Clear Sky Science · en
Reduced ROS-associated prophage induction in a lepA mutant contributes to increased fluoroquinolone persistence in Salmonella Typhimurium
Why some germs survive even our strongest drugs
When doctors use powerful antibiotics to treat serious infections, most bacteria die quickly. Yet a tiny minority can slip into a dormant state, ride out the attack, and later regrow the infection. These hardy survivors, called persister cells, do not carry classic resistance genes, so they are hard to predict and harder to eliminate. This study explores why certain Salmonella bacteria are more likely to form persisters during treatment with the widely used fluoroquinolone antibiotic ciprofloxacin, and how tiny bursts of chemically reactive oxygen and hidden viruses inside the bacteria can tip the balance between death and survival.
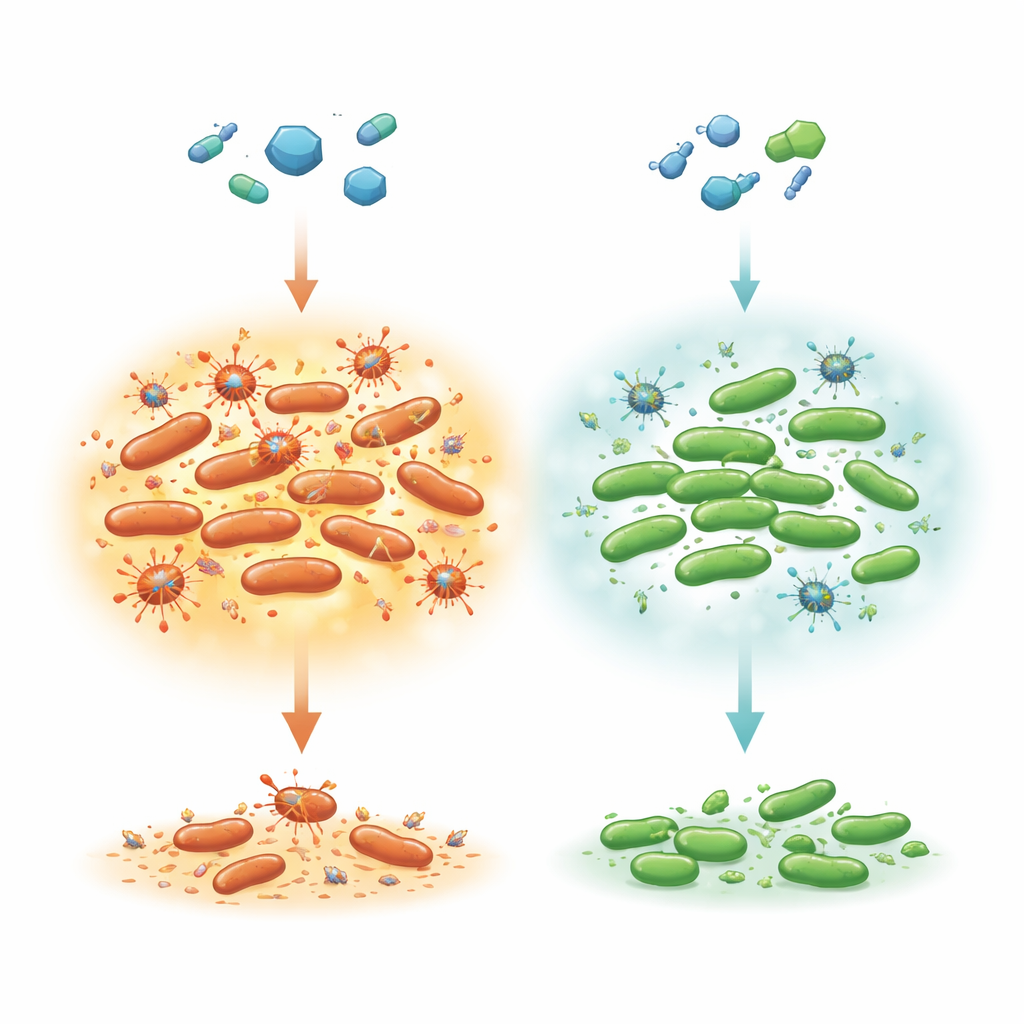
Hidden survivors inside an antibiotic attack
Most bacteria in a population are rapidly killed by an effective antibiotic, but a very small subpopulation dies much more slowly. This "two-step" killing pattern is a hallmark of persister cells, which simply hunker down until the drug is gone, then resume normal growth. The authors focused on Salmonella Typhimurium, a common cause of foodborne illness that can require ciprofloxacin treatment in vulnerable patients. Earlier work in Escherichia coli suggested that removing a translation factor protein called LepA made cells unusually tolerant to several antibiotics by reducing the buildup of reactive oxygen species (ROS) — chemically aggressive by-products of metabolism that can damage DNA and proteins. Here, the researchers asked whether LepA plays a similar role in Salmonella and how this interacts with viruses (prophages) that lie dormant inside the bacterial chromosome.
What changed in bacteria missing a single protein
The team compared normal Salmonella with a version lacking the lepA gene. They exposed both to three major classes of cell-killing antibiotics: ciprofloxacin (a fluoroquinolone), ampicillin (a beta-lactam), and kanamycin (an aminoglycoside). For ampicillin and kanamycin, both strains behaved similarly, showing the usual rapid die-off of most cells and a plateau of rare persisters. In contrast, after ciprofloxacin treatment, the lepA mutant consistently left behind more persister cells than the normal strain. Measurements of ROS using a fluorescent dye showed that ciprofloxacin triggered strong ROS formation, especially hydroxyl radicals, and that these levels were lower in the lepA mutant. Oxygen-consumption assays indicated that mutant cells ramped up respiration — and thus ROS production — more slowly, suggesting that subtle defects in the cell’s energy machinery keep oxidative stress lower and favor survival under drug pressure.
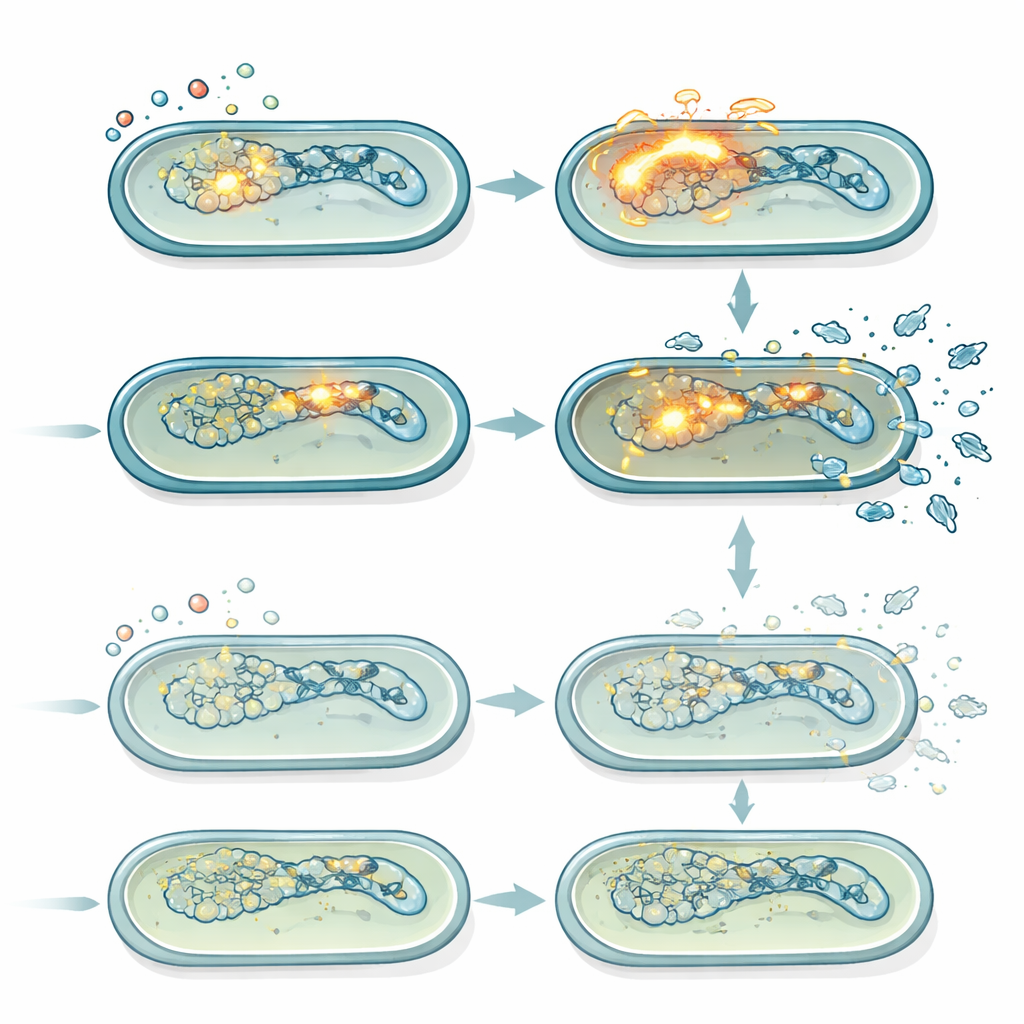
Silent viruses that turn lethal under stress
Salmonella Typhimurium carries several prophages, dormant viral genomes that can awaken when the bacterial DNA is damaged. When activated, these prophages switch to a lytic program, replicate, and cause the host cell to burst. Using gene expression measurements and fluorescent reporter strains, the authors showed that ciprofloxacin strongly activated the bacterial SOS DNA-damage response and, in normal cells, triggered robust induction of one prophage in particular (Gifsy-1). This induction was weaker in the lepA mutant, matching its lower ROS levels. Plaque assays that count active phage particles confirmed that, at moderate ciprofloxacin doses, the normal strain continually produced more phages over time than the lepA mutant. When all prophages were genetically removed, the survival advantage of the lepA mutant at moderate ciprofloxacin largely disappeared, revealing that the key difference between strains was how strongly ROS-driven prophage activation contributed to killing.
Turning down oxygen stress changes who lives
The researchers then used glutathione, a natural antioxidant, to dampen ROS during ciprofloxacin treatment. In normal, prophage-carrying Salmonella, glutathione expanded the subpopulation of cells that did not show prophage induction and increased the fraction of persisters. In a prophage-free background, the same treatment slowed early killing but no longer boosted persister levels, underscoring that the survival benefit was tied to prophage suppression. At very high ciprofloxacin doses, antioxidants again increased survival far more in prophage-positive cells than in prophage-free ones, indicating that ROS-triggered prophage activation can be important even under intense drug pressure. Similar patterns were seen in E. coli strains carrying the well-known lambda prophage: ciprofloxacin, but not ampicillin, drove prophage-mediated lysis and reduced persisters in normal cells, whereas lepA mutants and prophage-free strains avoided this additional killing.
What this means for fighting stubborn infections
The work supports a simple but powerful idea: for bacteria that harbor dormant prophages, antibiotic-induced ROS can push these hidden viruses into action, adding an extra wave of killing that shrinks the pool of drug-tolerant persisters. In Salmonella lacking LepA, lower ROS means weaker prophage activation and more cells that quietly survive ciprofloxacin. This suggests that, in some settings, carefully harnessing ROS and prophages could make existing antibiotics more effective — a concept known as phage–antibiotic synergy. At the same time, boosting ROS carries risks, because prophages can also spread virulence and resistance genes to other bacteria. The study highlights how a single bacterial protein, the state of the cell’s metabolism, and the presence of silent viruses together determine whether an antibiotic course clears an infection or leaves behind survivors that may cause trouble later.
Citation: Braetz, S., Karp, M., Nerlich, A. et al. Reduced ROS-associated prophage induction in a lepA mutant contributes to increased fluoroquinolone persistence in Salmonella Typhimurium. Sci Rep 16, 12721 (2026). https://doi.org/10.1038/s41598-026-47552-0
Keywords: antibiotic persistence, reactive oxygen species, prophages, Salmonella Typhimurium, ciprofloxacin