhodovulum sulfidophilum, long used as a model for bacterial photosynthesis because it can grow in both oxygen‑poor light and fully oxygen‑rich conditions, and can tolerate high levels of sulfur. Its core light‑using machine is a combined light‑harvesting 1–reaction center complex, or LH1–RC, embedded in internal membranes. Using high‑resolution cryo‑electron microscopy, the team determined the structure of this complex down to 1.81 angstroms—fine enough to see individual pigments, lipids, and even hundreds of water molecules. They found an open ring of 16 repeating light‑harvesting units wrapped around a central reaction center where charge separation occurs, with a deliberate gap that seems to be the only path for key molecules to move in and out.
A Special Wire for Moving Electrons
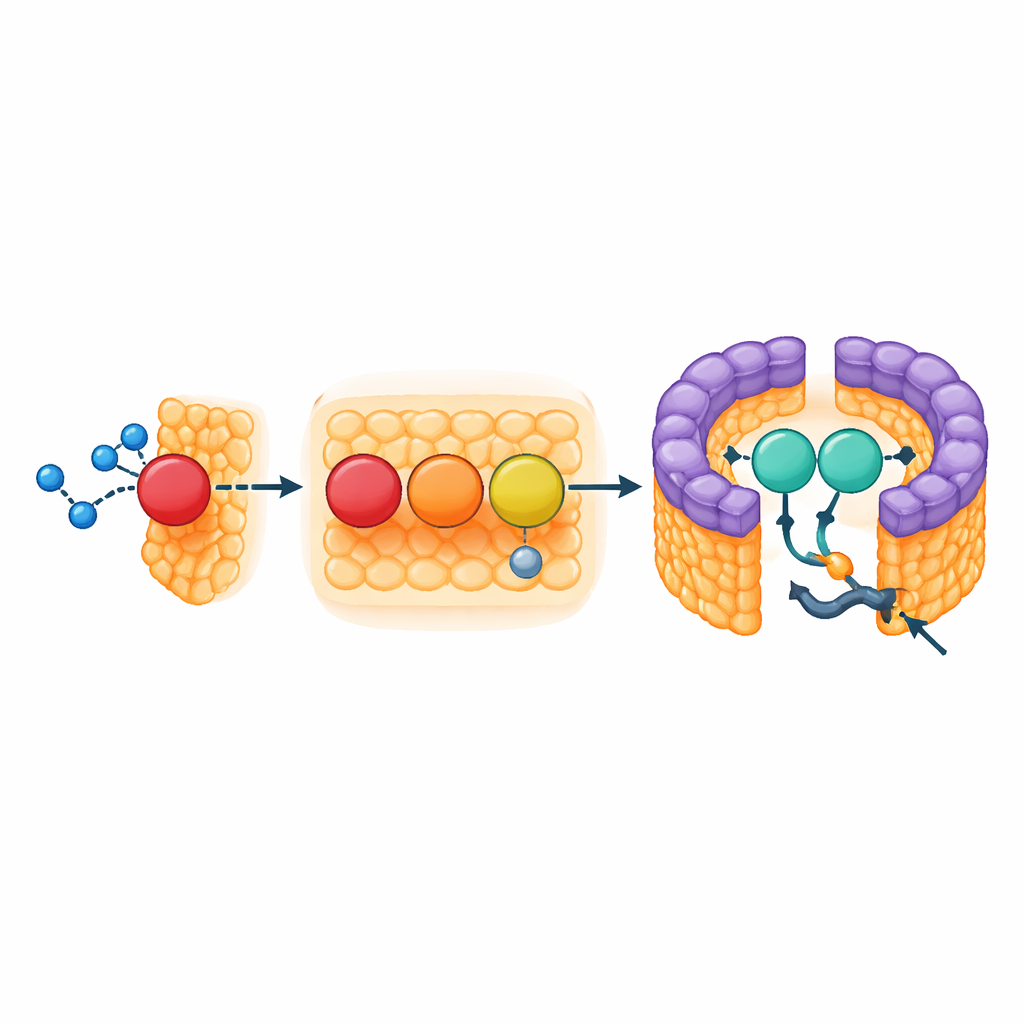
At the heart of the reaction center is a protein subunit that acts like a built‑in wire for moving electrons, known as a cytochrome. In many related bacteria this wire carries four metal‑bearing heme groups, but in Rhodovulum sulfidophilum there are only three. This raised a puzzle: how can electrons flow in efficiently without the usual outermost heme? The new structure shows that the middle heme (called heme‑2) is ligated, or held, by an unusual cysteine amino acid instead of the methionine seen in other species. This subtle swap greatly lowers its redox potential, making it behave more like the missing outer heme in other bacteria. The surface around this heme is open and shaped to let soluble electron‑donating proteins approach, hinting that this is a key landing site for incoming electrons.
An Extra Metal Helper Near the Electron Path
Unexpectedly, the team discovered a nonheme iron atom nestled close to this middle heme, coordinated by a histidine from the cytochrome and five water molecules. Spectroscopic measurements confirmed its presence and showed that it sits very close to heme‑2, but farther from the other two hemes. This suggests the extra iron may act as a relay station, briefly accepting electrons from soluble donors before passing them to the cytochrome’s metal centers. By combining structural and electron‑spin measurements, the authors argue that heme‑2, aided by this nearby iron cluster, is the likeliest first recipient of electrons from the bacterium’s soluble cytochromes, helping the organism cope with large energy gaps between donors and acceptors in its sulfur‑rich environment.
A Supporting Brace at the Ring Opening
The LH1 part of the complex forms a tight, pigment‑laden fence that captures light and funnels the excitation to the reaction center, but its ring is not fully closed. At the gap sit two structural elements: the membrane‑spanning start of the cytochrome subunit and a small three‑helix protein the authors call protein‑3h. Protein‑3h is derived from a larger gene in the bacterium’s respiratory chain (a pseudo‑gene known as urf1) and appears to have been repurposed. In the LH1–RC complex, it wedges into the gap and grabs onto both the cytochrome and neighboring light‑harvesting units, acting as a brace that stabilizes the opening. Because the dense carotenoid pigments block other routes, this opening likely serves as the sole gateway for quinone molecules that carry electrons away from the reaction center into the broader membrane network.
Why This Micro Machine Matters
Taken together, the results show how Rhodovulum sulfidophilum has fine‑tuned its photosynthetic hardware for life in variable, sulfur‑rich coastal waters. It uses a triheme cytochrome with an unusual metal binding site to accept electrons flexibly from different donors, a tightly sealed light‑harvesting ring to capture and hold energy, and a repurposed three‑helix protein to both stabilize and shape the only exit route for mobile carriers. For non‑specialists, the key message is that even single‑celled organisms can evolve intricate, modular solar devices whose parts are swapped, trimmed, and reassigned over time—offering natural design principles for robust, efficient light‑driven systems.
Citation: Yue, XY., Wang, GL., Kosaki, S. et al. Structural insights into the photochemistry of the LH1–RC complex from the marine purple phototrophic bacterium Rhodovulum sulfidophilum. Commun Biol 9, 502 (2026). https://doi.org/10.1038/s42003-026-09755-z
Keywords: bacterial photosynthesis, light harvesting complex, electron transfer, cryo electron microscopy, sulfur cycling