Clear Sky Science · en
Impact of culture dimensionality and matrix composition on morphology, phenotype and drug response in pancreatic cancer models
Why Growing Tumors in New Ways Matters
Pancreatic cancer is one of the deadliest cancers, in part because it is often found late and resists many common drugs. To develop better treatments, researchers rely on tumor cells grown in the lab as stand‑ins for real cancers. But there is a basic question behind this approach: does the way we grow these cells change how they behave and respond to medicines? This study explores whether growing pancreatic tumor cells in flat layers or three‑dimensional clusters, and in different gel‑like materials, truly changes what we learn about the disease and its treatment.
From Patient Tumor to Lab-Grown Models
The researchers collected tumor samples from 12 patients with pancreatic ductal adenocarcinoma, the most common form of pancreatic cancer. From each tumor, they tried to create three kinds of lab models: a classic flat layer of cells in a dish (2D culture) and two types of three‑dimensional “mini‑tumors,” or organoids, grown in different commercial gel matrices called Matrigel and Cultrex. These gels mimic the soft, protein‑rich material that surrounds cells inside the body. The team also checked which genetic mutations were present to confirm that these lab‑grown models truly came from cancer cells and carried well‑known driver mutations. 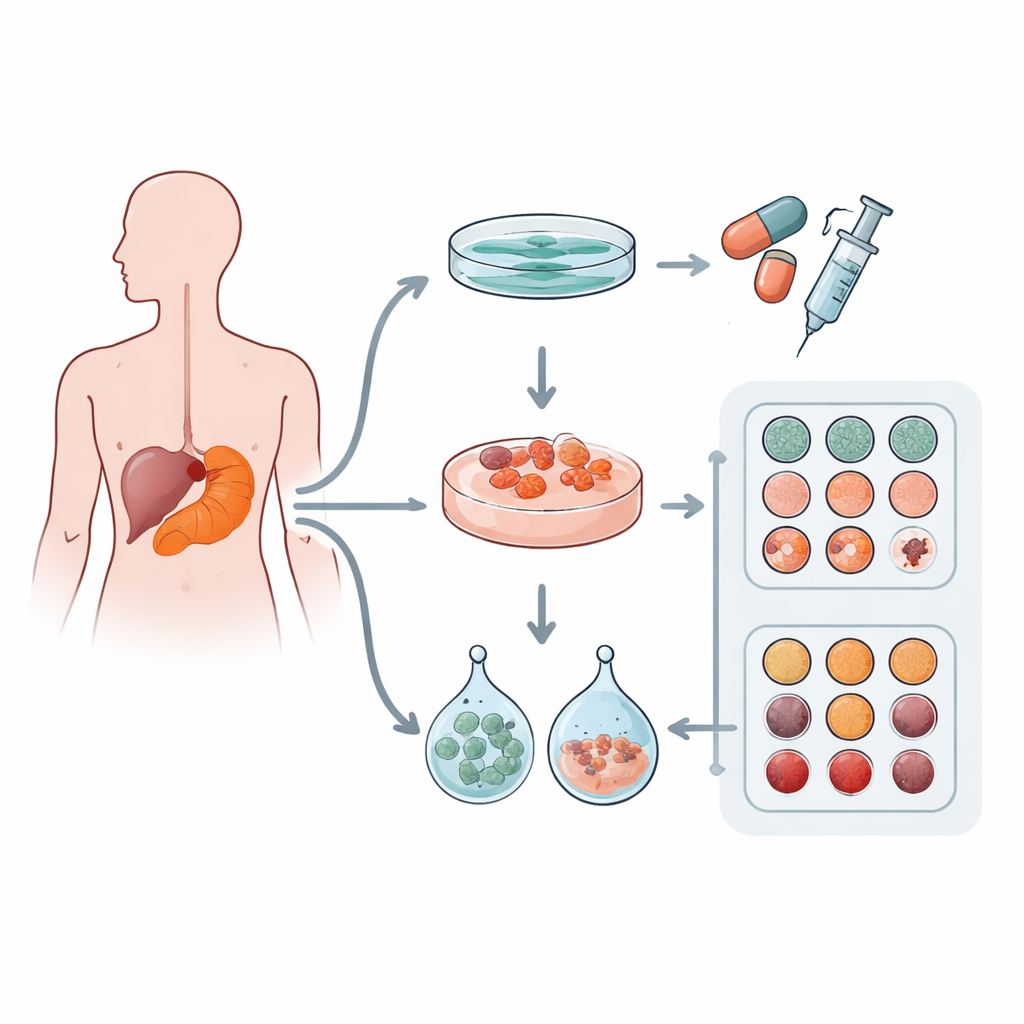
How Tumor Cells Grow and Look in the Lab
When they compared how the cells grew, the differences between flat and 3D cultures were striking. In 2D, cells spread out as thin sheets with little clustering, a pattern far removed from how tumors appear in the body. In contrast, organoids grown in 3D gels formed compact spheres or hollow, cyst‑like structures that more closely resembled the gland‑like architecture of real pancreatic tumors. Interestingly, the exact shape of these organoids depended more on the individual patient’s tumor than on which gel was used: the better‑differentiated cancers formed gland‑like organoids, while a more aggressive, poorly differentiated tumor produced dense, nest‑like clusters. Across conditions, 3D cultures divided faster and needed less time before their first split than the flat cultures, echoing higher activity levels seen in their cell division marker, Ki‑67.
Keeping the Tumor’s Identity Intact
The team then asked whether these different ways of growing cells might distort their basic identity. Using staining techniques similar to those used in hospital pathology labs, they looked at key proteins that define pancreatic duct cancers. Markers such as CK19 and GATA6, which signal a duct‑like cell type, were preserved in both flat and 3D systems. Proteins tied to the tumor’s genetic makeup, including p53 and SMAD4, showed patterns that matched the original tumors. A marker of cell division, Ki‑67, was higher in all lab models than in the original tissue, reflecting the growth‑promoting conditions of culture. Another marker, CDX2, rose especially in flat cultures, hinting that some aspects of cell identity can shift in the lab. Overall, however, both 2D and 3D approaches maintained the core features of the patients’ cancers.
Testing Chemotherapy in Different Lab Worlds
The heart of the study was to see whether culture format—flat versus 3D, and choice of gel—changes how tumor cells respond to common chemotherapy drugs used for pancreatic cancer, including gemcitabine, 5‑fluorouracil, oxaliplatin, SN‑38 and paclitaxel. Across the small patient set, drug sensitivity patterns were remarkably similar between 2D cultures and both types of organoids. Three‑dimensional models tended to be slightly more drug‑resistant, which matches their more tumor‑like structure and limited drug penetration, but these differences were modest and not statistically strong. What did stand out was how much responses varied from patient to patient: for several drugs, especially gemcitabine, some patients’ cells were far more resistant than others, regardless of how the cells were grown. In two patients who actually received gemcitabine after surgery, the lab results mirrored reality: the patient whose models were more resistant relapsed sooner, and their cultures showed higher levels of MDR1, a protein known to pump drugs out of cells. 
What This Means for Future Cancer Testing
For a lay reader, the key takeaway is reassuring: the main message coming from lab tests of standard chemotherapy drugs seems to depend much more on the individual patient’s tumor than on whether the cells are grown flat or as 3D clusters, or on which common gel is used. That means existing lab systems are reasonably robust for comparing how patients’ tumors respond to these drugs. At the same time, 3D organoids better mimic the real tumor’s structure and growth behavior, making them especially valuable for studying disease biology and possibly for more complex or targeted therapies. Together, these findings support the growing use of patient‑derived organoids as realistic, patient‑specific testbeds that could one day help doctors tailor treatments for people with pancreatic cancer.
Citation: Doelvers, F., Wansch, K., Kuehn, A. et al. Impact of culture dimensionality and matrix composition on morphology, phenotype and drug response in pancreatic cancer models. Sci Rep 16, 12346 (2026). https://doi.org/10.1038/s41598-026-47856-1
Keywords: pancreatic cancer, patient-derived organoids, 3D cell culture, chemotherapy response, drug resistance