Clear Sky Science · en
The C16orf87 protein is a subunit of the MIER corepressor complex controlling embryonic development and cell migration
Why a Small Protein Matters for Growth and Health
Every cell in our body has to manage an enormous amount of genetic information, turning the right genes on or off at the right time. This paper uncovers the role of a previously mysterious human protein, C16orf87, now proposed to be renamed HDAC Interacting Protein (HDIP). The researchers show that this small molecule helps control how tightly our DNA is packed, how cancer cells move, and how embryos develop—linking basic gene control to cell behavior and body shape in a living animal. 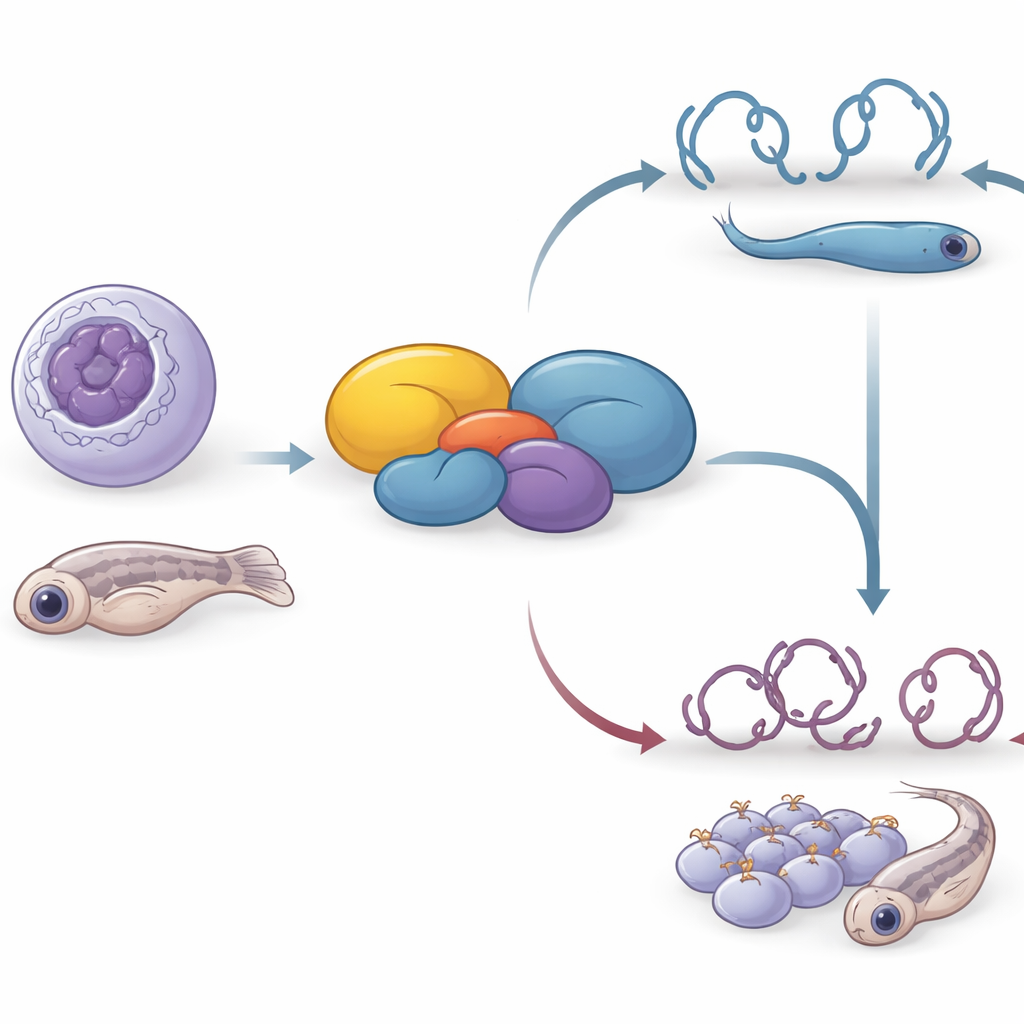
A Hidden Helper in DNA Packaging
DNA inside our cells is wrapped around proteins to form a structure called chromatin, which can be more open or more compact. This packing strongly influences whether genes are active or silent. Enzymes known as histone deacetylases (HDACs) help tighten chromatin and shut down gene activity. They rarely work alone; instead, they sit inside larger protein teams called corepressor complexes. Many members of these complexes are still poorly understood. By combining protein chemistry, structural prediction, and cell experiments, the authors identify C16orf87 as a missing piece of one such team, the MIER corepressor complex, which includes the key enzyme HDAC1.
How HDIP Connects a Gene-Silencing Machine
The researchers found that C16orf87 physically associates with HDAC1, HDAC2, and the scaffold proteins MIER1 and MIER3, which help bring HDACs to specific stretches of DNA. Using mass spectrometry and pull-down experiments in human cells, they show that C16orf87 acts as a connector between HDAC1 and MIER1 inside the MIER complex. When C16orf87 levels were reduced by RNA interference, or completely removed by CRISPR-based gene editing, the levels of several histone proteins changed, suggesting that this connector helps maintain the right balance of DNA-packaging components. Advanced structure predictions with AlphaFold3 further support a model in which C16orf87 is mostly flexible but uses a short, well-formed tail to dock between HDAC1 and the central MIER1 region, stabilizing their partnership without directly changing HDAC1’s chemical activity. 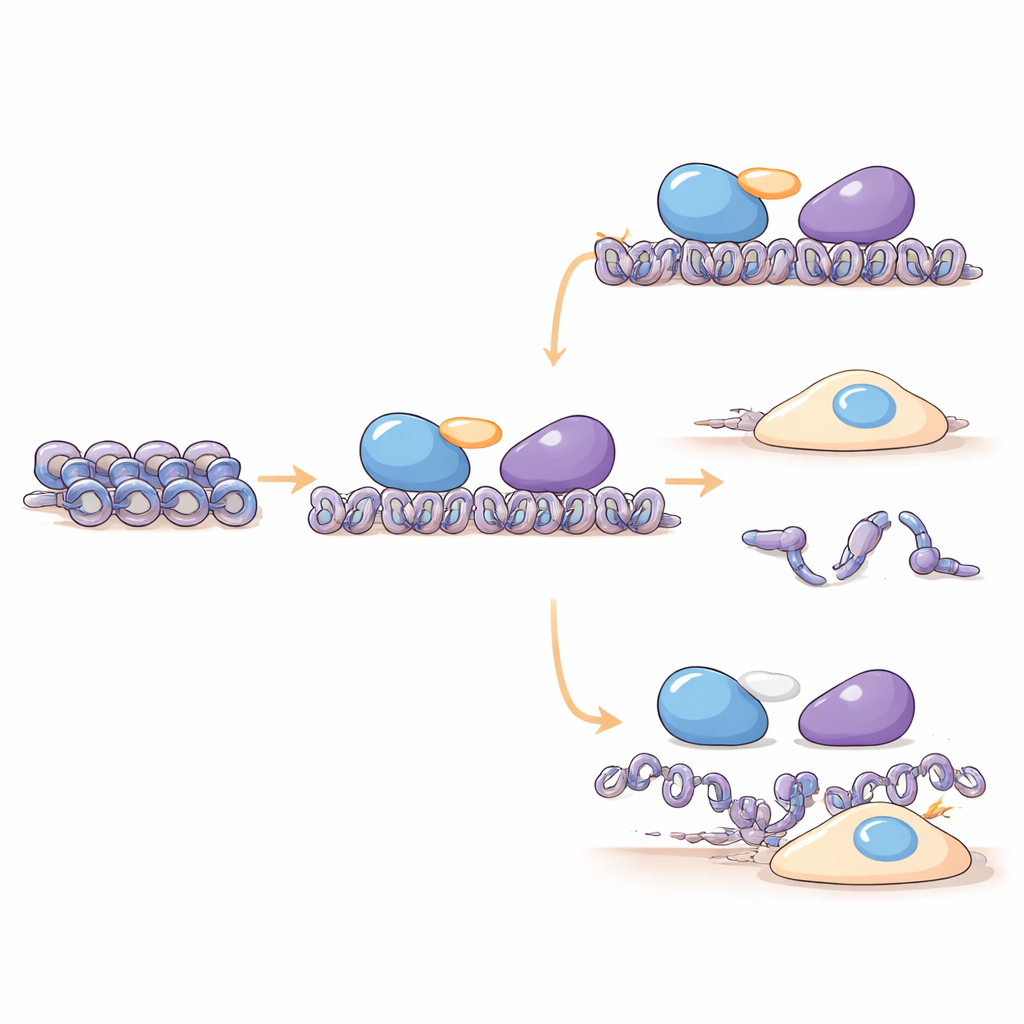
Changing the Connector Changes Chromatin and Cell Behavior
To see what happens when this connector protein is missing, the team deleted C16orf87 in a human pancreatic cancer cell line. The edited cells showed reduced levels of several core histones but, perhaps surprisingly, remained viable and even replicated their DNA slightly faster. However, when the scientists tested how these cells migrated across a surface—a key behavior for processes like wound healing and cancer spread—the cells without C16orf87 moved significantly more slowly. Genome-wide measurements of chromatin accessibility (using ATAC-seq) revealed that loss of C16orf87 made many specific DNA regions more open, particularly at sites previously known to be targeted by HDAC1/2 and MIER proteins. Some of these regions lie near genes involved in cell signaling and stress responses, and shifts in chromatin openness often went hand-in-hand with changes in gene activity.
From Cells in a Dish to Developing Fish
Because HDAC-containing complexes are known to guide early development, the researchers next asked whether the same connector protein matters in a whole organism. They disrupted the zebrafish version of the gene, called C7H16orf87, using CRISPR in embryos. Compared with their siblings, fish lacking functional C7H16orf87 were shorter, had smaller eyes and body surface area, and showed a more curved back and altered head-to-body angle. These defects reveal that the protein is not absolutely required for survival but is important for proper shaping and growth of the developing body, reinforcing the idea that fine-tuned chromatin control is essential during embryogenesis.
What This Means for Future Research and Medicine
Taken together, the findings paint C16orf87/HDIP as a flexible adaptor that helps assemble a specific gene-silencing complex and direct it to selected spots in the genome. By influencing which regions of DNA are tightly packed versus accessible, HDIP shapes patterns of gene activity that affect cell migration in human cancer cells and body formation in zebrafish larvae. Because HDACs are already targets of anti-cancer drugs, understanding their supporting partners like HDIP may open doors to more precise therapies that nudge only particular gene programs, rather than broadly blocking all HDAC activity. This work turns an obscure gene name into a functional story, linking molecular connections inside the nucleus to visible changes in how organisms grow and move.
Citation: Punga, T., Larsson, M., Mujica, E. et al. The C16orf87 protein is a subunit of the MIER corepressor complex controlling embryonic development and cell migration. Sci Rep 16, 13907 (2026). https://doi.org/10.1038/s41598-026-50740-7
Keywords: chromatin regulation, histone deacetylase, embryonic development, cell migration, MIER complex