Clear Sky Science · en
Structural and In silico safety evaluation of asciminib degradation products with a validated stability indicating HPLC method for genotoxic impurity determination
Why the safety of a cancer pill keeps changing over time
Many modern cancer drugs are taken for months or years, which means the pills must stay safe and effective over long periods. But medicines slowly break down, producing new molecules that were never part of the original recipe. This study looks at how the leukemia drug asciminib falls apart under harsh conditions, how to detect the tiny breakdown products, and whether computer tools suggest that any of them might be harmful.
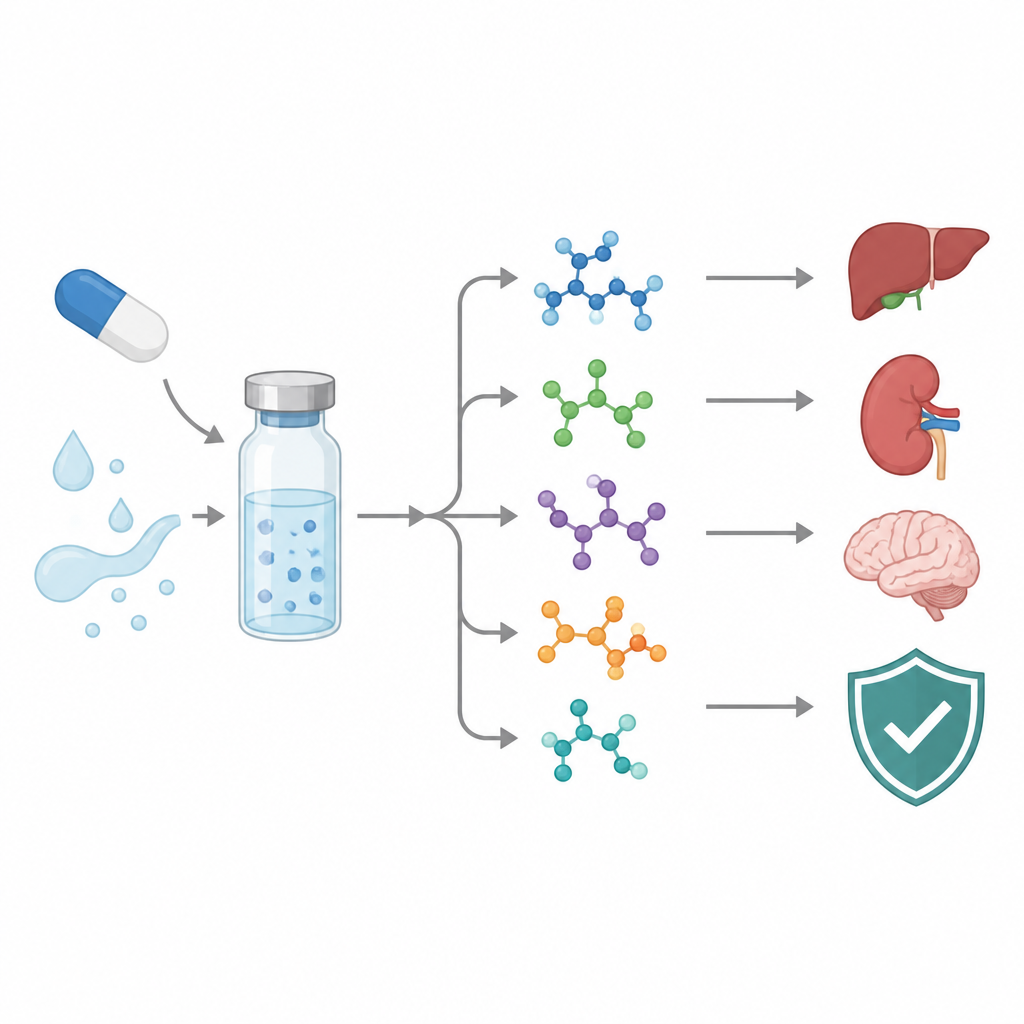
Following a cancer drug as it breaks apart
Asciminib is a targeted pill used for a blood cancer called chronic myeloid leukemia. Like all drugs, it can react with acids, bases, light, heat, or oxygen during manufacturing, storage, or inside the body. These reactions create degradation products, and a few may be “genotoxic,” meaning they could damage DNA. Regulators now require companies to search for such by-products, figure out what they are, and keep their levels under tight control. The researchers set out to map how asciminib degrades, identify the new molecules that appear, and develop a practical lab test that separates and measures the drug and its impurities.
Building a lab test that can see the smallest traces
The team designed and fine-tuned a liquid chromatography method, a workhorse separation technique in pharmaceutical labs. They tested several columns and solvent mixtures before choosing conditions that cleanly separated asciminib from three known genotoxic impurities and a range of unknown breakdown products. The method was checked for accuracy, sensitivity, and robustness, following international guidelines. It could pick up very low impurity levels and showed consistent performance even when conditions were slightly varied. This makes it suitable for routine quality control to ensure that each batch of tablets meets strict purity limits.
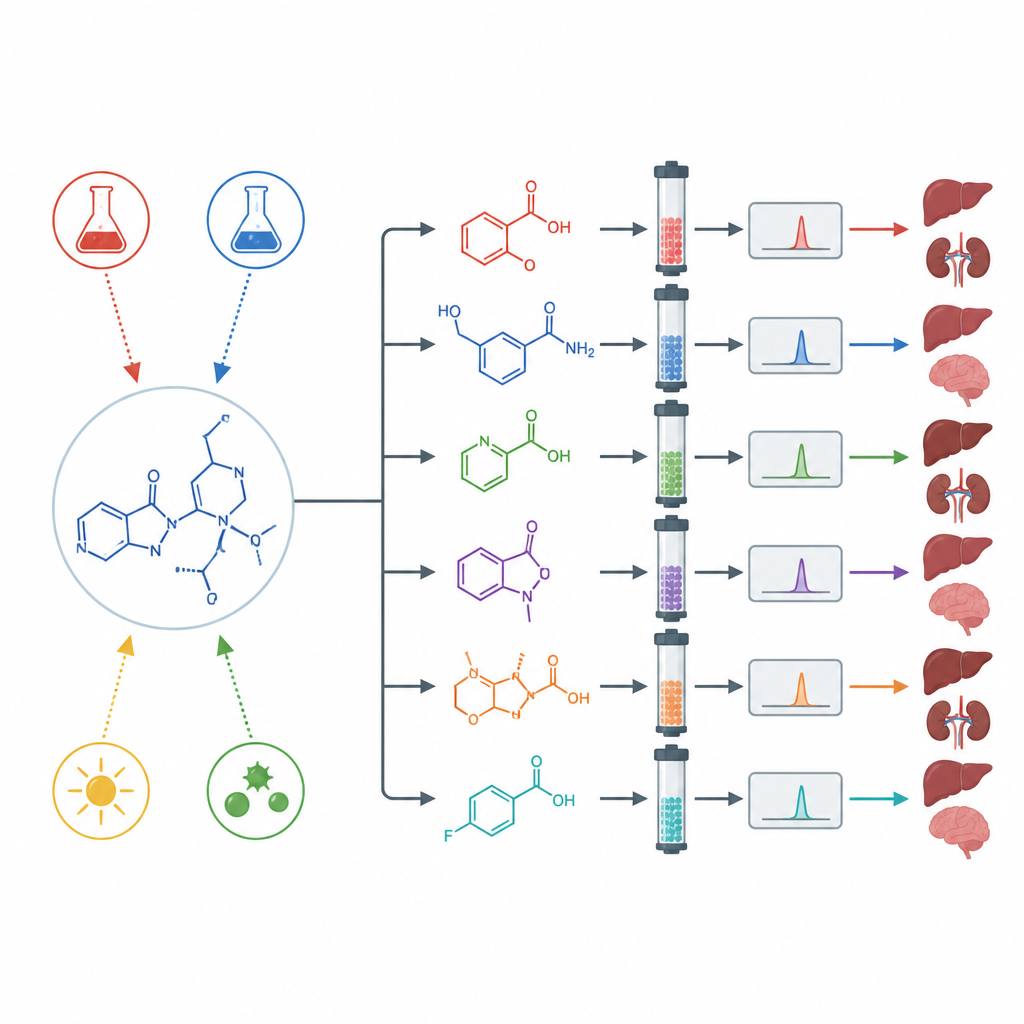
Stressing the drug to reveal hidden by-products
To see how asciminib behaves under extreme conditions, the scientists performed “forced degradation” tests. They exposed the drug to strong acid, strong base, oxidizing chemicals, heat, water, and bright light including ultraviolet. Under neutral water, heat, and normal light, the drug barely changed. But under acid, base, oxidizing agents, and ultraviolet light, it broke down noticeably and formed several distinct new molecules. Using preparative separation, high-resolution mass spectrometry, and nuclear magnetic resonance, the team isolated and worked out the structures of multiple degradation products formed in each type of stress.
Peeking at safety and behavior with computer models
Because testing every breakdown product in animals or people is slow and expensive, the researchers turned to in silico tools that estimate toxicity and drug-like behavior from chemical structure. These programs suggested that all the identified degradation products fall into a moderate toxicity band, but their predicted risks differ. Some showed signals of possible liver, kidney, or nervous system effects, while others looked milder. Several were flagged for potential cancer-causing or gene-damaging activity, and their ability to move through the body and cross into the brain varied widely. Although these predictions are not proof of harm, they highlight which impurities deserve the closest experimental scrutiny.
What this means for patients and drug makers
For people taking asciminib, the study does not change how the drug is used, but it does strengthen the safety net around it. The authors show that asciminib is quite stable in normal storage but can form a handful of distinctive breakdown products under harsher conditions. They provide a detailed lab method to watch for these molecules and initial computer-based clues about which ones might be more worrying. Together, this work helps manufacturers and regulators track the long-term safety of asciminib and offers a template for evaluating other cancer drugs as they age.
Citation: Shaik, R.B., Padala, S.K.R., Gupta, M. et al. Structural and In silico safety evaluation of asciminib degradation products with a validated stability indicating HPLC method for genotoxic impurity determination. Sci Rep 16, 14965 (2026). https://doi.org/10.1038/s41598-026-44693-0
Keywords: asciminib, drug degradation, genotoxic impurities, HPLC analysis, in silico toxicity